Perianal and periumbilical dermatitis: Report of a woman with group G streptococcal infection and review of perianal and periumbilical dermatoses
Main Content
Perianal and periumbilical dermatitis: Report of a woman with group G streptococcal infection and review of perianal and periumbilical
dermatoses
Joseph R Kallini1 BS, Philip R Cohen2,3,4 MD
Dermatology Online Journal 19 (4): 3
1. Medical School, Baylor College of Medicine, Houston, Texas2. The University of Houston Health Center, University of Houston, Houston, Texas
3. Department of Dermatology, University of Texas Medical School at Houston, Houston, Texas
4. Department of Dermatology, The University of Texas MD Anderson Cancer Center, Houston, Texas
Abstract
PURPOSE: We describe a woman with perianal and periumbilical dermatitis secondary to group G Streptococcus, summarize the salient features of this condition, and review other cutaneous conditions that clinically mimic streptococcal dermatitis of the umbilicus. BACKGROUND: Periumbilical and perianal streptococcal dermatitis are conditions that commonly occur in children and usually result from beta-hemolytic group A Streptococcus. Rarely, non-group A streptococcal and staphylococcal infections have been reported in adults. MATERIALS AND METHODS: A 31-year-old woman developed perianal and periumbilical group G streptococcal dermatitis. Symptoms were present for six months and were refractory to clotrimazole 1 percent and betamethasone dipropionate 0.05 percent cream. RESULTS: The etiology of perianal and periumbilical dermatitis is unclear, but is perhaps explained by virulence of previously asymptomatic colonized bacteria. Perianal streptococcal dermatitis is more common in children. A number of adult infections have been reported, most of which were secondary to group A beta-hemolytic Streptococcus. Men are more often affected than women. Group G Streptococcus is rarely the infective etiology of perianal streptococcal dermatitis. This condition presents as a superficial well demarcated erythematous patch on clinical examination. Diagnosis is ascertained by diagnostic swabs and serological tests: antistreptolysin O (ASO) or anti-DNase titer. Treatments include oral amoxicillin, penicillin, erythromycin, and mupirocin ointment. CONCLUSIONS: Our patient expands on the clinical presentation typical of streptococcal dermatitis. We describe a rare occurrence of an adult woman infected with non-group A Streptococcus. Several conditions can mimic the presentation of perianal streptococcal dermatitis. Although rare, group G Streptococcus should be considered in the setting of virulent infections usually attributed to group A species. Streptococcal dermatitis can be added to the list of conditions affecting the umbilicus.
Introduction
Periumbilical and perianal streptococcal dermatitis is more commonly observed in children and usually results from beta-hemolytic group A streptococcal infection. Omphalitis may be caused by various bacterial pathogens. We report a woman with periumbilical and perianal beta-hemolytic group G streptococcal infection, summarize the differential diagnosis of perianal streptococcal dermatitis, and review other cutaneous conditions that clinically mimic streptococcal dermatitis of the umbilicus.
Case report
A 31-year-old woman experienced vulvar pruritus for six months and subsequently developed a periumbilical, perianal, and vulvar rash. She was initially seen in a Woman’s Clinic with a presumed diagnosis of candidal intertrigo versus allergic contact dermatitis. She was prescribed a combination cream consisting of clotrimazole 1 percent and betamethasone dipropionate 0.05 percent (Lotrisone cream). She returned for evaluation four months later with no resolution of symptoms.
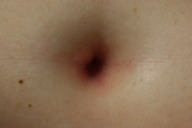 |
| Figure 2c |
|---|
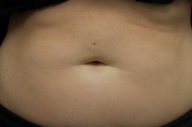
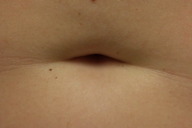
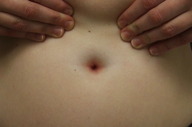
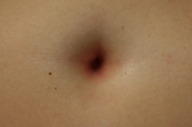
Cutaneous examination showed confluent erythematous patches in the umbilicus, anus, and labia majora. The clinical differential diagnoses included partially treated psoriasiform dermatitis and a bacterial infection. She was prescribed clobetasol propionate 0.05 percent ointment twice daily to the affected sites. Swabs of the affected regions were also cultured for bacteria.
