Evaluation of open-label topiramate for scar therapy
Published Web Location
https://doi.org/10.5070/D38960f67rMain Content
Evaluation of open-label topiramate for scar therapy
Nathan A Shapira MD PhD, Mary Lessig BS, Tanya K Murphy MD, April M Annis JD BS, and Marty Lazoritz MD
Dermatology Online Journal 9 (5): 3
Department of Psychiatry, College of Medicine and the Evelyn F. and William McKnight Brain Institute, University of Florida,
Gainesville, Florida. shapira@psychiatry.ufl.edu
Abstract
A 3-month open-label pilot study was carried out to assess the safety, tolerability, and effect of the antiepilectic topiramate on the cosmetic appearance of scars. Ten adult subjects with discolored or raised scars at least 2 years old were given an oral dosage of 15 mg per day of topiramate for 1 month. The dosage was then increased to 30 mg per day if there was minimal or no improvement. The safety of topiramate was assessed in this study by reviewing adverse events and vital signs. Efficacy outcomes included a Clinician Global Impression Scale (CGI) to document changes such as thickness and color. Digital photos were taken with consistent variables. In addition, two independent medical reviewers blindly reviewed the photos. The Rosenberg Self-Esteem Scale was done at each visit to measure patients' levels of self-esteem. Side effects were generally mild with the most common being language problems (n = 3) and sleep disturbances (n = 3). All subjects completed the study and experienced at least minimal thinning and decreased coloration (usually redness) of their scars. Based on CGI-assessment data at 3 months, two subjects were very much improved, four were much improved, and four had minimal improvement. One independent medical reviewer arranged before and after treatment picture sets for ten out of ten subjects. The other independent medical reviewer arranged before and after treatment pictures sets for nine out of ten subjects (both p-values < 0.025). The data indicate that topiramate may be a safe and effective treatment for scar therapy.
Introduction
The formation of and healing of scars has been studied for over 2,000 years, beginning in China [1]. However, there continues to be a need for new treatments as the current techniques and technologies offer inconsistent and often limited results [2]. Current treatments for scars include cryosurgery, dermabrasion, laser camouflage, physical therapy, radiotherapy, silicon gel sheets, steroid injections, surgery, tissue implantation, and topical products (such as onion extract gel) [3].
In a previous open-label pilot trial of topiramate in adult Prader-Willi syndrome (PWS) subjects evaluated for appetite regulation, we had observed decreased skin picking and apparent improved healing of open lesions and scars [4]. After initiating topiramate, the skin surrounding the lesions appeared less red and swollen, and established scars (age > 2 years) appeared lighter in appearance (Shapira, unpublished data). We observed this effect occurring within a few months, even at low doses of topiramate (25-50 mg/day). PWS is a genetic disorder characterized by mental retardation, hyperphagia (with a high risk of obesity) and self-injurious behavior (SIB) (such as skin picking) [5]. In light of our findings in PWS, we hypothesized that topiramate may also be effective in scar therapy in individuals without SIB.
To determine whether topiramate is effective in scar therapy, we conducted an investigational pilot study in subjects with raised or discolored scars that were at least 2 years old to minimize any natural occurring improvement. We evaluated subjects' scars with a Clinician Global Impression Scale (CGI), and two independent medical reviewers blindly reviewed digital pictures of the scars.
Methods
This pilot trial was a 3-month, open-label, flexible-dose study where the efficacy and safety of topiramate was tested in patients with scars that were both raised or discolored and at least 2 years old. Digital pictures were taken of subjects scars with an 8 × 10-inch 18-percent gray card (CPM, Inc., Dallas, Texas) to match lighting and conditions. Additionally, half the pictures were taken with a ½ ×1¾-inch white label (Avery, Pasadena, California) placed near the scar to aid in determining color and sharpness. The pictures were taken with the same camera, a Canon PowerShot G2 (Canon, Inc., Tokyo, Japan), with the same distance between the camera and the scars at each visit. The camera was set up in macro mode, with the same camera stand and a black drop cloth for Caucasian subjects and an off-white drop-cloth for the one African-American subject. In order to minimize shadows, the camera system was activated by RemoteCapture (Canon Inc., Tokyo, Japan) directly into the computer as a JPEG file. The image size was 2272 × 1704 pixels, and the image quality was large and superfine. The lighting remained consistent throughout the study, with the same GE Photobulbs (General Electric Company,-USA, Fairfield Connecticut) and the same distance between the lighting and the subjects. All pictures were taken in the same windowless room. The two independent medical reviewers viewed the pictures in Adobe Photoshop 6.0 (Adobe Systems Incorporated, Mountain Valley, California) and conducted a blinded review of matched pictures, and assigned them into categories of before and after.
