Collagen XVIII and fibronectin involvement in bullous scleroderma
Published Web Location
https://doi.org/10.5070/D367t4c63mMain Content
Collagen XVIII and fibronectin involvement in bullous scleroderma
Soraya Neves Marques Barbosa dos Santos MD PhD1, Gisele Viana de Oliveira MD 2, André Luiz Pasqua Tavares MD2, André Ricardo Massensini PhD2, Lorenza Machado de Souza Carvalhaes PhD2, Ritva Reljasvaara PhD3, Gregory Thomas Kitten PhD2
Dermatology Online Journal 11 (1): 17
Hospital Semper, Belo Horizonte, Minas Gerais, Brasil1. sorayasantos@doutoradermatologista.com.brInstituto de Ciências Biológicas, Universidade Federal de Minas Gerais, Belo Horizonte,
Minas Gerais, Brasil2 Collagen Research Unit, Biocenter Oulu and Department of Medical Biochemistry and Molecular Biology, University of Oulu,
Oulu, Finland3
Abstract
Background.—Endostatin, an anti-angiogenic C-terminal fragment of collagen XVIII, has been recently reported to play a role in scleroderma
pathogenesis, but collagen XVIII immunohistochemistry in scleroderma skin has still not been performed. Bullous scleroderma,
a rare form of scleroderma, may have altered angiogenic and lymphangiogenic characteristics.
Objective.—Our aim is to report a rare case of bullous scleroderma, studying the presence of fibronectin and collagens type I, III and
XVIII in sclerodermic skin.
Methods.—We describe the progression of bullous scleroderma in a 67-year-old patient since the first symptoms. Histological and immunohistochemical
aspects of skin biopsies are compared to normal skin from a patient without scleroderma and are correlated with the pathogenesis
of the disease. Indirect immunofluorescence measured by laser confocal microscopy allows quantitative determination of fibronectin
and collagens type I, III and XVIII.
Results and conclusions.—Dermo-epidermal cleavage, fibrosis and inflammation are the main histological findings. The dermal distribution and amounts
of collagens and in the scleroderma patient are similar to normal skin. Conversely, both fibronectin and collagen XVIII are
increased in scleroderma skin, suggesting their involvement in the pathogenesis of bullous scleroderma.
Clinical synopsis
A 67-year-old female patient presents to the Dermatology Service of Health Council of Belo Horizonte City, Brazil, complaining of dry, thick skin. Clinical examination shows one large sclerotic plaque in the chest region, with atrophic parts and wine-colored borders extending to the inframammary area, and two well-delineated plaques occupying both sides of the prescapular area. By 3 months, the lesions extend over the dorsum, chest, abdomen, and legs. A few days later, vesicles and blisters begin to arise over the extensor region of the limbs. A viscous and clear secretion is associated with the lesions, some of which become infected or formed crusts, mainly on the legs (Fig. 1). At the end of the fourth month, the overall skin presents with atrophy or sclerosis, with few islands of normal skin.
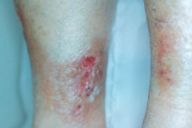
|
| Figure 1 |
|---|
| Blisters and sclero-atrophic plaque on the leg, after 4 months of disease. |
The hemogram, thyroid, and kidney-function exams do not reveal abnormalities. Lyme disease antibodies (IgM and IgG) were negative and chest x-ray and upper endoscopy do not show abnormalities. Serology for Borrelia burgdorferi display negative results and serum eosinophils show normal values in multiple blood evaluations. An ANA test was positive (1:160, specked pattern).
Methods
During the 4-month observation, skin biopsies are obtained from the borders of abdominal plaques and from the apparently healthy skin. One control skin sample is obtained from discarded material of a patient submitted to elective surgery.
The study has been approved by the City Health Department of Belo Horizonte and by the University Ethics Committee, and the patient has given informed consent for histological and immunohistochemical studies.
Histology.—Samples fixed in paraformaldehyde are processed for eosin/hematoxylin, Gomori trichrome, and colloidal iron histochemistry. Hyperkeratosis, epidermal atrophy, vacuolization of basal cells, dermal edema and sclerosis, perivascular lymphohistiocytic infiltrate, multifocal vesiculation and dermo-epidermal fends are seen in the scleroderma skin. Thick and hyaline collagen fibers are found, as well as lymphatic ectasia and pilosebaceous atrophy that extends to the hypodermic region (Fig. 2). Mucin investigation displays negative results (using PAS, Gomori and colloidal iron). The apparently normal skin from the patient with scleroderma shows histological signs of the disease, with thick collagen fibers and lymphatic ectasia.