Beta-hemolytic group G Streptococcus grew from both the umbilicus and the anus, confirming the diagnosis of periumbilical and perianal streptococcal dermatitis. The patient was prescribed erythromycin 500 mg four times daily for 10 days because she was allergic to penicillin. She cleaned the affected areas with chlorhexidine 4 percent solution once daily and applied mupirocin 2 percent ointment three times daily to the sites. She stopped the clobetasol ointment. After one week of this regimen, the patient no longer experienced pruritus and erythema; repeat bacterial cultures from the umbilicus and anus only showed normal skin flora.
Discussion
Perianal dermatitis was originally known as perianal cellulitis prior to the 1990s. The old term was unfortunately misleading owing to the contrasting clinical manifestations of this disease [1]. Unlike conventional bacterial cellulitis (typically warm, tender plaques with ill-defined and advancing borders), perianal dermatitis is often characterized by an erythematous, sharply demarcated, pruritic lesion. The condition has also been termed perianal streptococcal disease because beta-hemolytic group A Streptococcus is the most common etiology of perianal dermatitis in infants and children. Non-group A beta hemolytic Streptococcus and Staphylococcus aureus species are also responsible pathogens for a significant number of individuals [2, 3, 4].
Group G Streptococcus species is a rare pathogenic etiology for perianal streptococcal dermatitis. Indeed, only a few individuals have been reported as having this organism as the cause of their condition (Table 1) [5]. Group G Streptococci are similar to group A and B strains in that they are beta-hemolytic. Group G can be either bacitracin resistant (like most group B strains) or sensitive (similar to group A organisms). Thus, group G organisms mimic group A and group B pathogens with many current biochemical analyses. In addition, the M protein (a moiety that confers virulence) of group G matches those of some group A streptococcal strains that cause rheumatic fever. Group G also has a cross-reactive streptolysin O, so infection can cause anti-streptolysin O titers to rise similar to the phenomenon observed with group A streptococcal infection [6].
Infection with Group G Streptococcus can be associated with severe bacteremia; this is attributed to the presence of the M protein [7]. The pathogen has also been shown to trigger a “toxic-shock-like syndrome” similar to group A organisms [8]. Indeed, Brandt and Spellerberg have described a spectrum of clinical diseases secondary to group G Streptococcus that are similar to those caused by group A Streptococcus [9]. Group G Streptococcus has also been implicated in the development of abscesses, empyema, endocarditis, epiglottitis, exophthalmitis, fasciitis, impetigo, meningitis, necrotizing osteomyelitis, pharyngitis, pneumonia, puerperal fever, and septic arthritis in immunocompromised patients [6].
To the best of our knowledge, only a few reports of streptococcal dermatitis secondary to group G exist in the literature (Table 1). One group of investigators in Germany described two adult patients with perianal group G streptococcal dermatitis. One of the individuals also had a history of Crohn disease [5].
Bacteria, considered to be normal flora, may be present in the perianal and vulvar regions. The perineum is usually colonized by Escherichia coli, coagulase-negative Staphylococcus, micrococci, and a variety of aerobic coryneforms. Indeed, Acinetobacter species are also found in 15 percent of patients. Organisms regularly isolated from the vulva include coagulase-negative Staphylococcus, micrococcus, coryneforms, and enterococcus species; in addition, group B Streptococcus may be found in small numbers [10].
Perianal streptococcal dermatitis is either uncommon or underreported. Although the number of affected individuals may be greater than those found in the literature, only about 100 patients with perianal streptococcal dermatitis have been reported [2]. The mean age of patients with group A streptococcal infection is 3.8 years, whereas that of non-group A infection is 2.7 years [2]. Most patients are children between 6 months and 10 years old. More boys than girls have been reported. Although more common in children, a number of adults with this condition have also been described, most of whose perianal streptococcal disease was also secondary to group A beta-hemolytic Streptococcus [11].
The pathophysiology of perianal dermatitis is poorly understood. Bacterial species may descend from the throat to distal portions of the gastrointestinal tract. Evidence that supports this hypothesis is the discovery that 53.8 percent of patients with group A perianal Streptococcus and 51.9 percent of non-group A perianal Streptococcus had associated pharyngeal colonization [2]. However, asymptomatic throat colonization is more rare in adults than children. Therefore, local trauma and repeated use of detergents that diminish the lipid barrier of the skin may also play a role in infectivity [12].
Signs and symptoms of perianal streptococcal disease include perianal erythema, edema, maceration, soreness, pruritus, difficult defecation, anal leakage and soiling, mucoid discharge, bleeding, constipation, and sometimes even proctocolitis [12, 13]. The duration of symptoms can range from one month to one year.
