Actinomycetoma of the chest wall attributed to after reconstructive surgery
Published Web Location
https://doi.org/10.5070/D36g2866ffMain Content
Actinomycetoma of the chest wall attributed to Nocardia nova after reconstructive surgery
Joana Antunes1, David Pacheco1, Rita Travassos1, Hortênsia Sequeira2, Paulo Filipe1,2, Manuel Sacramento Marques1
Dermatology Online Journal 18 (1): 4
1. Serviço de Dermatologia, Hospital de Santa Maria, Lisboa, Portugal2. Laboratório de Micologia, Faculdade de Medicina de Lisboa, Lisboa, Portugal
Abstract
A 29-year-old man, presented with multiple ulcers, nodules, abscesses, fistulae, and atrophic scars, over the right chest wall. Six years prior, the patient had a car accident, which resulted in skin loss of the right arm, shoulder, thoracic wall. In addition, he suffered a supracondylar fracture; orthopedic surgery and skin grafts were required. Material discharging from sinus tracts was obtained for mycological and bacteriological studies. Direct microscopic examination revealed small white grains. Cultures on Sabouraud and Lowenstein-Jensen media isolated orange-white colonies suggestive of Nocardia. PCR assay identified Nocardia nova. Thoracic and right upper limb CT showed signs of chronic osteomyelitis. Treatment with imipenem/cilastatin for 8 weeks, followed by amoxicillin clavulanate for 6 months, resulted in healing of lesions and improvement in the patient’s general health. Primary cutaneous nocardiosis remains a diagnostic challenge. Nocardia are soil-borne filamentous gram-positive bacteria. Identification of characteristic granules on examination of discharge smears from discharge or tissue biopsy is essential for diagnosing mycetoma. Because grain discharge is discontinuous, multiple clinical specimens should be submitted for microscopic examination and culture. Sulfonamides have been the mainstay of Nocardia actinomycetoma treatment. However, our patient’s strain was resistant to Co-trimoxazole. Therefore, treatment with imipenem followed by amoxicillin clavulanate was favored, with good clinical and analytical response.
Nocardia species are ubiquitous, soil-borne, filamentous, aerobic, gram-positive, weakly acid-fast bacteria, members of the Actinomycetales order [1, 2]. Nocardiosis can manifest both as localized or disseminated disease. Most cases of nocardiosis take the form of disseminated disease and typically affect immunocompromised individuals. The most common pathogen is Nocardia asteroides, which is acquired through the respiratory tract as airborne spores or mycelial fragments [3].
Cutaneous nocardiosis can result from primary cutaneous infection or be secondary to hematogenous dissemination, usually from an initial pulmonary focus of disease. Primary cutaneous nocardiosis follows cutaneous inoculation by any of the pathogenic Nocardia, with N. brasiliensis being responsible for 80 percent of cases [4]. It can present as 3 distinct clinical variants: 1) superficial skin and soft tissue infection (pustules, cellulitis, abscesses or paronychia); 2) lymphocutaneous or lymphonodular infection that simulates sporotrichosis; and 3) a deeper and more common type of infection, mycetoma [2, 5]. Mycetoma caused by bacteria (aerobic actinomycetes) is classified as Actinomycetoma [6].
Mycetoma is a chronic, granulomatous infection involving the skin, subcutaneous tissue, and underlying structures. Clinically, it produces an area of localized swelling with nodules that develop multiple sinus tracts; elimination of macroscopic colored grains is a common feature. However, even though the clinical picture may be suggestive of mycetoma, definite diagnosis requires visualization and positive culture of the grains, which is often the limiting process [7]. Identification of the etiological agent is also essential for institution of adequate treatment and a favorable outcome.
We report an interesting case of extensive primary cutaneous nocardiosis of the chest wall following major trauma.
A 29-year-old man from Guinea-Bissau presented with a 4-year history of multiple inflammatory nodules, abscesses, and intermittently discharging sinuses, over the right anterior and posterior thoracic wall and shoulder. The patient also reported malaise, anorexia, and unintended weight loss.