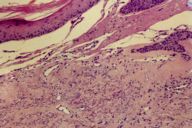
|
| Figure 2 |
|---|
| Thick and hyaline collagen fibers, lymphatic ectasia and pilosebaceous atrophy extending into the hypodermic region on the scleroderma skin. |
Immunohistochemical studies.—Part of the samples were fixed in a -20° C solution of 80 percent methanol and 20 percent DMSO and processed for immunohistochemistry. They were embedded in paraplast, and 3-µm tissue sections were mounted on slides. The distribution and levels of collagens type I and III (Research Diagnostics Inc.®, Flanders, NJ) and XVIII (Antibody against the N-terminal noncollagenous region of human collagen XVIII anti-all huXVIII, polyclonal, rabbit anti-human), and fibronectin (Research Diagnostics Inc.®, Flanders, NJ) were investigated in both scleroderma and control patients using specific primary antibodies and fluorescent-labeled secondary antibody (Cappel Laboratory®, W. Chester, PA).
Laser confocal microscopy and image analysis.—Images were captured using the confocal laser scanning microscope (Bio-Rad MRC 1024 / Zeiss Axiovert 100), 40X objective, magnification 400, and analyzed with Confocal Assistant® and Image Tool® softwares. The region corresponding to the connective tissue was divided in two distinct regions: region 1 (superficial connective tissue, corresponding to papillary dermis, including epidermis), and region 2 (deep connective tissue, corresponding to the reticular layer of the dermis approximately 50 µm below the basement membrane, under the superficial vascular plexus). In order to analyze the amount of collagens quantitatively, the intensity of fluorescence was measured in nine randomly selected areas in each region (three areas per section, in a total of three sections). Confocal-microscope parameters were adjusted for background staining level observed in the epithelial tissue on each section in order to allow for comparisons between different samples.
Results

|
|
| Figure 3a | Figure 3b |
|---|---|
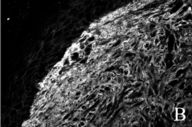
| Fibronectin deposition in normal (fig. 3a) and scleroderma skin (3b), showing increased deposition in the scleroderma. | |
|
|
| Figure 3c | Figure 3d |
|---|---|
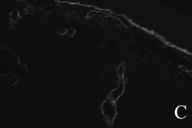
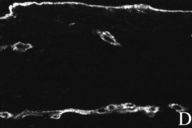
| Collagen XVIII in normal (3c) and scleroderma patient (3d). | |
|
| Figure 4 |
|---|
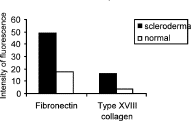
| Fluorescent intensity of collagen type XVIII and fibronectin in normal and scleroderma skin. |
Types I and III collagens were not significantly different for either normal or scleroderma patient skin. Diffuse deposition of both of these collagens was seen in the dermal compartments (data not shown). Fibronectin staining revealed a threefold increase in the scleroderma patient when compared to normal skin (Figs. 3a and 3b). Collagen-XVIII labeling was positive in the basement membrane of the skin and around blood vessels in both scleroderma and normal skin. Fluorescence intensity was increased fourfold for the scleroderma skin (Figs. 3c, 3d, and 4).
Discussion
We report a rare case of a generalized bullous scleroderma. The lesions progressed until covering almost 100 percent of the total body surface area, with an increased number of blisters and vesicles in the limbs. Histological findings showed lymphangiectasia, different degrees of edema in the dermal region, and a thick dermis with fibrosis and sclerosis, which is compatible with late-phase scleroderma [1]. Increased fluorescence intensity for both fibronectin and type-XVIII collagen was seen in the scleroderma skin, although this report is only a case report, and those findings should be confirmed in a larger series of patients.
The bullous form of scleroderma is rare and often misdiagnosed, in part because of clinical and histological similarities to lichen sclerosus et atrophicus, but also because of confusing classification [1, 2]. Morphea profunda, bullous morphea profunda, bullous morphea, bullous scleroderma, and generalized scleroderma have similar clinical descriptions and might well be considered as one type [1, 2].
