Eosinophilic fasciitis secondary to intravenous iron infusions
Published Web Location
https://doi.org/10.5070/D39sm798wcMain Content
Eosinophilic fasciitis secondary to intravenous iron infusions
Bahar F Firoz MD MPH1,2, Leonard H Goldberg MD FAAD1,2, Jennifer Landau BS1, Valda Kaye MD3, Louis Berman MD4
Dermatology Online Journal 16 (5): 5
1. Derm Surgery Associates, Houston, Texas. goldb1@dermsurgery.org2. Departments of Dermatology, Weill Cornell Medical College, Methodist Hospital, Houston, Texas
3. Twin Cities Dermatopathology, Plymouth, Minnesota
4. Rheumatology, Houston, Texas
Abstract
A 43-year-old African-American female with anemia secondary to uterine leiomyomas and menorrhagia presented with induration and stiffness of the right arm and hand four weeks after receiving intravenous iron infusions at multiple infusion sites along the right proximal forearm. Multiple intravenous sites between her right antecubital fossa and wrist had to be used because developing pain necessitated the site changes. The iron infusions were performed because the patient had refused blood transfusions and her symptoms failed to resolve on oral iron supplementation. The skin induration persisted and progressed for several months at which time a skin biopsy was performed. The skin histology was consistent with eosinophilic fasciitis and her complete blood count was notable for a peripheral eosinophilia. Because of the location of the fibrosis and the time proximity in relation to her infusions, a relationship between the iron infusions and eosinophilic fasciitis was made. Cutaneous fibrosis has been linked to immunologic dysfunction, autoantibody production, tissue hypoxia, and vascular damage, which may have been contributing factors in this patient. Eosinophilic fasciitis has been linked to certain drugs and chemicals, notably L-tryptophan ingestion and the statin family of drugs.
Observation
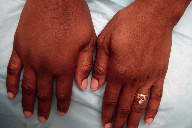 | 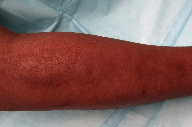 |
| Figure 1 | Figure 2 |
|---|
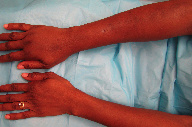 |
| Figure 3 |
|---|
A 43-year-old woman presented with edema of the hand, soft-tissue swelling, joint stiffness, and intermittent and had a peau d’orange appearance with overlying hyperpigmented, mottled patches. These changes had been present for approximately four months and the edema and induration continued to progress up the right proximal forearm toward the right shoulder (Figures 1, 2, and 3). The left upper extremity was unchanged.
The patient’s past medical history was notable for uterine leiomyomas, menorrhagia with heavy uterine bleeding, and anemia with a hemoglobin as low as 7 g/dL. The patient refused blood transfusions for the anemia although she was symptomatic with hypotension and syncope unresponsive to oral iron supplementation. Because of these persistent symptoms, she was started on intravenous iron sucrose infusions (Venofer). Venofer consists of an aqueous complex of polynuclear iron (III) – hydroxide in sucrose (20 mg elemental iron/mL) and contains no dextran (a glucose polymer) or modified dextran. Multiple IV sites had to be used between the right antecubital fossa and the right wrist because of pain during the infusions. The patient subsequently developed phlebitis after intravenous iron administration that was treated with oral non-steroidal anti-inflammatory medications.
Approximately one month after the infusions, the patient began to develop edema, pain, and the overlying skin changes noted above. The changes continued to progress despite the cessation of intravenous therapy.
Lab results
The following studies were obtained. Antinuclear antibody was positive at 1:162, with a centromere pattern and the sedimentation rate was 40 mm/Hr (0-20). A complete blood count was notable for a peripheral eosinophilia of 8.8 percent (0-7%). A routine chemistry panel, liver function panel, thyroid function panel, urinalysis, anti-Smith, anticardiolipin, creatine kinase, aldolase, antiscleroderma-70, anti-Ro, anti-La, and anti-ribonucleoprotein were within normal limits. Age appropriate cancer screening was negative.
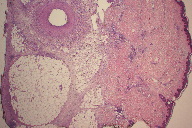 |  |
| Figure 4 | Figure 5 |
|---|
 |  |
| Figure 6 | Figure 7 |
|---|
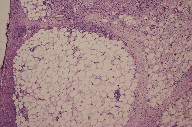
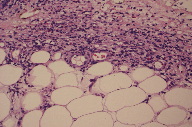
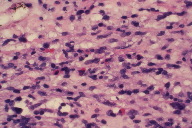
Two skin biopsies of the right forearm were obtained. The first exhibited prominent dermal fibrosis suggestive of scar. Minimal chronic inflammation was present. The second biopsy was an elliptical wedge incision to the fascia of the distal forearm, which showed an inflammatory reaction associated with septal widening and edema from the deeper dermis extending into the subcutaneous fat. The biopsies revealed a thickening of the interlobular fibrous septae with an associated infiltrate of lymphocytes, histiocytes, plasma cells, and eosinophils. These factors were also associated with a thickening of the deep tissue and extension into the deep dermis. Based on the histologic evaluation of the biopsies (Figures 4 through 7) a diagnosis of eosinophilic fasciitis was made.
