Imiquimod 5 percent cream and the treatment of cutaneous malignancy
Published Web Location
https://doi.org/10.5070/D34vw339w4Main Content
Imiquimod 5 percent cream and the treatment of cutaneous malignancy
Daniel Navi1 and Arthur Huntley2
Dermatology Online Journal 10 (1): 4
From the School of Medicine1, and the Department of Dermatology2, University of California Davis. achuntley@ucdavis.edu
Abstract
Imiquimod is a novel synthetic compound that is a member of the imidazoquinolone family of drugs. This class of drugs, which also includes a more potent member, resimiquimod (R-848), is unique having the properties of topical immune response modifiers and stimulators. Imiquimod resembles a nucleoside analogue and is known for its potent induction of endogenous antiviral pro-inflammatory mediators. This article is a review of the relevant literature as it relates to the off-label applications of imiquimod 5 percent cream for treatment of cutaneous preneoplastic and neoplastic conditions including its recently approved indication for treating actinic keratoses.
Introduction
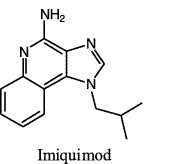
|
| Figure 1 |
|---|
Imiquimod is a novel synthetic compound that is a member of the imidazoquinolone family of drugs. This class of drugs, which also includes a more potent member, resimiquimod (R-848), is unique having the properties of topical immune response modifiers and stimulators. Imiquimod resembles a nucleoside analogue (fig.1) and is known for its potent induction of endogenous antiviral pro-inflammatory mediators [1]. Following publication of several prospective studies showing that topical application of imiquimod is a safe and effective treatment for external anogenital and perianal warts, in 1997 the Food and Drug Administration approved its licensure for these manifestations of human papilloma virus infection [2]. In 2004 imiquimod 5 percent received FDA approval for the treatment of actinic keratosis (AK). As of this writing it is still marketed in the United States as a 5 percent cream (Aldara™, 3M Pharmaceuticals) for its two FDA approved usages. In recent years the off-label use of imiquimod 5 percent cream in dermatological practice, however, has shown this unique drug to be efficacious in a many dermatological conditions. This article is a review of the relevant literature as it relates to the off-label applications of imiquimod 5 percent cream for treatment of cutaneous preneoplastic and neoplastic conditions including its recently approved indication for treating AK.
Mechanism of action
Both in vivo and in vitro studies point to the role of imiquimod as an immune modulator via its binding to the Toll receptor 7 (TLR-7) present on dendritic cells, macrophages, and monocytes [1]. Subsequent activation of these cells leads to release of pro-inflammatory mediators IFN-α, TNF-α, IL-1, IL-12, IL-6, IL-8, and IL-10, among others. These cytokines are believed to then drive the activation of the adaptive immune response toward the TH-1 or cell-mediated pathway and inhibit the TH-2 pathway. This modulation of the immune response, along with creation of an antiviral state including upregulation of NK-cell activity via induction of 2'5'oligoadenylate synthetase, is thought to be important for control of viruses and tumors [3]. Additionally, topical application of imiquimod has been shown to enhance the functional maturation and migration of Langerhan cells to regional lymph nodes, thus enhancing antigen presentation to naïve T cells and inducing a more specific immune response [1]. Recent findings shed some light on a possible direct action of imiquimod on basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). The addition of imiquimod resulted in the induction of Fas receptors on tumor cells, and is postulated to lead to their subsequent apoptosis via binding of FasR to the Fas ligand, induction by neighboring BCC cells, and infiltration of lymphocytes. Berman et al. demonstrated that BCC cells in 3 of 4 patients treated with imiquimod for a 2-week period were positive for FasR, while all 5 vehicle-treated patients had FasR negative BCC cells at the end of the treatment period [4]. A number of other studies also provide evidence for direct antineoplastic activity of imiquimod via induction of apoptosis in tumor cells, which normally have mechanisms that resist apoptosis. In one study tumor-specific apoptosis induced by imiquimod was demonstrated both in SCC cell lines as measured by a number of apoptotic signals (Bcl-2, capsases, etc.) and in BCC tumors in vivo [5].