Cutaneous examination shows a superficial cutaneous erythematous patch, which has clearly defined borders [12]. However, several patients have exhibited psoriasiform dermatitis; one patient not only had a “beefy-red,” psoriasiform eruption with superficial crusting, but also an associated digital desquamation [14]. Several familial cases have been reported [15].
The clinical differential diagnoses of perianal streptococcal disease are listed in Table 2 [1-5]. Because the symptoms of this condition are nonspecific, patients are frequently misdiagnosed and incorrectly treated with topical antifungal agents, topical corticosteroids, and even systemic antiparasitic medications [16]. In addition, perianal lichen sclerosis, flexural psoriasis, guttate psoriasis, and intertrigo have been reported to be associated with perianal dermatitis, which further complicates the ability to establish the correct diagnosis [12, 14-17].
The umbilicus is the most commonly colonized region in neonates. It is often colonized by S. aureus and occasionally colonized by beta-hemolytic group A Streptococcus shortly after birth. Indeed, these organisms may spread from infant to infant in hospital nurseries [10].
Group B Streptococcus may also colonize the newborn. However, this usually occurs in the setting of positive vaginal cultures in the mother. One study found that 69.2 percent of newborns delivered vaginally were colonized by group B Streptococcus when maternal vaginal and/or amniotic fluid cultures were positive, whereas only 5.6 percent of the newborns were colonized by this species in the setting of negative maternal cultures [18].
Although perianal streptococcal dermatitis is more frequently described in the literature, there are some notable reports of individuals with analogous periumbilical bacterial infection. One report describes massive growth of hemolytic group C Streptococcus in the umbilicus of a 32-year-old woman. The patient presented with extreme periumbilical pain exacerbated by movement. Cutaneous examination demonstrated erythematous, exudative skin and grayish malodorous pus appearing as a ring [19].
Another study described an outbreak of S. aureus infections following discontinuation of antiseptic cord care on a neonatal unit. The most common site of infection was the umbilicus. The study not only showed one cluster of group A Streptococcus, but also two clusters of methicillin-resistant S. aureus [20].
Several cutaneous conditions can affect the umbilicus; several of these can also clinically mimic periumbilical streptococcal dermatitis (Table 3) [21, 22, 23]. Diagnostic swabs for microbiological analysis are recommended. Serological tests, such as the antistreptolysin O (ASO) test or anti-DNase titer, can also be helpful in the diagnosis. However, a negative finding does not exclude Streptococcus, especially local cutaneous infections [5].
Early antibiotic treatment results in dramatic and rapid improvement of streptococcal disease. The treatment of choice for children is amoxicillin 40 mg/kg/day, divided into three doses [15]. For adults, intramuscular or oral penicillin may suffice. Erythromycin is a suitable alternative in patients such as ours who are allergic to penicillin [14]. In addition to systemic medication, adjuvant topical therapy may be incorporated: chlorhexidine 4 percent solution for cleaning the affected area and/or application of mupirocin 2 percent ointment [15]. Treatment options for group G Streptococcus are similar to group A species [5].
Conclusion
Beta-hemolytic group A Streptococcus is the most common etiology of perianal dermatitis in infants and children, although a significant number of individuals have non-group A beta hemolytic Streptococcus or S. aureus as a causative pathogen. Group G Streptococcus species is rare; only a few individuals have been reported. The pathophysiology of perianal dermatitis is poorly understood. It has been proposed that bacteria may descend from the throat throughout the gastrointestinal tract. Signs and symptoms of perianal streptococcal disease include perianal erythema, edema, maceration, soreness, pruritus, difficult defecation, anal leakage and soiling, mucoid discharge, bleeding, constipation, and occasionally proctocolitis. Patients are frequently misdiagnosed and incorrectly treated because of the nonspecific symptoms of this condition. Moreover, periumbilical streptococcal dermatitis is even more rare. Diagnostic swabs for microbiological analysis are recommended. Serological tests, such as the antistreptolysin O (ASO) test or anti-DNase titer, can also be helpful in the diagnosis. Our patient expands both the currently reported epidemiology and clinical presentation of streptococcal dermatitis because she is an adult woman infected with non-group A Streptococcus. Several conditions can mimic the presentation of streptococcal disease. Although rare, group G Streptococcus should be considered in the setting of virulent infections usually attributed to group A species. Streptococcal dermatitis can be added to the list of conditions affecting the umbilicus.