Only patients who were between ages of 18-65, with at least 1 scar that was at least 2 years old and raised or discolored, were accepted in the study. Subjects were enrolled from August 14, 2002, to May 1, 2003. All subjects signed written, informed consent as approved by the Health Science Center Institutional Review Board of the University of Florida. Subjects were recruited by local advertising. Inclusion criteria included that the scars must have closed skin around them. Subjects could have obtained scars from various measures such as accidents, burns, lacerations, or surgery. Exclusion criteria included subjects who self-injured, women who were pregnant or lactating, women who were not taking contraceptive measures, participants who were displaying clinically significant suicidality or homicidality, a current or recent DSM-IV diagnosis of substance abuse or depression, clinically unstable diseases, participants on drugs that might interfere or interact with topiramate, participants with a history of kidney stones, and participants who were diagnosed with PWS. Out of the ten subjects, one was African-American, and nine were Caucasian. There were nine females and one male.
At baseline, the oral medication, topiramate, was given in a dosage of 15 mg/day with instruction to be taken in the evening. After 1 month on 15 mg/day, participants came in for their first month visit. At this visit the safety and tolerability of topiramate was reported, and, if there was none to minimal improvement, the dosage was adjusted to 30 mg/day for the next 2-month period barring side effects. If there was more than minimal improvement after 1 month, then the dosage was kept at 15 mg/day.
To test the safety of the drug topiramate, the following safety measures were established at screening, baseline, and 1-month and 3-month visits. A laboratory evaluation was conducted at screening, which included a pregnancy test, a blood screening, urinalysis, and an EKG if the subject was older than 40. Topiramate has been reported in healthy volunteers to have side effects, such as a decrease in attention and verbal fluency [6]. Thus, two scales, the Controlled Oral Word Association Test (COWAT) [7] and the Semantic Oral Word Association Test (SOWAT) [7], were conducted at every visit. In addition, the Rosenberg Self-Esteem Scale (RSE) was conducted at every clinical visit [8, 9]. These data were analyzed by repeated pairwise t-test comparing baseline, and 1-month and 3-month values.
Scar characteristics were documented by digital picture. At every visit photographic documentation (digital pictures) were taken of the scars. The investigator (NAS) rated the CGI. The CGI was defined as no improvement (0 % to < 25 % decrease in scar characteristics such as thickness and coloration), minimally improved (25 % - < 50 %), much improved (50 % to < 75 %), or very much improved (75 % and above).
SPSS statistical software for Windows, version 9.0 (SPSS Inc., Chicago, IL) was used to perform the binomial test and the t-test.
Results
|
|
||
| Variable | Female (n = 9) | Male (n = 1) |
|---|---|---|
| Age, mean + SD (range), y | 31.1 + 12.0 | 32 |
| Ethnicity: |
|
|
| African-American | 1 | 0 |
| White | 8 | 1 |
| Types of scars: |
|
|
| Accidental | 4 | 0 |
| Acne | 1 | 0 |
| Burn | 1 | 0 |
| Chicken Pox | 1 | 0 |
| Laceration | 1 | 0 |
| Surgery | 8 | 1 |
| Vaccination | 1 | 0 |
| Previous nontherapeutic therapies: |
|
|
| Collagen Injection | 1 | 0 |
| Laser Treatment | 0 | 1 |
| Onion Extract Gel | 4 | 1 |
| Radiation therapy | 1 | 0 |
| Silicone patch | 2 | 0 |
| Steroids | 0 | 1 |
| Surgery | 2 | 0 |
| Vitamin-E | 1 | 0 |
|
|
||
Table 1 summarizes the key demographic information for this study. Of fifteen subjects with scars who completed screening and signed the informed consent, ten subjects met inclusion and exclusion criteria and initiated topiramate. All ten subjects completed the study. Five subjects were administered the dosage of 15 mg/day for 3 months. Four subjects were administered 15 mg/day for 1 month, and then the dosage was increased to 30 mg/day for the remaining 2 months. One subject initially increased to 30 mg/day after 1 month but subsequently decreased to 15 mg/day because of language problems. The mean ± SD daily dose of topiramate at the end of the study was 21 ± 7.7 mg/day.
Eight out of the ten patients enrolled in the study reported a total of 19 adverse events. Most were mild and transient. The most common side effects were language problems (n = 3), sleep disturbances (n = 3), anorexia (n = 2), and dizziness (n = 2) followed by one of each of the following: anhedonia, confusion, congestion, headache, muscle pain, nervousness, numbness, paresthesia, and pruritus. No side effects resulted in discontinuation of a subject. Only one subject (language problems) lowered the topiramate dose (from 30 mg/day to 15 mg/day).