X-rays of the right upper extremity were performed which were within normal limits with a notable absence of carpal tunnel syndrome or radiculopathy. A venous ultrasound of the right upper extremity was also performed, and normal flow was identified in the right jugular, subclavian, ulnar and radial veins. It was noted that the area of tenderness in the right mid-upper arm did not involve deeper tissue or vascular structures.
Discussion
Eosinophilic fasciitis is a clinical entity first reported by Shulman in 1975 and is recognized as a variant of morphea or scleroderma [1, 2]. Initially, patients describe edema and pain of the involved extremities, which is usually symmetric but spares the hands, feet, and face. The etiology is unknown, although it has been associated with strenuous physical activity in 30 percent of patients. Other potential factors/causes include systemic drugs, trauma, arthropod bites, and borreliosis. A recent review of eosinophilic fasciitis in twelve patients over ten years evaluated characteristics of the disease [3]. All patients had skin induration, whereas seven displayed peau d’orange and three had morphea. Only four patients’ symptoms were exercise induced. Upper extremity involvement was seen in nine patients. Two patients had concomitant malignancy, specifically chronic lymphocytic leukemia and breast cancer. Two patients exhibited a monoclonal gammopathy, and two exhibited positive anti-nuclear antibodies, like our patient.
It does seem more than plausible that this patient’s eosinophilic fasciitis is related to the intravenous iron infusions because the disease onset was 3 weeks after the infusions and the changes were localized to the proximal forearm, the site of all of the infusions. According to the package insert for Venofer, the most commonly reported side effects are changes in taste, low blood pressure, fever, shivering, injection site reactions, and nausea. Non-serious allergic reactions rarely occur. Skin disorders were described as uncommon but included itching, hives, rash, and redness. Isolated cases of joint swelling and muscle cramps and muscle pain are also described. An analysis of FDA data from 2001 to 2003 revealed few complications resulting from iron sucrose infusions with Venofer [4]. Of 8,837,000 patients receiving Venofer, 18 had an allergic reaction. None had an anaphylactoid reaction, 1 had a fatal reaction, 12 had hypotension, 10 had nausea, and 10 had dyspnea (other complications were noted in fewer than 10 people) [4]. It is possible that this patient may have suffered local tissue hypoxia associated with the trauma following repeated failed intravenous attempts and/or persistent inflammation because of phlebitis. These factors, including the iron itself, may have stimulated the patient’s immunologic response. Additionally, since these compounds are intended to be long acting, their introduction into the subcutaneous tissue could have produced a prolonged reaction.
To safely administer iron intravenously, it is injected as a complex completely surrounded by sucrose, a stabilizing carbohydrate [5]. The iron-carbohydrate complex decreases the rate at which the iron is released to bind to ferritin or transferrin, and, ultimately, to hemoglobin [5]. Sucrose is not metabolized and is eliminated unchanged in the urine, which can lead to increased excretions of albumin, enzyme (N-acetyl-β-glucosaminidase), and cytokine (chemokine monocyte chemoattractant protein-1) [6]. Iron sucrose infusions are believed to cause untoward complications such as inflammation, endothelial dysfunction, and renal injury [6]. The FDA has not reported any side effects relating to the sucrose in Venofer.
In order to confirm the link between these two events causally, it would be necessary to re-challenge the patient with another infusion in the opposite arm, which of course was not done. Systemic immunosuppression including prednisone, mycophenolate mofetil and methotrexate were initiated in this patient with improved joint mobility and reduced edema and pain.
Eosinophilic fasciitis laboratory results usually include an elevated sedimentation rate, peripheral eosinophilia, and hypergammaglobulinemia. The degree of eosinophilia does not necessarily correlate with disease severity and is not a useful marker of treatment response or disease activity [3]. The disease has also been associated with myeloproliferative disorders, paraneoplastic conditions, and pancytopenia, anemia, and thrombocytopenia [7]. Our patient had anemia due to heavy uterine bleeding that predated the eosinophilic fasciitis and resolved with the intravenous iron infusions. It is difficult to exclude the possibility that her anemia and eosinophilic fasciitis were related. This patient did have normal age-appropriate cancer screening tests and no evidence of a gammopathy.
Treatment of eosinophilic fasciitis is usually initiated with oral corticosteroids and a response is usually seen within a few weeks to months. If oral corticosteroids do not elicit a response, other immunosuppressive medications such as hydroxychloroquine, methotrexate, dapsone, cyclosporine, cyclophosphamide, PUVA or infliximab may be used alone or in combination [8, 9, 10, 11].
