Necrolytic acral erythema: A case not associated with hepatitis C infection
Published Web Location
https://doi.org/10.5070/D35f42g3b3Main Content
Necrolytic acral erythema: A case not associated with hepatitis C infection
Austin Liu MD1, Christof P Erickson MD2, Clay J Cockerell MD2, Sylvia Hsu MD1
Dermatology Online Journal 14 (6): 10
1. Department of Dermatology, Baylor College of Medicine, Houston, Texas. shsu@bcm.edu2. University of Texas Southwestern Medical Center, Dallas, Texas
Introduction
Necrolytic acral erythema (NAE) is a poorly understood and infrequently described dermatologic entity. It was first described over 10 years ago in 7 Egyptian patients [1]. It is considered by many to be part of the family of necrolytic erythemas. This group of disorders also includes acrodermatitis enteropathica/acquired zinc deficiency, biotin deficiency, essential fatty acid deficiency, niacin deficiency (pellagra), and necrolytic migratory erythema. These conditions share many similarities, both histologically and clinically. The differences are primarily attributed to their diverse etiologies. The importance of distinguishing between the necrolytic erythemas stems from their distinct treatments and prognoses.
All reported cases of NAE thus far have been shown to occur exclusively in patients with hepatitis C virus (HCV) infection. For this reason, NAE has been regarded as a cutaneous sign of infection by this particular virus. The majority of reports have described Egyptian patients. This may be a result of the higher rate of HCV infection in Egypt (approximately 22%) compared to the worldwide incidence (3%). Other conditions recognized to have an association with HCV infection include: lichen planus, leukocytoclastic vasculitis, and porphyria cutanea tarda. We report a case of NAE in a patient who did not have any evidence of HCV infection.
Case report
A 56-year-old Caucasian woman was referred to our clinic with a one month history of an erythematous, pruritic, and painful papular eruption on the dorsal toes, fingers and knees. Lesions of the dorsal hands and knees healed within two weeks without treatment. However, papules on the dorsal toes progressed to develop painful blisters associated with a severe burning sensation. The patient could not identify any aggravating factors and had not received any medications for her symptoms. At the time of presentation, she noted a decrease in pain, pruritus, and level of erythema. The patient had a medical history significant for Crohn's disease, psoriasis/psoriatic arthritis, fibromyalgia, hypothyroidism, and hyperlipidemia. Her medications included: thyroid tablets (Armour thyroid) 120 mg once daily; atorvastatin, 20 mg once daily; alprazolam; estrogen HRT, 0.625 mg twice daily; rabeprazole, 20 mg twice daily; trazodone hydrochloride, 50 mg once nightly; fluconazole, 100 mg once daily; mesalamine, 800 mg three times daily; etanercept, 50 mg twice weekly; and duloxetine, 60 mg once daily.
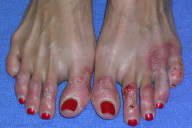
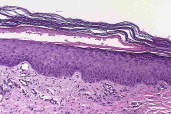
On physical examination, well-demarcated erythematous plaques with scattered bullae were present on the dorsal toes bilaterally (Fig. 1). No lesions were noted on the knees or dorsal hands. A 4-mm punch biopsy from the left dorsal foot was sent for histology and another was sent for direct immunofluorescence (DIF) examination. Histopathology showed orthokeratosis overlying an epidermis with prominent pallor in the upper third of the epidermis and extensive ballooning of superficial keratinocytes (Fig. 2). The dermis showed a sparse perivascular lymphocytic infiltrate. Direct immunofluorescence examination of the tissue did not reveal IgG, IgA, IgM or C3 deposition. The patient was seronegative for hepatitis A, B, and C viruses. Liver enzyme levels were within normal range: alkaline phosphatase, 107 U/L (reference range, 38-126 U/L); alanine aminotransferase, 17 U/L (reference range, 5-54 U/L); and aspartate aminotransferase, 28 U/L (reference range, 5-41 U/L). Plasma zinc level was 79 mcg/dL (reference range, 60-130 mcg/dL); niacin, 5.07 ug/mL (reference range, 0.50-8.45 ug/mL); free fatty acids, 0.21 mmol/L (reference range, 0.07-0.88 mmol/L); biotin, 140 pg/mL (reference range, 200-500 pg/mL); and glucagon, <50 pg/mL (reference range, <60 pg/mL).
