Facial angiofibromas treated with topical rapamycin: An excellent choice with fast response
Published Web Location
https://doi.org/10.5070/D36tb7n61mMain Content
Facial angiofibromas treated with topical rapamycin: An excellent choice with fast response
Teresa Truchuelo MD, Blanca Díaz-Ley MD, Luis Ríos MD, Javier Alcántara MD, Pedro Jaén MD
Dermatology Online Journal 18 (1): 15
Ramón y Cajal Hospital Madrid, Madrid, SpainAbstract
Tuberous sclerosis (TS) is the second most common genodermatosis in our country and one of its main characteristics is the presence of facial angiofibromas. These benign tumors can be really bothersome for some patients and there is not a gold-standard treatment. Laser therapy has been used with good responses but it is a painful option and recurrence is guaranteed. TS develops as a result of a mutation of one of two genes, TSC1 or TSC2, which encode for hamartin and tuberin, respectively. TSC1 and TSC2 are tumor suppressors that inhibit mTOR, which if mutated results in mTOR activation, leading to an increase in protein translation. This eventually induces formation of hamartomatous tumors in patients with TSC. Oral rapamycin had been reported to be effective for the treatment of various tumors, apparently because of its action of inhibiting the m-TOR complex. Recently it has been suggested that the drug may be effective when applied topically. We report the 6th case of facial AF treated with topical rapamycin, 1 percent, once per day. An excellent response was achieved surprisingly rapidly. We propose this option as a safe and effective therapy.
Introduction
An 11-year-old girl with a personal history of tuberous sclerosis (ST) came to our hospital. She had numerous signs of the disease: multiple angiomyolipomas in the kidney without functional impairment, cortical tubers, and epilepsy, which had resolved. Skin changes included ash leaf-like hypopigmented macules, Koenen tumors, and angiofibromas. The patient wanted to treat the numerous, disfiguring papules on her cheeks. She had previously received treatment for this reason with CO2 laser, which produced a good response, but was poorly tolerated. In addition, the lesions had recurred rapidly.
Case report
On physical examination, numerous firm erythematous papules with a telangiectatic surface were located on the cheeks (Figure 1). The appearance of the facial papules was consistent with facial angiofibromas.
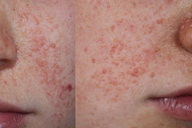 | 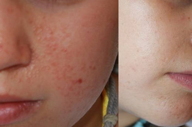 |
| Figure 1 | Figure 2 |
|---|
After informed consent, we decided to treat the lesions with a novel and promising therapy: topical rapamycin. For this purpose the following formulation was prescribed: rapamycin, 1 percent, in Dexeryl® cream, once a day. Laboratory findings (blood count and chemistry with liver enzymes) were normal at the beginning and also after one and 4 weeks of treatment. Blood levels of rapamycin were undetectable. The treatment was well tolerated and improvement was already observed 2 days after beginning treatment (Figure 2a). One month after starting treatment the lesions had improved in size and color, and 2 weeks later they had almost disappeared, at which point the treatment was stopped (Figure 2b).
Discussion
Tuberous sclerosis is an autosomal dominant genetic disease (the second most common in our country). It is a neurocutaneous syndrome characterized by the formation of hamartomas in the brain, skin, kidneys, heart, and other organs [1, 2, 3]. Up to 90 percent of patients with ST have cutaneous lesions. Among them, hypopigmented patches are the first ones to develop, followed by the shagreen plaque. During puberty periungueal fibromas usually appear. Facial angiofibromas may be present at any age but they usually develop in late infancy and progressively increase in number and size afterwards. An angiofibroma consists of vascular and interstitial cells that express angiogenic factors such as vascular endothelial growth factor (VEGF) [4]. They represent the most frequent lesion in ST (74.5% to 89.6%) [1]. These facial papules are generally asymptomatic, but are cosmetically bothersome because they are usually located on visible facial areas and can have a significant impact on a patient’s quality of life. An effective treatment with limited toxicity is needed [3].
Tuberous sclerosis develops as a result of a mutation of one of two genes, TSC1 or TSC2, which encode for hamartin and tuberin, respectively. TSC1 and TSC2 are tumor suppressors that inhibit mTOR, which if mutated results in mTOR activation. This leads to an increase in protein synthesis that eventually induces the formation of hamartomatous tumors in patients with TSC [1, 3, 4].
Rapamycin is a drug, which belongs to the group of molecules known as the m-TOR inhibitors; it has been traditionally used in transplant recipients [5, 6]. It has powerful antiproliferative and immunosuppressant activity. Although similar in structure to cyclosporine and tacrolimus, it is not a calcineurin inhibitor and its safety profile is much higher [4, 5]. It modulates different signaling pathways, which lead to an immunosuppressive state, but it also promotes an antiangiogenic effect. The drug has an influence in pathways important in regulation of the cell growth and apoptosis. This could explain its role in ST hamartomas [5]. Rapamycin inhibits the progression from G1 phase to S phase, suppresses T-lymphocyte and antibody production, and inhibits keratinocytic proliferation and neutrophilic inflammatory activity. It also decreases the levels of the vascular endothelial growing factor (VEGF) [4, 5].
The reduction of various tumors, such as AF in ST patients, has been described after the oral administration of rapamycin [4, 7-11]. Topical administration of rapamycin was shown to be effective in mice with ST in 2008 [12] and it has recently been published as being useful in humans for the skin lesions of ST. Our case is the 6th one in the literature that reports the healing of AF after topical rapamycin treatment [3, 5, 13].