The pathogenic events that lead to blister formation are still unknown. Templeton (1941) suggested that bullous lesions were formed after lymphatic obstruction, caused by the extensive fibro-edematous process [2]. Tuffanelli (1975) correlated the blisters with lymphangiectasia in the superficial dermis. He proposed that dermal fibrosis would progressively cause dilation of lymph vessels, culminating with the formation of the blisters [3]. Pautrier (1929) attributed the bullous lesions to trophic disturbances after vascular abnormalities [4]. Local trauma has also been implicated in the pathogenesis of the disease [5], and lymphatic dilation is sometimes absent [6, 7]. Daoud described thirteen scleroderma patients with subepidermal blisters associated with perivascular inflammation [8].
In agreement with other authors, we found evidence that the lymphatic dilation might be involved in the pathogenesis of the disease [3, 8]. Increased concentrations of fibronectin and collagen XVIII were also seen in this patient. Although types I and III collagens can vary greatly in the different phases of scleroderma, fibronectin has consistently been reported as increased [9], confirming our results. Collagen XVIII, which is normally localized in the basement membranes of the skin and blood vessels [10], has never been investigated in tissues of patients with scleroderma. However, endostatin!, the C-terminal fragment derived from collagen XVIII, is a potent anti-angiogenic factor. High endostatin serum levels have been recently reported in scleroderma patients [11].
A role for endostatin in scleroderma has not yet been established, but it may contribute to the ischemic symptoms of the disease. One hypothesis that deserves further investigation is that, similar to VEGF-C [12], endostatin might play a role in both angiogenesis and lymphangiogenesis in scleroderma [11].
ACKNOWLEDGMENTS: Antibody against N-terminal noncollagenous region of human collagen XVIII (anti-all huXVIII, polyclonal, rabbit anti-human) was a generous gift from Dr. Taina Pihlajaniemi, University of Oulu, Finland, and it has been described previously by Saarela et al [10].
References
1. Fleischmajer R, Gay S, Meigel WN, Perlish JS. Collagen in the cellular and fibrotic stages of scleroderma. Arthritis Rheum. 1978 May;21(4):418-28. PubMed2. Templeton HJ et al. Localized scleroderma with bullae. Arch Dermatol. 1941; 111: 361-4.
3. Tuffanelli DL. Letter: Lymphangiectasis due to scleroderma. Arch Dermatol. 1975 Sep;111(9):1216. PubMed
4. Pautrier LM. Sclerodermie à évolution rapide em plaques multiples: Importance dês lesions vasculaires initiales et tardives dans l`étude de la sclérodermie. Bull. Soc. Fr. Dermatol. 36: 928-38, 1929.
5. O’Leary PA. In discussion on morphea. Arch. Dermatol. Syphiliol. 70:387-8, 1954.
6. Su WP, Greene SL. Bullous morphea profunda. Am J Dermatopathol. 1986 Apr;8(2):144-7. PubMed
7. Daoud MS, Su WP, Leiferman KM, Perniciaro C. Bullous morphea: clinical, pathologic, and immunopathologic evaluation of thirteen cases. J Am Acad Dermatol. 1994 Jun;30(6):937-43. PubMed
8. Synkowski DR, Lobitz WC Jr, Provost TT. Bullous scleroderma. Arch Dermatol. 1981 Mar;117(3):135-7. PubMed
9. Chen ZY, Maricq HR, Pilia PA, Dobson RL, Silver RM, Ainsworth SK. Fibronectin distribution in nailfold biopsies of scleroderma (systemic sclerosis) patients. Acta Derm Venereol. 1985;65(3):185-9. PubMed
10. Saarela J, Ylikarppa R, Rehn M, Purmonen S, Pihlajaniemi T. Complete primary structure of two variant forms of human type XVIII collagen and tissue-specific differences in the expression of the corresponding transcripts. Matrix Biol. 1998 Jan;16(6):319-28. PubMed
11. Hebbar M, Peyrat JP, Hornez L, Hatron PY, Hachulla E, Devulder B. Increased concentrations of the circulating angiogenesis inhibitor endostatin in patients with systemic sclerosis. Arthritis Rheum. 2000 Apr;43(4):889-93. PubMed
12. Skobe M, Detmar M. Structure, function, and molecular control of the skin lymphatic system. J Investig Dermatol Symp Proc. 2000 Dec;5(1):14-9. Review. PubMed
© 2005 Dermatology Online Journal