Comment
Necrolytic acral erythema is a dermatologic condition that has been uniformly associated with HCV infection. The lesions are known to occur consistently on the dorsal feet and the most common presenting symptoms have been reported to be burning pain and/or itching [2, 3]. Clinically, the initial erythematous lesions develop into scaly, lichenified, hyperkeratotic plaques with dusky erythematous rims [2, 4, 5]. The most common histological findings include psoriasiform epidermal hyperplasia and necrosis of keratinocytes [2, 3, 4, 6].
Scaly, erythematous lesions on acral locations can be observed in both psoriasis and NAE. However, the scale of psoriasis is classically silvery white and that of NAE is darker and more verrucous [7]. Furthermore, NAE can present with flaccid blisters [4] and it typically spares the palms and soles [2]. However, there has been a case report of a patient with NAE whose palms and soles were affected with scaly, hyperkeratotic lesions with fissures and erosions [6]. If clinical presentation does not allow for accurate differentiation between the two conditions, histological examination may prove useful. Specifically, the papules and plaques of psoriasis do not possess the necrotic keratinocytes seen with NAE [4, 7]. Although our patient has a history of psoriasis, the histopathological findings of the current lesions were not suggestive of psoriasis.
As previously mentioned, many pathological entities share similarities with NAE. In view of the negative HCV test, we felt it was necessary to rule out the other disorders that make up the family of necrolytic erythemas. This group includes: acrodermatitis enteropathica/acquired zinc deficiency, biotin deficiency, essential fatty acid deficiency, niacin deficiency (pellagra), and necrolytic migratory erythema. We provide a more comprehensive overview of these conditions due to the importance of accurately differentiating among them.
Acrodermatitis enteropathica describes an inherited autosomal recessive disorder resulting in zinc deficiency in infancy. However, the term is also used by many to describe acquired deficiencies in zinc concentration. Such cases are most commonly attributed to disorders of the gastrointestinal tract including chronic inflammatory bowel disease, pancreatic insufficiency, and steatorrhea. Our patient's history of Crohn's disease therefore raised the index of suspicion for this particular disorder. The condition often presents with a triad of periorificial dermatitis, diarrhea, and alopecia. Furthermore, angular stomatitis and glossitis are often concurrently present.
Biotin deficiency is a rare condition with a clinical presentation similar to that of zinc deficiency. In adults, it can be caused by impaired absorption of biotin or excessive intake of raw egg whites because they contain avidin that binds to biotin. Laboratory results from our patient demonstrated the serum biotin level to be below normal. However, the patient did not exhibit the clinical manifestations of biotin deficiency. Specifically, erosive and xerotic lesions are typically seen periorificially and on the extremities in biotin deficient individuals. Alopecia and seborrheic dermatitis can be present as well. The initial lesions demonstrated by our patient were located on the knees, dorsal hands, and toes. Subsequently, only the dorsal toes were affected and there was never any periorificial involvement. Based on clinical and histopathological findings, biotin deficiency was not believed to be the etiology in this case.
Essential fatty acid deficiency results from inadequate intake of fatty acids which cannot be synthesized by the human body. It is seen with low-fat diets and insufficient protein intake. Scaly xerotic lesions and alopecia can be apparent with this condition. The diagnosis can be established clinically and by demonstrating decreased plasma levels of linoleic acid and arachidonic acid, and increased levels of eicosatrienoic acid.
Niacin deficiency is classically associated with the "3 Ds": dermatitis, diarrhea, and dementia. The cutaneous lesions can often be observed on sun-exposed areas including the face, neck, dorsal hands and feet. Erythema and blistering occur along with associated pain and pruritus. Diagnosis is established by noting decreased levels of plasma niacin.
Necrolytic migratory erythema is a cutaneous disorder often associated with an underlying glucagon-secreting α2-pancreatic islet cell tumor (glucagonoma). In absence of this tumor, the most commonly associated disorders have been reported to be hepatocellular dysfunction and hypoalbuminemia [8]. Specifically, the hepatic disorders were attributed to alcoholic liver disease and hemochromatosis. Additionally, infection with hepatitis B virus (HBV) has also been recognized as a causative factor [9]. Clinically, a migrating erythema with vesicles, bullae, and erosions can be seen on the face, abdomen, genital region and lower extremities. Additional symptoms include glossitis, stomatitis, anemia, alopecia, and diabetes. Individuals with glucagonomas often undergo clearance of these dermatologic manifestations upon resection of the tumor or administration of somatostatin or somatostatin analogs.