Nature of published studies
The nature of the published studies on the off-label applications of imiquimod range from anecdotal case reports to phase III clinical trials. Because of its action inducing an inflammatory response favoring the apoptosis of tumor cells (i.e., the activation of TH-1 pathway, stimulation of NK cells and killer T cells, and maturation of dermal antigen presenting cells) it is not surprising that the majority of the published studies examine the efficacy of imiquimod 5 percent cream in treating cutaneous oncological lesions. Table 1 summarizes the current stage of research on off-label applications of topical imiquimod cream for such lesions.
| Cutaneous Lesion | Stage of Study |
| Actinic Keratosis | Phase III Clinical Trial |
| Superficial and Nodular BCC | Phase III Clinical Trial |
| Sclerodermiform BCC | Case Report |
| Bowen's Disease/SCC in Situ | Phase II Clinical Trial |
| Invasive Squamous Cell Carcinoma | Case Report |
| Lentigo Maligna | Case Report |
| Metastatic Melanoma | Case Report |
| Mycosis Fungoides | Case Report |
| Keratoacanthoma | Case Report |
| Extramammary Paget's Disease | Case Report |
Published findings
Actinic keratosis
Promising results of Phase II studies on actinic keratosis have been recently reported. One such study examined the efficacy of imiquimod for lesions on scalp, forehead, dorsal forearm, neck, and dorsal hand. The 5 percent cream was applied up to three-times per week for 12 weeks, although rest periods were allowed for subjects with troublesome local reactions. Of the treatment group, 84 percent experienced complete clinical and histological clearance of actinic keratoses, and 8 % had partial clearance. Recurrence after 1 year of treatment was seen in 10 % of patients [6]. In a similar study imiquimod was applied to lesion sites for 6-8 weeks depending on the severity of the application site reactions. All six patients in this study demonstrated histological clearing with no apparent signs of persisting lesions. No recurrences were observed during the 2-12 month followup period [7].
Salasche et al. performed an open-label study on the treatment of AKs and proposed a regimen of cycled therapy [8]. They used a rigid 4-week active intervention phase followed by a 4-week imposed rest period. During the treatment phase, imiquimod was applied once daily, 3 times a week. At the end of the 4-week rest phase, the treatment phase was repeated if any AKs were still present. They used up to three treatment phases. They concluded that their cycled approach of alternating 4 weeks of active therapy with 4 weeks of rest was able to achieve high efficacy while minimizing adverse effects.
Superficial basal cell carcinoma
The first dose-response study on the treatment of superficial basal cell carcinoma with imiquimod 5 percent cream was published in 1999. There were 35 patients randomized to several dosing regimens of imiquimod or a vehicle cream for a maximum treatment period of 16 weeks or 2 weeks following clinical clearance of lesions. Rest periods of up to 7 days were allowed for patients with severe localized reactions. Superficial BCC lesions cleared (on the basis of histological examination) in all patients (100 %) dosed twice a day, once a day, and three-times a week. Clearance was seen in 60 % of patients dosed twice weekly, 50 % of patients dosed once weekly, and 9 % of patients treated with vehicle. The median duration of treatment was 10 weeks for the twice-daily group, 13 weeks for once-daily group, and 14.5 weeks for three-times-weekly group [9].
Similar results were observed in later study encompassing a multicenter 6-week dose-response trial of 99 patients. Complete histological clearance was seen in 100 % (3/3) of patients in twice-daily regimen, 87.9 % (29/33) of patients in the once-daily regimen, 73.3 % (22/30) of patients in twice-daily three-times-per-week regimen, and 69.7 % (23/33) of patients in the once-daily, three-times-per-week regimen. The median duration of treatment for complete clearance was 10-16 weeks [10].
A larger Phase II study published in 2002 also assessed the efficacy of various dosing regimens on clearance of superficial BCC lesions. Patients were randomized to a 12-week treatment of lesions with imiquimod in one of four dosing groups. Histological examination of lesions 6-weeks post-treatment revealed complete response rates in 100 % (10/10), 87.1 % (27/31), 80.8 % (21/26), and 51.7 % (15/29) for patients in the twice-daily, once-daily, five-times-a-week, and three-times-a-week imiquimod groups, respectively, and 18.8 % (6/32) in the vehicle group. The authors conclude that daily or five-times-a-week dosing for 12 weeks is highly efficacious and has acceptable safety profiles [11]. To assess the effect of occlusion on response rates at low dosing frequencies a multicenter clinical trial published in 2002 randomized patients to one of 4 treatment arms: imiquimod application 2 or 3 days per week either with or without occlusion. The highest histological complete response rate of 87 % was seen in the 3-days-per-week with occlusion groups, but occlusion did not have a statistically significant effect on response rate at either dosing frequency. The response rate in 3-days-per-week groups, however, was significantly higher than the 2-days-per-week groups, which had response rates of 43 % and 50 % for patients with and without occlusion, respectively [12].
