Generalized pruritic eruption in a patient with chronic kidney disease
Published Web Location
https://doi.org/10.5070/D37s48c7kfMain Content
Generalized pruritic eruption in a patient with chronic kidney disease
Collette McCourt MB BCh1, Andrea Corry MB BCh1, Maureen Walsh MB BCh2, Clifford McMillan MB BCh3
Dermatology Online Journal 16 (4): 10
1. Department of Dermatology, Belfast City Hospital, Belfast, County Antrim, Northern Ireland. collmccourt@hotmail.com2. Department of Pathology, Royal Victoria Hospital, Belfast, County Antrim, Northern Ireland
3. Department of Dermatology, Belfast City Hospital, Belfast, County Antrim, Northern Ireland
Case report
A 67-year-old gentleman presented with a 7-month history of a widespread pruritic eruption, clinically consistent with prurigo nodularis. His past history included ischaemic heart disease, hypertension, hypercholesterolemia, peripheral vascular disease, and chronic kidney disease stage 5 (CKD).
Laboratory investigations revealed a macrocytic anemia with normal thyroid function tests, B12, and folate. Autoimmune screen, liver function tests, plasma protein electrophoresis, and immunoglobulins were normal. Renal profile revealed CKD stage 5. Response to conventional treatment (potent topical steroids and anti-histamines) was poor, thus a skin biopsy was performed and sent for histopathology, confirming prurigo nodularis.
 |
| Figure 1 |
|---|
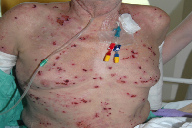
Shortly after presentation he commenced on hemodialysis 3 times per week, which improved his skin for several months before he relapsed. Cyclosporine was then commenced (2 mg/kg) and produced a short-lived response; he was subsequently admitted for in-patient treatment. At this time he was noted to have superficial erosions interspersed with crusted nodules. No mucosal involvement or intact blisters were noted. (Figure 1)
Skin biopsies were sent, this time for both histopathology and immunofluorescence. Histopathology from a nodule showed hyperkeratosis overlying a thickened epidermis with a prominent granular cell layer. The basal layer was intact. The underlying dermis was vascularised with moderate chronic inflammation composed mainly of lymphocytes and plasma cells. Occasional eosinophils were seen but no blister was identified (similar to previous biopsy). (Figures 2 and 3) Direct immunofluorescence showed linear deposits of C3 complement, fibrin, and IgG along the basement membrane. Indirect immunofluorescence revealed anti-basement membrane antibody at a titre of greater than 1 in 80.
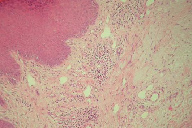 | 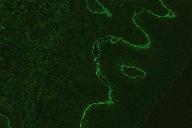 |
| Figure 2 | Figure 3 |
|---|---|
A diagnosis of pemphigoid nodularis was made and his skin improved dramatically within a week after starting oral prednisolone (40 mg once daily).
Discussion
Pemphigoid nodularis is a condition now recognized in the literature as a variant of bullous pemphigoid [1, 2, 3]. This disease entity consists of the classical excoriated papules and nodules of prurigo nodularis with overlapping features of bullous pemphigoid (BP) [1].
The time interval from the appearance of nodules to blister formation varies between reports and in some cases blisters never occur, despite evidence of circulating and tissue bound anti-basement membrane antibodies [1].
Biopsy taken from a nodular lesion demonstrates classical changes associated with prurigo nodularis [2]. Immunofluoresence, however, usually demonstrates linear basement membrane zone deposition of IgG +/- C3 and in most cases anti-basement membrane antibodies [2].
The pathogenesis of this condition is still uncertain, however various theories are postulated. One theory is that the two conditions are co-incidental, occurring simultaneously in the same patient [1].
Another is that trauma from repeated scratching may result in the expression of critical antigens usually “hidden” from the immune system [4]. In a susceptible patient, after exposing these antigens, an immune response may then be triggered, resulting in BP antibodies [1, 4, 5]. This phenomenon may be related to “epitope spreading” [4] which has also been implicated in the development of other autoimmune conditions such as pemphigus vulgaris [4].
To our knowledge there are no reported cases of pemphigoid nodularis occurring in patients on hemodiaylsis, however, there have been cases of BP in patients on hemodialysis localized to their venous access sites [5] and patients with CKD.
In the CKD and hemodialysis population, a possible trigger for blister formation may be elevated levels of CD23 caused by end stage renal disease +/- hemodialysis [6]. CD23 is a low affinity IgE receptor expressed on mature B lymphocytes [7] and elevated levels have been noted in patients with CKD and patients on hemodialysis, relative to controls [6].
Moreover, in a study of patients with bullous pemphigoid that looked at CD23 expression and serum IgE, increased CD23 expression on B cells in individuals with severe bullous pemphigoid correlated with elevated IgE levels when compared with moderate BP and control individuals [7]. The authors suggest that up-regulation of surface CD23 may be involved in IgE synthesis and thus the pathogenesis of BP [7]. Unfortunately serum IgE was not sent at diagnosis in this case.
This case highlights the importance of considering skin biopsy for both histopathology and immunofluorescence in patients with prurigo nodularis resistant to conventional treatment, especially if erosions and crusts are noted. More specifically, it should be considered in treatment resistant patients with CKD; our case highlights the fact that the diagnosis may be missed if skin biopsies are sent for histopathological analysis only.
References
1. Powell AM, Albert S, Gratian MJ and Bittencourt R et al. Pemphigoid Nodularis (non-bullous): a clinicopathological study of five cases. Br Journal Dermatol. 2002; 147(2): 343-349. [PubMed]2. Bourke JF, Berth-Jones J, Gawkrodger DJ and Burns DA. Pemphigoid nodularis: a report of two cases. Clin Exp Dermatol. 1994; 19(6): 496-499. [PubMed]
3. Cliff S and Holden CA. Pemphigoid nodularis: a report of three cases and review of the literature. Br Journal Dermatol. 1997; 136(3): 398-401. [PubMed]
4. Chan LS, Vanderlugt CJ, Hashimoto T, NishikawaT et al. Epitope Spreading: Lessons from autoimmune skin diseases. J Invest Dermatol. 1998; 110(2): 103-109. [PubMed]
5. Pardo J, Rodriguez-Serna M, Mercader P and Fortea JM. Localized bullous pemhigoid overlying a fistula for hemodialysis. J Am Acad Dermatol. 2004; 51(2 Suppl): S131-2. [PubMed]
6. Descamps-Latscha B, Herbelin A, Nguyen AT, De Groote D et al. Soluble CD23 as an effector of immune dysregulation in chronic uremia and dialysis. Kidney Int. 1993; 43(4): 878-884. [PubMed]
7. Inaoki M, Sato S and Takehara K. Elevated expression of CD23 on peripheral blood B lymphocytes from patients with bullous pemphigoid: correlation with increased serum IgE. J Dermatol Sci. 2004; 35(1): 53-9. [PubMed
© 2010 Dermatology Online Journal