Two years before the onset of the skin lesions, the patient had been involved in a car accident in Guinea-Bissau, in which he was thrown to the street. The accident resulted in skin loss of the right arm, shoulder, and chest wall along with a supracondylar fracture. He subsequently had orthopedic surgery and skin grafts in Senegal. His previous medical history was otherwise unremarkable.
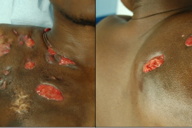
On physical examination, the patient was emaciated and sub-febrile (tympanic temperature 37.6°C). Over the anterior and posterior right chest wall and right shoulder there were multiple, well delineated, round and oval ulcers, covered in granulation tissue. In some cases, large inflammatory masses resulted. When palpated, foul-smelling, purulent and serosanguineous material would drain from different orifices, showing the presence of sinuses and fistulae (Figure 1). Some lesions were superimposed on areas of keloid scarring. Keloids and contractions impaired his movement. Enlarged, tender lymphadenopathy was palpable in the right axilla.
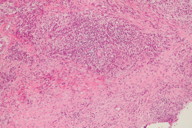
Routine laboratory tests showed chronic disease anemia (Hb 10.9 g/dL), elevated C-reactive protein (7.9 mg/dL), and chronic Hepatitis B. Serologies for HIV 1 and 2 were negative. Chest X-ray was normal and Quantiferon-gold was negative. Histological examination of an incisional skin biopsy collected from the back revealed an ulceration with granulation tissue above abundant fibrosis and mixed inflammatory infiltrate. Prominent abscess formation and a few multinuclear giant cells, suggestive of infectious granulomatous disease were demonstrated (Figure 2). Periodic acid-Schiff and Ziehl-Neelsen staining of skin biopsy were both negative. Thoracic and upper-limb computed tomography showed densification of soft tissue and signs of chronic osteomyelitis of the humerus and ulna.
The discharge from the sinus was collected daily for bacteriological and mycological examination. The first bacteriological cultures isolated methicillin-sensitive Staphylococcus aureus.
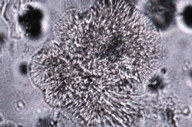
Direct microscopic examination of exudate, stained with lactophenol blue, revealed small, soft, white grains (~0.5 mm) (Figure 3). Multiple cultures on Sabouraud dextrose agar and Lowenstein-Jensen medium, incubated at 24°C and 37°C, developed powdery, orange-white colonies, compatible with Nocardia spp. (Figure 4). PCR assay of those isolates, performed at the Observatoire Français des Nocardioses (Lyon, France), identified N. nova. In vitro antibiotic susceptibility testing rendered it resistant to ampicillin, trimethoprim/sulfamethoxazole and ciprofloxacin, but sensitive to amoxicillin clavulanate, imipenem/cilastatin and erythromycin.
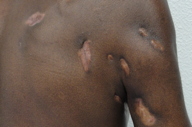
The patient was started on imipenem/cilastatin 500 mg IV q6h, for 8 weeks, with complete healing of skin lesions and improvement of general status (Figure 5). After hospital discharge, he was medicated with amoxicillin clavulanate 875+125mg and referred for an orthopedic and a physical therapy evaluation until the osteomyelitis resolved and he recovered upper limb mobility.
Nocardia nova was first described by Michio Tsukamura in 1982, who separated N. asteroides sensu strictu into N. asteroides and N. nova [8]. Nevertheless, it was only established as a distinct species when Yano et al. clearly differentiated it from N. asteroides and N. farcinica through DNA hybridization analysis [9]. It was further characterized by Wallace et al. who described white colonies, which developed an orange pigmentation after 7 to 10 days. The organism demonstrated arylsulfatase activity, susceptibility to erythromycin, moderate susceptibility for ampicillin, and resistance to amoxicillin clavulanate and carbenicillin [10]. However, antibiotic susceptibility testing performed for our isolate demonstrated that it was resistant to ampicillin and sensitive to amoxicillin clavulanate, which may mean an incomplete characterization of the species by Wallace et al, or already an adaptive evolution of N. nova.
In a review of indexed literature, we found 5 other reports of cutaneous nocardiosis caused by N. nova (Table 1). The reports span all clinical patterns of cutaneous nocardiosis and the majority were patients with some type of immunosuppression. Our patient had no comorbidities, but had suffered major trauma and undergone reconstructive surgery. Whether infection was acquired as a result of the accident or was iatrogenically caused remains unclear.