Currently, authors appear divided over the perception of NAE as a distinct entity from NME. Abdallah et al. [2] argued that the two are separate conditions based on clinical findings. Specifically, NME typically affects the intertriginous areas with less propensity for an exclusively acral distribution. However, Nofal et al. [4] suggested NAE to be a variant of NME and coined the term, "acral NME." They based this on similarities between the two disorders including: acral involvement, association with chronic hepatitis infection, and similar histological findings. Other authors agree with this argument and also consider NAE to be a form of NME [9]. Regardless of the exact categorization of NAE, it is important to recognize its clinical manifestations and the previously reported, consistent association with HCV infection. This may help lead to earlier diagnoses of subclinical hepatitis and the institution of proper treatment. Our case, however, suggests that NAE can occur without a concurrent HCV infection. It may therefore be prudent not to rule out the possibility of NAE simply based on seronegative hepatitis studies.
Different therapeutic options for NAE have been previously studied. Oral zinc has been reported to have varying degrees of efficacy ranging from: no effect [2] to mild/moderate improvements [3] to complete/near complete remissions [7, 10]. Abdallah et al. [7] reported complete remission within 2 months of initiating oral zinc therapy and a reduction of the dosage was followed by an exacerbation of symptoms. They recommended oral zinc as a less toxic form of therapy for patients with NAE.
Additionally, many clinicians have also reported successful outcomes using interferon α-2b. Specifically, complete/near complete improvements were noted with interferon α-2b alone [2], and also with combinations of interferon α-2b & zinc [5] and interferon α-2b & ribavirin [3, 6]. Khanna et al. [5] reported the first case of NAE in the United States and treated the patient with a combination of interferon α-2b and zinc. Complete remission was noted within six months and the clearance was still present after three and a half years. Furthermore, Hivnor et al. [6] treated a patient with NAE using interferon α-2b and ribavirin. Significant improvement was noted within two months despite a persistently elevated hepatitis C viral load. The near-complete remission was sustained at follow-up four years later.
Our patient represents a case of NAE based on clinicopathological features, but without the previously reported relationship with HCV infection. This suggests the possibility that NAE may also occur either as an isolated phenomenon or in association with another, as yet unidentified, pathological condition. Future reports may provide more clarification on this matter and perhaps identify any other disease entities with a pathophysiological connection to NAE.
References
1. el Darouti M, Abu el Ela M. Necrolytic acral erythema: a cutaneous marker of viral hepatitis C. Int J Dermatol. 1996 Apr;35(4):252-6. PubMed2. Abdallah MA, Ghozzi MY, Monib HA, Hafez AM, Hiatt KM, Smoller BR, Horn TD. Necrolytic acral erythema: a cutaneous sign of hepatitis C virus infection. J Am Acad Dermatol. 2005 Aug;53(2):247-51. PubMed
3. El-Ghandour TM, Sakr MA, El-Sebai H, El-Gammal TF, El-Sayed MH. Necrolytic acral erythema in Egyptian patients with hepatitis C virus infection. J Gastroenterol Hepatol. 2006 Jul;21(7):1200-6. PubMed
4. Nofal AA, Nofal E, Attwa E, El-Assar O, Assaf M. Necrolytic acral erythema: a variant of necrolytic migratory erythema or a distinct entity? Int J Dermatol. 2005 Nov;44(11):916-21. PubMed
5. Khanna VJ, Shieh S, Benjamin J, Somach S, Zaim MT, Dorner W Jr, Shill M, Wood GS. Necrolytic acral erythema associated with hepatitis C: effective treatment with interferon alfa and zinc. Arch Dermatol. 2000 Jun;136(6):755-7. PubMed
6. Hivnor CM, Yan AC, Junkins-Hopkins JM, Honig PJ. Necrolytic acral erythema: response to combination therapy with interferon and ribavirin. J Am Acad Dermatol. 2004 May;50(5 Suppl):S121-4. PubMed
7. Abdallah MA, Hull C, Horn TD. Necrolytic acral erythema: a patient from the United States successfully treated with oral zinc. Arch Dermatol. 2005 Jan;141(1):85-7. PubMed
8. Marinkovich MP, Botella R, Datloff J, Sangueza OP. Necrolytic migratory erythema without glucagonoma in patients with liver disease. J Am Acad Dermatol. 1995 Apr;32(4):604-9. PubMed
9. Kitamura Y, Sato M, Hatamochi A, Yamazaki S. Necrolytic migratory erythema without glucagonoma associated with hepatitis B. Eur J Dermatol. 2005 Jan-Feb;15(1):49-51. PubMed
10. Najarian DJ, Lefkowitz I, Balfour E, Pappert AS, Rao BK. Zinc deficiency associated with necrolytic acral erythema. J Am Acad Dermatol. 2006 Nov;55(5 Suppl):S108-10. PubMed
© 2008 Dermatology Online Journal