Nail toxicity related to taxanes
Published Web Location
https://doi.org/10.5070/D381h8p24mMain Content
Nail toxicity related to taxanes
Y Abou Mourad, M Matta-Muallem, and A Shamseddine
Dermatology Online Journal 9(3): 15
From the Department of Internal Medicine, Department of Dermatology, American University of Beirut-Medical Center, Beirut,
Lebanon. as04@aub.edu.lbAbstract
A 70-year-old woman with non-small-cell lung cancer developed severe nail toxicity while she was being treated with docetaxel at three-week intervals. Docetaxel is a chemotherapeutic agent of the taxane family. Taxanes are well known to cause nail changes, but mainly when used on a weekly basis.
Introduction
The taxanes, paclitaxel and docetaxel, are effective chemotherapeutic drugs used in the treatment of many malignant diseases including breast, lung, ovary, bladder, and prostate cancer. Taxanes are usually administered every 3-weeks, but weekly regimens are also used. Cutaneous toxicity, such as erythema and desquamation, has been reported with taxanes. Nail changes are also described in different series and case reports, usually when the weekly regimen is used or when these drugs are combined with other cytotoxic drugs. We report a case in which severe nail toxicity developed using docetaxel as a single agent on an every 3-week regimen.
Case report
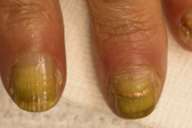 |
| Figure 1 |
|---|
| Nails demonstrating blackish discoloration, periungual erythema, and partial loss of nails (onycholysis). |
A 73-year-old woman had a confirmed diagnosis of non-small-cell lung cancer with mesenteric lymph node metastasis documented by fine needle aspiration in March 2000. She received first-line chemotherapy with Cisplatin and Vinorelbine with complete disappearance of metastatic disease. In April 2002, she was started on second-line chemotherapy with the single agent, docetaxel, as 80 mg/m every 3 weeks, because of the appearance of a new lesion involving the left adrenal gland. Shortly after starting docetaxel, she developed nail changes involving both hands and feet. Dyspigmentation and blackish discoloration of the nail plates were initially noted, followed by onycholysis along with periungual tenderness, swelling, and erythema (Fig. 1). With further continuation of docetaxel, partial loss of the nails with mild pain occurred but did not interfere with function (grade 2 according to the common toxicity criteria of the National Cancer Institute, version 2.0). Discontinuation of chemotherapy was not necessary, and the last cycle was given on December 31, 2002.
Discussion
Nail abnormalities are a common side effect of systemic chemotherapy. Taxanes probably cause nail changes more often than other drugs, and these changes include nail pigmentation, splinter hemorrhage and subungual hematoma, Beau lines, acute paronychia, and onycholysis. The common toxicity criteria of the National Cancer Institute (version 2.0) describes two grades of nail changes. Grade 1 describes discoloration, ridging (koilonychias), or pitting. Grade 2 describes partial or complete loss of a nail or pain in the nail bed.
Nail pigmentation results from toxic effects to the matrix epithelium, which results in melanocyte activation and melanonychia. The mechanism of splinter hemorrhage and subungual hematoma is not clear. One possible explanation is that these are due to taxane-induced thrombocytopenia and vascular abnormalities. Acute toxic damage to the proximal nail fold causes paronychia; whereas damage to nail bed and nail plate causes detachment with onycholysis. The most typical changes are Beau lines, which are signs of arrest in epithelial proliferation.
Spazzapan et al. reported nail toxicity related to weekly administration of taxanes. [1] A total of 58 patients were entered into a phase II trial of weekly paclitaxel in metastatic breast cancer using the Perez schedule. [1, 2] Nail toxicity occurred in 16 patients (27.6 %), mostly grade 2. In two cases, a chemotherapy delay was required in order to manage the nail toxicity, which caused limitation in function and pain. Perez et al. documented toxicity incidences of 17 percent (grade 1) and only 3 percent (grade 2).[2] Hainsworth et al. did not report nail disorders at all.[3] They used weekly docetaxel in the treatment of elderly patients with advanced breast cancer. Spazzapan et al. stressed the fact that nail toxicity is typical for the weekly schedule of taxanes.
By reporting this first case of nail toxicity on an every 3-week regimen of docetaxel, we want to point out that the nail toxicity occurring with taxanes is not limited to patients receiving the weekly regimen but can occur in patients on the tri-weekly schedule of treatment. Clinicians should be aware of the side effects of taxanes on nails that may be severe, leading to impaired quality of life or to discontinuation of chemotherapy.
References
1. Simon Spazzapon, Diana crivellari, Davide Lombardi, et al.: Nail Toxicity related to weekly Taxanes: An important issue requiring a change in Common Toxicity Criteria Grading? J Clin Oncol 2002; 21: 4404-4405.2. Perez EA, Vogel CL, Irwin DH, et al.: Multicenter phase II trial of weekly paclitaxel in women with metastatic breast cancer. J Clin Oncol 2001; 19: 4216-4223.
3. Hainsworth JD, Buris HA III, Yardley DA, et al.: Weekly docetaxel in the treatment of elderly patients with advanced breast cancer: A Minnie Pearl Cancer Research Network phase II trial. J Clin Oncol 2001; 19: 3500-3505.
© 2003 Dermatology Online Journal