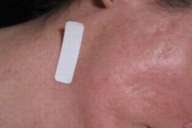
|
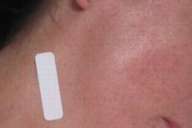
|
| Figure 1A | Figure 1B |
|---|---|
| Figure 1. Right side of face for 41 year-old woman with acne scars. A, Baseline visit. B, After 3 months of topiramate 15 mg/day. | |
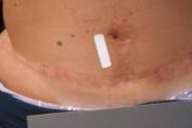
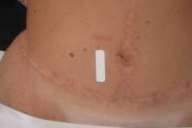
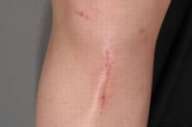
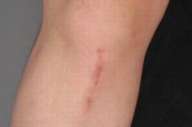
All subjects experienced at least minimal thinning and decreased coloration (usually redness) of their scars. Based on the investigator's CGI assessment data at 3 months, two subjects were very much improved, four were much improved, and four had minimal improvement. Figure 1 shows a 41-year-old female with acne scars. The right side of her face is shown at baseline (Fig. 1A) and at month 3 (Fig. 1B) after taking topiramate 15 mg/day throughout the study. Figure 2 shows scars on the abdomen of a 37-year-old female from gastric bypass surgery 8 years ago and an abdominoplasty 3 years ago. Her abdomen is shown at baseline (Fig. 2A) and at month 3 (Fig. 2B) of taking topiramate 15 mg/day for 1 month and 30 mg/day for 2 months. Figure 3 shows scars on a 34-year-old female from knee surgery of 3 years and 6 weeks age. Her right knee is shown at baseline (Fig. 3A) and at month 3 (Fig. 3B) after taking topiramate 15 mg/day for 1 month and 30 mg/day for 2 months.
One independent medical reviewer arranged before and after picture sets for ten out of ten subjects. The other independent medical reviewer correctly arranged before and after picture sets for nine out of ten subjects. Both p-values < 0.025 by binomial test indicate that the appearance of the scars changed significantly between the baseline and 3-month visit. In terms of safety assessments (weight, COWAT, SOWAT, and RSE), there were no significant differences.
Discussion
The results of this investigational pilot study, conducted in ten patients with scars that were raised or discolored, and at least 2 years old, suggest that the antiepileptic medication topiramate may be a novel and effective treatment for scar therapy. Among the ten participants in the study, topiramate was well tolerated. This is the first published study evaluating the effect of topiramate on the cosmetic appearance of scars, and we are unaware of any other oral medication that has shown efficacy in this area. Further controlled studies are needed to determine the optimal frequency and dose of topiramate for scar therapy.
Topiramate is an antiseizure medication that comes only in oral form [10]. Some participants indicated that previous use of topical medications had caused allergic reactions. Topiramate, designated chemically as 2,3:4,5-bis-O-( 1-methylethylidene )-β-D-fructopyranose sulfamate, is an antiepileptic medication approved by the Food and Drug Administration, for the treatment of epilepsy [11] in adults and children with partial onset seizures and primary generalized tonic-clonic seizures down to age 2 [10]. The doses (15 and 30 mg/day) utilized in this study were low compared to the recommended dose for topiramate as adjunctive therapy in epilepsy (400 mg/day in 2-divided doses) [10].
In this study, topiramate was well tolerated as evidenced by no subjects discontinuing, and only one subject decreasing her dose as the result of side effects. There were no significant changes for COWAT and SOWAT scores. Interestingly, there was also no significant change in self-esteem as measured by RSE, but this is likely a result of very high self-esteem (35.2 out of 40) described by participants at baseline. Additionally, topiramate has been noted in the treatment of epilepsy to be associated with decreased appetite and weight. This weight loss tends to be dose dependent and more prevalent in individuals who are overweight [11]. Although subjects lost a mean of 2.6 kg over the 3 months, it was not statistically significant, which could be the result of the small sample size, length of the trial, or low dose.
The mechanism by which topiramate may be effecting scar tissue is unknown. Several possible mechanisms of action have been identified for topiramate [11]: (1) facilitation of GABAergic activity at a nonbenzodiazepine site on γ-aminobutyric acid (GABAA) receptors; (2) antagonism of kainite/AMPA type glutamate receptors; and (3) state-dependent blockade of voltage-activated sodium channels.