References
1. Krol AL: Perianal streptococcal dermatitis. Pediatr Dermatol 1990;7:97-100. [PubMed]2. Mostafa WZ, Arnaout HH, El-Lawindi MI, Zein El-Abidin YM: An epidemiologic study of perianal dermatitis among children in Egypt. Pediatr Dermatol 1997;14:351-354. [PubMed]
3. Teillac-Hamel D, De Prost Y: Perianal streptococcal dermatitis in children. Eur J Dermatol 1992;2:71-74.
4. Heath C, Desai N, Silverberg NB: Recent microbiological shifts in perianal dermatitis: Staphylococcus aureus predominance. Pediatr Dermatol 2009;26:696-700. [PubMed]
5. Scheiba N, Hartschuh W: Perianal streptococcal dermatitis caused by β-hemolytic group G streptococci in two adults [in German]. Hautarzt. 2011;62:131-133. [PubMed]
6. Vartian C, Lerner PI, Shlaes DM, Gopalakrishna KV: Infections due to Lancefield group G streptococci. Medicine 1985;64:75-88. [PubMed]
7. Cohen-Poradosu R, Jaffe J, Lavi D, Grisariu-Greenzaid S, Nir-Paz R, Valinsky L, Dan-Goor M, Block C, Beall B, Moses AE. Group G streptococcal bacteremia in Jerusalem. Emerg Infect Dis. 2004 Aug;10(8):1455-60. [PubMed]; PMCID: PMC3320404.
8. Bomke AK, Vagts DA, Podbielski: Toxic-shock-like syndrome caused by β-hemolytic Streptococcus group G with a multi-morbid patients with erysipelas [in German]. Dtsch Med Wochenschr 2006;131:263-266. [PubMed]
9. Brandt CM, Spellerberg B: Human infections due to Streptococcus dysgalactiae subspecies equisimilis. Clin Infect Dis 2009;49:766-772. [PubMed]
10. Chiller K, Selkin BA, Murakawa GJ: Skin microflora and bacterial infections of the skin. J Invest Dermatol Symp Proc 2001;6:170-174. [PubMed]
11. Brazilai A, Choen HA: Isolation of group A streptococci from children with perianal cellulitis and from their siblings. Pediatr Infect Dis J 1998;17:358-360. [PubMed]
12. Neri I, Bardazzi F, Marzaduri S, Patrizi A: Perianal streptococcal dermatitis in adults. Br J Dermatol 1996;135:796-798. [PubMed]
13. Barnett BO, Frieden IJ: Streptococcal diseases in children. Sem Dermatol 1992;11:3-10. [PubMed]
14. Nowicki MJ, Bishop PR, Parker PH: Digital desquamation--a new finding in perianal streptococcal dermatitis. Clin Pediatr (Phila) 2000;39:237-239. [PubMed]
15. Rasi A, Pour-Heidari N: Association between plaque-type psoriasis and perianal Streptococcal cellulitis and review of the literature. Arch Iran Med 2009;12:591-594. [PubMed]
16. Kokx NP, Comstock JA, Facklam RR: Streptococcal perianal disease in children. Pediatrics 1987;80:659-663. [PubMed]
17. Ledoux M, Chazerain V, Saiag P, Mahé E: Streptococcal perianal dermatitis and guttate psoriasis. Ann Dermatol Venereol 2009;136:37-41. [PubMed]
18. Matorras R, Garcia-Perea A, Usandizaga JA, Omeñaca F: Natural transmission of group B Streptococcus during delivery. Int J Gynaecol Obstet 1989;30:99-103. [PubMed]
19. Juhlin L, Glade C: A woman with umbilical inflammation continuing for 9 years after delivery. J Am Acad Dermatol 2001;44:687-688. [PubMed]
20. Allen KD, Ridgway EJ, Parsons LA: Hexachlorophane powder and neonatal staphylococcal infection. J Hosp Infect 1994;27:29-33. [PubMed]
21. Chaitra V, Rajalakshmi T, Mohanty S, Lahoti NK, George A, Idiculla J: Actinomycosis in urachal remnants: A rare cause of pseudotumor. Indian J Urol 2011;27:545-546. [PubMed]
22. Cohen PR, Robinson FW, Gray JM. Omphalith-associated relapsing umbilical cellulitis: recurrent omphalitis secondary to a hair-containing belly button bezoar. Cutis. 2010;86:199-202. [PubMed]
23. Sarma DP, Teruya B. 'Lint ball' omphalitis, a rare cause of umbilical discharge in an adult woman: a case report. Cases J. 2009;2:7785. [PubMed]
© 2013 Dermatology Online Journal