Nodular basal cell carcinoma
The occlusion study discussed above also included patients with nodular BCC lesions. In this group of patients the 3-days-per-week regimen with occlusion resulted in a complete histological clearance rate of 65 % versus 50 % in the no-occlusion group, a statistically insignificant finding. Furthermore, these response rates were not statistically different from those attained from the 2-days-per-week groups [12]. The lower overall efficacy of imiquimod 5 percent cream for the treatment of nodular BCC versus superficial BCC seen in this study has also been demonstrated in other studies. One such study was a Phase II clinical trial comparing efficacy of various dosing regimens in a 6-week study in Australia and New Zealand and a 12-week study in United States. In both studies histological examination of lesion site 6-weeks post-treatment showed the highest clearance rate in the once-daily-for-7-days-per-week groups, with 71 percent of patients in the 6-week study and 76 percent of patients in the 12-week study having complete response following treatment. As the authors point out these response rates were lower than the nearly 88 percent response rates seen in studies of superficial BCC lesions [13].
Sclerodermiform BCC
One case report demonstrating success in the treatment of a sclerodermiform BCC lesion on the right temple was found in the literature. Following 3-times-weekly application for 16 weeks, a histological examination of the lesion site showed only scar tissue. No recurrences were seen 9 months after completion of treatment [14].
| BCC subtype | Treatment duration | Dosing regimen | Percent complete histological clearance |
| Superficial BCC | 10-14.5 Weeks | 2 × a day | 100 % |
| once a day | 87.1-100 % | ||
| 3 × a week | 51.7-100 % | ||
| Nodular BCC | 6-12 Weeks | 3 × a week | 50-65 % |
| once a day | 71-76 % | ||
| Sclerodermiform BCC | 16 weeks | 3 × a week | 100 % |
Squamous cell carcinoma in situ (Bowen's disease)
A number of studies have addressed the efficacy of imiquimod 5 percent cream in the treatment of Bowen's disease (i.e., SCC in situ). One of the larger ones was a Phase II, open-label study of sixteen patients with single lesions located on their legs (15 patients) and shoulder (1 patient). This 2001 study assessed histological clearance of the lesions 6 weeks following a 16-week treatment regimen of once-daily application of imiquimod 5 percent cream. Although only ten patients completed 16 weeks of treatment (others self treated for 4-8 weeks), 93 percent (14 of 15) showed complete clearance in post-treatment biopsy specimens. Furthermore the treatment site was tumor free in the 6-month followup visit in all thirteen patients who attended the followup [15]. Such long-term recurrence-free resolution of Bowenoid lesions treated with imiquimod have been observed in a number of case reports. One such case report is of a patient with a nose lesion who had no histological sign of recurrence 12 months following a 9-week treatment with imiqimod [16]. Another study reported the case of a patient with SCC in situ of the penis who was treated with imiquimod once daily for a total of 24 days with a 1-month rest period after the first 11 days. The lesion was histologically resolved, and the patient was tumor free 18 months following treatment [17].
Invasive squamous cell carcinoma
We found one study in the literature supporting the efficacious role of imiquimod 5 percent for treatment of invasive SCC [14]. The two patients were immunosuppressed renal transplant patients, one with the lesion on the temple and the other on the sternum. The patient with temple lesion was treated three times a week for 12 weeks and showed scarring in a biopsy obtained at week 16 and no evidence of residual tumor at a six-month followup. A patient with SCC on sternum was treated three times a week for 5 weeks followed by twice a week for 7 weeks because of uncomfortable local reaction. This patient had complete clearance at week 12 and was tumor free at an 8-month followup.
Lentigo maligna
The first reported case on the therapeutic use of imiquimod 5 percent for lentigo maligna (LM) was in 2000 by Ahmed and Berth-Jones [18]. The patient was an elderly man reluctant to have surgery who was therefore treated with topical imiquimod. Total treatment period was 7 months with frequent rest periods allowed for localized reactions to subside. There was complete histological and clinical clearance at follow-up and no evidence of recurrence at 9 months. Other case reports have shown similar successes with LM lesions. One such case report involves a patient with recurrent LM who was initially treated with CO2 laser [19]. Imiquimod application once or twice a day for 3 months resulted in no apparent residual LM on two punch biopsies. Because a complete excision was not done and long-term efficacy is unknown, the authors recommended that their patient continue treatment of the primary site for 12 months. The efficacy of imiquimod for the treatment of LM also supported by a recent Phase I clinical trial consisting of 30 subjects with histologically confirmed lesions [20]. This study by Naylor et al. assessed clinical and histological clearance of lesions 4 weeks after a 12-week treatment regimen of once daily application of imiquimod. Of the 28 evaluable subjects 26 (93 %) had a complete response while 2 were nonresponders (i.e., they had no application site reaction and had persistent tumor visibly and on biopsy at week 16). Furthermore, of the 28 subjects 80 percent were followed and had no evidence of relapse at 1 year.