All the reviewed cases had successful clinical outcomes, but only after adjusting treatment to the culture and antibiotic sensitivity testing results, which appears to be the critical step in effectively managing nocardiosis.
Demonstration of the organism from clinical specimens, like granules, pus, or aspirate fluid, is the mainstay of diagnosis for primary cutaneous nocardiosis [5]. However, because the discharge of mycetoma grains is only sporadic, repeated sample collection is warranted whenever this diagnosis is suspected. This concept should be particularly enforced in non-endemic areas, such as Europe and the US, where this disease is very uncommon, but may become more prevalent with the continuous flow of travelers or immigrants from endemic regions.
ACKNOWLEDGEMENTS: The authors would like to thank Professor Patrick Boiron and Drs. Verónica Rodríguez-Nava and Emmanuelle Bergeron of Observatoire Français des Nocardioses, Laboratoire de Mycologie – Faculté de Pharmacie, Université de Lyon, Lyon, France, for performing PCR assay and antibiotic susceptibility testing on our samples.
References
1. Tan CK, Lai CC, Lin SH, et al. Clinical and microbial characteristics of nocardiosis including those caused by emerging Nocardia species in Taiwan, 1998-2008; Clin Microbiol Infect 2010;16(7):966-72 [PubMed]2. Lerner PI. Nocardiosis. Clin Infect Dis. 1996;22(6):891-903 [PubMed]
3. Baraboutis IG, Argyropoulou A, Papastamopoulos V, et al. Primary sternal osteomyelitis caused by Nocardia nova: Case report and literature review. Braz J Infect Dis. 2008;12(3):257-9 [PubMed]
4. Dodiuk-Gad R, Cohen E, Ziv M, et al. Cutaneous nocardiosis: report of two cases and review of the literature. Int J Dermatol. 2010;49(12):1380-5 [PubMed]
5. Inamadar AC, Palit A. Primary cutaneous nocardiosis: A case study and review. Indian J Dermatol Venereol Leprol 2003;69(6):386-91 [PubMed]
6. Lichon V, Khachemoune A. Mycetoma: A review. Am J Clin Dermatol. 2006;7(5)315-21 [PubMed]
7. Fahal AH. Mycetoma: a thorn in the flesh. Trans R Soc Trop Med Hyg. 2004;98(1):3-11 [PubMed]
8. Tsukamura M. Numerical analysis of the taxonomy of Nocardiae and Rhodococci. Microbiol Immunol. 1982;26(12):1101-19 [PubMed]
9. Yano I, Imaeda T, Tsukamura M. Characterization of Nocardia nova. Int J Syst Bacteriol. 1990;40(2):170-4
10. Wallace RJ, Brown BA, Tsukamura M, et al. Clinical and laboratory features of Nocardia nova. J Clin Microbiol. 1991;29(11):2407-11 [PubMed]
11. Schiff TA, Sanchez M, Moy J, et al. Cutaneous nocardiosis caused by Nocardia nova occurring in an HIV-infected individual: A case report and review of the literature. J Acquir Immune Defic Syndr. 1993;6(7):849-51 [PubMed]
12. Burucoa C, Breton I, Ramassamy A, et al. Western blot monitoring of disseminated Nocardia nova infection treated with clarithromycin, imipenem, and surgical drainage. Eur J Clin Microbiol Infec Dis 1996;15(12):943-7 [PubMed]
13. Shimizu A, Ishikawa O, Negai Y, et al. Primary cutaneous nocardiosis due to Nocardia nova in a healthy woman. Br J Dermatol. 2001;145(1):154-6 [PubMed]
14. Inamadar AC, Palit A, Peerapur BV, et al. Sporotrichoid nocardiosis caused by Nocardia nova in a patient infected with human immunodeficiency virus. Int J Dermatol. 2004;43(11):824-6 [PubMed]
15. Arora G, Friedman M, MacDermott RP. Disseminated Nocardia nova infection. South Med J. 2010;103(12):1269-71 [PubMed]
© 2012 Dermatology Online Journal