In terms of possible direct effects of topiramate on skin, one recent open-label pilot study in eight subjects found that topiramate may ameliorate psoriasis [12]. Subjects were initially given a low dose of topiramate (usually 15 mg/day) and gradually titrated upwards each week typically by 15 mg/day to a mean final dose of 56 mg/day. Overall, subjects were treated for at least 4 months and experienced a mean reduction in their psoriasis area and severity index (PASI) from 16.1 to 7.1.
The optimal timing of topiramate for scar therapy is also unknown. It is interesting that one subject in our study experienced a reduction in 2 different scars, an older, colored, and raised laparoscopic right knee scar (3 years old), and a recent (6 week old) laparoscopic scar, (to the left of the older scar). Intriguingly, the most dramatic improvement occurred in the recent laparoscopic scar (Fig. 3). Therefore, it is possible that earlier administration of topiramate may afford better results than after a scar has been established. Four subjects have continued follow-up treatment of topiramate with continued thinning and lightening of their scar tissue. Furthermore, there has been no reported loss of positive scar changes by subjects who had received treatment with topiramate.
Because this study is a pilot study, the results should be considered with caution, as the number of subjects is small, and the study is not a controlled trial. Furthermore, other limitations included that there was only one male and one African-African, and that some subjects changed their tanning practices (either more or less) during the study (e.g., Fig. 2) or used moisturizer the day of their study visit on their scars. Use of moisturizer caused the skin to be more reflective of the lighting (e.g., Fig. 3A). However, positive improvements (such as raised scars becoming flush with the skin or scars becoming significantly less red) were not subtle as evidenced by the findings of the reviewers, particularly in subjects who experienced much or very much improvement. Additionally, seven of the ten subjects described that before topiramate they had failed various surgical or topical forms of scar therapy, and no subject described using any other treatments for scar therapy during the trial. Therefore, this pilot study demonstrates promising results for the therapeutic treatment of various scars. Future controlled studies are needed to address the safety and efficacy of topiramate on the cosmetic appearance of scar tissue and to determine whether earlier use after a wound occurs or other routes of administration (such as topical) would also have beneficial effects.
Acknowledgments/Disclosures: We thank Joanne A. Byars, B.S. for statistical help with this study. This study was supported by a grant from the Office of Technology Licensing, Research and Graduate Programs, University of Florida. Dr. Shapira is on the speaker's bureau for Ortho-McNeil and has also received grant/research support for unrelated research. Dr. Shapira and Ms. Lessig are on a patent application (PCT/US01/44923) relating to this study. Drs. Murphy, Lazoritz and Ms. Annis do not have any disclosures.
References
1. Fu X, Wang Z, Seng Z. Advances in wound healing research in China: from antiquity to the present. Wound Repair Regen. 2001 Jan-Feb;9(1):2-10. PubMed2. Wittenberg GP, Fabian BG, Bogomilsky, et al. Prospective, single blind, randomized, controlled study to assess the efficacy of the 585-nm flashlamp-pumped pulsed-dye laser and silicone sheeting in hypertrophic scar treatment. Arch Dermatol. 1999;135:1049-1055.
3. Menter, A, Niroomand, F. Wounds and Scars: Therapeutic Options. A Supplement to Drug Topics. 1998; 3:1-15.
4. Shapira NA, Lessig MC, Murphy TK, Driscoll DJ, Goodman WK. Topiramate attenuates self-injurious behaviour in Prader-Willi Syndrome. Int J Neuropsychopharmacol. 2002 Jun;5(2):141-5. PubMed
5. Martin A, Matthew S, Koenig K, et al. Prader-Willi syndrome. Am J Psychiatry. 1998;155,1265-1273.
6. Martin R, Kuzniecky R, Ho S, et al. Cognitive effects of topiramate, gabapentin, and lamotrigine in healthy young adults. Neurology. 1999;52,321-327.
7. Lezak, M. Nueropsychological assessment (2nd ed.), New York: Oxford University Press; 1996.
8. Shakespeare V, Postle K. A qualitative study of patients' views on the effects of breast-reduction surgery; a 2-year follow-up survey. Br J Plast. Surg. 1999;198-204.
9. Robert R, Meyer W, Bishop S, et al. Disfiguring burn scars and adolescent self-esteem. Burns. 1999;25(7):581-5.
10. Physicians Desk Reference(r), 2002 (c) Medical Economics Company, Inc. Montvale, NJ.
11. Privitera MD. Topiramate: a new antiepileptic drug. Ann Pharmacother. 1997 Oct;31(10):1164-73. Review. PubMed
12. Ryback R. Topiramate in the treatment of psoriasis: a pilot study. Br J Dermatol. 2002 Jul;147(1):130-3. PubMed
© 2003 Dermatology Online Journal