Metastatic melanoma
Perhaps the most promising off-label use of imiquimod has been for the treatment of cutaneous melanoma metastases, as evidenced by several recent published studies. There are three case reports that illustrate some of the complexities in this approach. The first reported case of successful treatment of cutaneous metastatic lesions was by Steinmann in 2000 [21]. The patient was a female with a lesion on the breast, too extensive for excision or radiotherapy. Three-times-weekly imiquimod was initiated along with continuing treatment with dacarbazine. There was complete clearing in imiquimod-treated lesions by 12 weeks and a biopsy at week 18 revealed apoptotic melanocytes surrounded with dense lymphocytic infiltrates [21]. In 2003 Wolf reported of two cases who had recurrent melanoma metastasis following several treatment modalities [22]. The first patient was an elderly woman with recurrent cutaneous metastasis of melanoma on the lower leg previously treated with wide excision and CO2-laser ablation. A staging procedure revealed no lymph node or visceral metastasis in this patient. The second patient was a middle-aged man with a lesion on the parietal scalp. He had a positive sentinel lymph node but no other evidence of metastasis. Following resection of the skin lesion and affected lymph nodes, adjuvant IL-2, and chemotherapy, he had multiple recurrences around surgical scars. As subsequent treatment modalities were unsuccessful, imiquimod treatment was initiated for both patients. Three-times-weekly application for 4 months resulted in complete clearing with no residual melanoma cells on biopsy in the first patient. She was recurrence free at 15 months. The same dosing regimen was applied to the second patient, and he was found to be biopsy negative for melanoma with no apparent recurrence at 8 months.
A 2002 study by Ugurel et al. involved a patient with satellite metastasis surrounding the excision site of a primary melanoma on the upper arm [23]. The patient refused further surgery, and daily application of imiquimod was initiated. The patient was monitored weekly by blood chemistries and hemograms, and ultrasound imaging of regional lymph nodes was done at 4-week intervals. After a total of 10 weeks of treatment, a biopsy of the lesion site revealed a lymphocyic infiltrate but no residual melanoma. Treatment was continued for 2 more weeks to achieve maintenance of therapeutic response. However, a biopsy-confirmed metastasis to the right axillary lymph node was discovered at week 12 post-therapy (and during continuation of treatment). The authors concluded that, although imiquimod may lead to eradication of skin metastasis of malignant melanoma, it may not prevent lymphogenous spread of tumor cells.
Mycosis fungoides
There is a single case report in the literature describing the successful treatment of a lesion of mycosis fungoides with imiquimod 5 percent cream. Most of the disease manifestations of the patient in this study had shown a clear remission during PUVA therapy combined with low-dose retinoids. The plaque on the face of this patient, however, was resistant to PUVA and retinoids, and thus imiquimod therapy was initiated. Daily application for 2 weeks resulted in complete clearance of the plaque, which remained recurrence free for a 12-month followup period [24].
Keratoacanthoma
Dendorfer et al. reported the successful therapeutic use of imiquimod 5 percent cream in four patients with 1-6 week histories of facial keratoacanthoma (KA) [25]. The treatment regimen was every-other-day application for 4-12 weeks (the endpoint was clinically apparent clearance). KA lesions fully regressed in all patients as assessed clinically with a treatment period of 4-6 weeks in three of the patients. Clinical clearance was confirmed histologically in two of the patients. No recurrences were seen during the 4-6 month followup period.
Extramammary Paget disease
Successful treatment of primary limited extramammary Paget disease (EMPD) in two patients was reported by Zamponga et al. in 2002 [26]. Lesions were located on the scrotum in the first patient and on the perineum and left inguinal area in the second patient. The first patient was treated for a total of 16 weeks and the second patient was treated every other day for 7.5 weeks. The first patient showed no clinical evidence of disease after 8 weeks of treatment, and a post-treatment biopsy revealed low-grade irritant dermatitis but no evidence of residual neoplasm. This patient remained disease free during the 1-year followup period. In the second patient, 5 biopsies were obtained 4 months after completion of therapy, and these revealed no evidence of Pagetoid cells and only mild chronic inflammation and dermal fibrosis. Similar findings were reported in a more recent case report by Qian et al. [27]. They observed complete clinical and histological resolution of noninvasive EMPD of the penis after 6 weeks of imiquimod application.