Immunologic dysfunction, autoantibody production, vascular damage, and increased collagen production have been implicated in the pathogenesis of cutaneous fibrosis [12, 13, 14, 15]. Hypoxia has been linked to the production of profibrotic cytokines, fibroblast activation, and collagen production. Recent research has also focused on transforming growth factor-beta (TGF-beta) and platelet derived growth factor (PDGF) and their stimulatory effects on fibroblasts. Future therapeutics may target such pathways to help inhibit or reverse fibrosis.
Certain chemicals and drugs have been linked to a wide spectrum of cutaneous fibrosis syndromes including eosinophilic fasciitis. Eosinophilic fasciitis has been linked to ingestion of L-tryptophan and the statin family of drugs [16, 17, 18]. Two case reports describe the development of eosinophilic fasciitis approximately one week after starting atorvastatin and eighteen months after starting simvastatin. Eosinophilia-myalgia syndrome, characterized by an acute phase of fever, myalgias, morbilliform eruption, and peripheral eosinophilia occurred as a result of contaminated L-tryptophan intake. Unfortunately even after the discontinuation of L-tryptophan, about half of the patients progressed to a chronic phase with sclerodermoid induration of the extremities and progressive peripheral neuropathy and myopathy [2]. It is not unlikely that intravenous iron may also be associated with eosinophilic fasciitis. We feel it is important to describe this to increase awareness regarding a potential relationship between intravenous iron and eosinophilic fasciitis.
References
1. Shulman LE. Diffuse fasciitis with eosinophilia: a new syndrome? Trans Assoc Am Physicians 1975;88:70-86. [PubMed]2. Connolly MK. Systemic Sclerosis (Scleroderma) and Related Disorders In: Bolognia JL, Jorizzo JL, Rapini RP, editors. Dermatology. London England: Elsevier; 2008. p. 591-592.
3. Bischoff L, Derk CT. Eosinophilic fasciitis: demographics, disease pattern and response to treatment: report of 12 cases and review of the literature. Int J Dermatol. 2007;47(1):29-35. [PubMed]
4. Chertow GM, Mason PD, Vaage-Nilsen O, et al. Update on adverse drug events associated with parenteral iron. Nephrol Dial Transplant. 2006;21:378-382.
5. Danielson BG. Structure, chemistry, and pharmacokinetics of intravenous iron agents. J Am Soc Nephrol. 2004;15 Suppl 2:S93-8.
6. Bishu K, Agarwal R. Acute injury with intravenous iron and concerns regarding long-term safety. Clin J Am soc Nephrol. 2006 Sep; 1 Suppl 1:S19-23.
7. Philpott H, Hissaria P, Warren L, et al. Eosinophilic fasciitis as a paraneoplastic phenomenon associated with metastatic colorectal carcinoma. Australas J Dermatol. 2008;49(1):27-29. [PubMed]
8. Haiduc VF, Erkan D, Kirou K, et al. Anti-neutrophil cytoplasmic antibody (c-ANCA) positive recurrent eosinophilic fasciitis responsive to cyclophosphamide: a clinical pathology conference held by the division of rheumatology at hospital for special surgery. HSS J. 2008;4(1):81-86. [PubMed]
9. Smith LC, Cox NH. Dapsone treatment for eosinophilic fasciitis. Arch Dermatol. 2008;144(7):845-847. [PubMed]
10. Tahara K, Yukawa S, Shoji A, et al. Long-term remission by cyclosporine in a patient with eosinophilic fasciitis associated with primary biliary cirrhosis. Clin Rheumatol. 2008;27(9):1199-201. [PubMed]
11. Tzaribachev N, Holzer U, Schedel J, et al. Infliximab effective in steroid-dependent juvenile eosinophilic fasciitis. Rheumatology 2008;67 (4):572-574. [PubMed]
12. Uitto J, Jimenez S. Fibrotic skin diseases: Clinical presentations, etiologic considerations, and treatment options. Arch Dermatol. 1990;126:661-664. [PubMed]
13. Falanga V, Qian SW, Danielpour D, et al. Hypoxia upregulates the synthesis of TGF-β1 by human dermal fibroblasts. J Invest Dermatol. 1991;97:634-637. [PubMed]
14. Falanga V, Zhou L, Yufit T. Low oxygen tension stimulates collagen synthesis and COL1A1 transcription through the action of TGF-β1. J Cell Physiol. 2002;191:42-50. [PubMed]
15. Border WA, Noble NA. Transforming growth factor- β in tissue fibrosis. N Engl J Med. 1994;331:1286-1292. [PubMed]
16. Noakes R, Spelman L, Williamson R. Is the L-tryptophan metabolite quinolinic acid responsible for eosinophilic fasciitis? Clin Exp Med. 2006;6(2):60-64. [PubMed]
17. DeGiovanni C, Chard M, Woollons A. Eosinophilic fasciitis secondary to treatment with atorvastatin. Clin Exp Dermatol. 2006;31(1):131-132. [PubMed]
18. Choquet-Kastylevsky G, Kanitakis J, Dumas V, et al. Eosinophilic fasciitis and simvastatin. Arch Intern Med. 2001;161(11):1456-1457. [PubMed]
© 2010 Dermatology Online Journal