In addition, topical rapamycin has been used for lesions of Kaposi sarcoma, psoriasis, and lichen planus [14, 15]. It has also been studied for its utility in mice with various lesions related to Birt-Hogg-Dubé syndrome with promising results [16].
The rapid resolution of the angiofibromas in our case is remarkable and this has not been reported before. We suggest that younger patients with lesions of shorter duration could be easier to treat.
Side effects reported in the literature after topical rapamycin administration are mild and local (irritation, acneiform eruption, and oral aphthous ulcers) [5]. No systemic effects have been reported in contrast with the oral administration of rapamycin. Rapamycin in a concentration of 1 percent in cream base appears to be effective and safe when applied topically and could be an excellent choice.
In conclusion, topical rapamycin seems to be an excellent therapeutic option for facial AF treatment in patients with SR. These tumors, despite being non-malignant lesions, can represent an important problem for patients. Further studies are needed to determine the proper concentration and frequency of use. In addition, new studies will be able to document the usual time to response and, it is hoped, to confirm the apparently excellent safety profile of topical rapamycin administration.
References
1. Schwartz, R.A., Fernández G, Kotulska K, Jóźwiak S. Tuberous sclerosis complex: advances in diagnosis, genetics, and management. J Am Acad Dermatol 2007;57(2):189-202. [PubMed]2. Fernández Guarino M., Boixeda P, Anaya MJ, Beldar P, Jaén P. Esclerosis tuberosa. Hallazgos clínicos en 67 pacientes. Actas Dermosifiliogr 2009;100:597-602. [PubMed]
3. Kaufman McNamara E, Curtis AR, Fleischer Jr AB. Successful treatment of angiofibromata of tuberous sclerosis complex with rapamycin. J Dermatolog Treat 2010: 1-3. [PubMed]
4. Hofbauer, GF., Marcollo-Pini A, Corsenca A, Kistler AD, French LE, Wüthrich RP, Serra AL. The mTOR inhibitor rapamycin significantly improves facial angiofibroma lesions in a patient with tuberous sclerosis. Br J Dermatol 2008;159(2):473-5. [PubMed]
5. Haemel, AK., O'Brian AL, Teng JM. Topical rapamycin: a novel approach to facial angiofibromas in tuberous sclerosis. Arch Dermatol 2010;146(7):715-8. [PubMed]
6. DeKlotz C., Ogram A, Singh S, Dronavalli S, MacGregor J. Dramatic Improvement of Facial Angiofibromas in Tuberous Sclerosis With Topical Rapamycin: Optimizing a Treatment Protocol. Arch Dermatol. 2011;147:1116-1117. [PubMed]
7. Bissler JJ, McCormack FX, Young LR, Elwing JM, Chuck G, Leonard JM, Schmithorst VJ, Laor T, Brody AS, Bean J, Salisbury S, Franz DN. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med 2008;358(2):140-51. [PubMed]
8. Franz DN, Leonard J, Tudor C, Chuck G, Care M, Sethuraman G, Dinopoulos A, Thomas G, Crone KR et al. Rapamycin causes regression of astrocytomas in tuberous sclerosis complex. Ann Neurol 2006;59(3):490-8. [PubMed]
9. Peces R, Peces C, Cuesta-López E, Pérez-Dueñas V, Vega-Cabrera C, Azorín S, Selgas R. Low-dose rapamycin reduces kidney volume angiomyolipomas and prevents the loss of renal function in a patient with tuberous sclerosis complex. Nephrol Dial Transplant 2010;25(11):3787-91. [PubMed]
10. Tarasewicz, A, Debska-Slizień A, Konopa J, Zdrojewski Z, Rutkowski B. Rapamycin as a therapy of choice after renal transplantation in a patient with tuberous sclerosis complex. Transplant Proc 2009;41(9): 3677-82. [PubMed]
11. Micozkadioglu H, Koc Z, Ozelsancak R, Yildiz I. Rapamycin therapy for renal, brain, and skin lesions in a tuberous sclerosis patient. Ren Fail 2010;32(10): 1233-6. [PubMed]
12. Rauktys R, Koc Z, Ozelsancak R, Yildiz I. Topical rapamycin inhibits tuberous sclerosis tumor growth in a nude mouse model. BMC Dermatol 2008; 8:1. [PubMed]
13. Valerón-Almazán P, Vitiello M, Abuchar A, Kerdel FA. Topical Rapamycin Solution to Treat Multiple Facial Angiofibromas in a Patient With Tuberous Sclerosis. Actas Dermosifiliogr. 2011 Aug 20. [PubMed]
14. Ormerod AD, Shah SA, Copeland P, Omar G, Winfield A. Treatment of psoriasis with topical sirolimus: preclinical development and a randomized, double-blind trial. Br J Dermatol 2005;152(4): 758-64. [PubMed]
15. Soria A, Agbo-Godeau S, Taïeb A, Francès C. Treatment of refractory oral erosive lichen planus with topical rapamycin: 7 cases. Dermatology 2009;218(1): 22-5. [PubMed]
16. Baba M, Furihata M, Hong SB, Tessarollo L, Haines D, Southon E.Kidney-targeted Birt-Hogg-Dubé gene inactivation in a mouse model: Erk1/2 and Akt-mTOR activation, cell hyperproliferation, and polycystic kidneys. J Natl Cancer Inst 2008:16;100(2): 140-154. [PubMed]
© 2012 Dermatology Online Journal