Common side effects and adverse reactions
Application-site reactions are commonly seen in patients undergoing topical imiquimod treatment. In most studies, the localized skin reactions are mild to moderate, and the severity is increased in a dose-dependent manner. The most frequently reported application-site reactions include pruritus, erythema, edema and discharge, papular rash, pain and tenderness, and occasionally hypopigmentation or hyperpigmentation. More severe localized reactions such as bleeding, crusting, erosions, and indurations occur more often in patients on a more intensive regimen. A dose-related severity of local reactions is illustrated in the study by Beutner et al. [8]. Rest periods were required by all patients applying imiquimod twice daily, half those on a once-daily regimen, one fourth of the three-times-weekly subjects, and by none of the other groups. Because such reactions have an effect on patient compliance, dosing regimens designed to minimize these untoward side effects are thought to achieve a relatively higher therapeutic response compared to more intensive regimens in most clinical trials.
Summary
Imiquimod is the first member of a new class of drugs that modulates and upregulates the immune system, thus potentiating an antiviral and antitumor activity. Although imiquimod is currently FDA-approved only for the treatment of actinic keratoses and anogenital warts, a growing body of evidence points to many other potential therapeutic applications of this drug. The results of phase III clinical trials and larger studies evaluating efficacy of imiquimod for the various above-mentioned conditions are awaited. There are several considerations involved in determining whether a patient with a cutaneous neoplastic or preneoplastic lesion might benefit from treatment with imiquimod. The patient should make an informed decision on treatment given information on the risks and benefits of current standard treatment for a particular condition compared to the risk-benefit profile of imiquimod.
When should imiquimod be considered for the treatment of cutaneous malignancy? There appears to be clear evidence of efficacy for Bowen's disease and basal cell carcinoma (especially the more superficial forms) [4, 5, 6, 7, 9, 11, 14]. Patients whose lesions have been unresponsive to other therapeutic modalities or who have had recurrence following standard therapy might also benefit from treatment with imiquimod. This treatment should also be considered for patients with contraindications to standard therapy.
References
1. Stanley, MA. Imiquimod and the imidazoquinolones: mechanism of action and therapeutic potential. Clin Exp Dermatol. 2002 Oct;27(7):571-7. PubMed2. Garland, SM. Imiquimod. Curr Opin Infect Dis. 2003 Apr;16(2):85-9. PubMed
3. Skinner, RB Jr. Dermatol Clin. 2003 Apr;21(2):291-300. PubMed
4. Berman B, Sullivan T, De Araujo T, and M. Nadji. Expression of Fas-receptor on basal cell carcinomas after treatment with imiquimod 5% cream or vehicle. Br J Dermatol. 2003 Nov;149 Suppl 66:59-61. PubMed
5. Schon M, Bong AB, Drewniok C, Herz J, Geilen CC, Reifenberger J, Benninghoff B, Slade HB, Gollnick H, and MP Schon. Tumor-selective induction of apoptosis and the small-molecule immune response modifier imiquimod. J Natl Cancer Inst. 2003 Aug 6;95(15):1138-49. PubMed
6. Walker, JK and C. Koenig. Is imiquimod effective and safe for actinic keratosis? J Fam Pract. 2003 Mar;52(3):184-5. PubMed
7. Stockfleth E, Meyer T, Beninghoff B, and E. Christophers. Successful treatment of actinic keratosis with imiquimod cream 5%: a report of six cases. Br J Dermatol. 2001 May;144(5):1050-3. PubMed
8. Salasche SJ, Levine N, Morrison L. Cycle therapy of actinic keratoses of the face and scalp with 5% topical imiquimod cream: An open-label trial. J Am Acad Dermatol. 2002 Oct;47(4):571-7. PubMed.
9. Beutner KR, Geisse JK, Helman D, Fox TL, Geinkel A, and ML. Owens. Therapeutic response of basal cell carcinoma to the immune response modifier imiquimod 5% cream. J Am Acad Dermatol. 1999 Dec;41(6):1002-7. PubMed
10. Marks R, Gebauer K, Shumack S, Amies M, Bryden J, Fox TL, and ML. Owens. Imiquimod 5% cream in the treatment of superficial basal cell carcinoma: results of a multicenter 6-week dose-response trial. J Am Acad Dermatol. 2001 May;44(5):807-13. PubMed
11. Geisse JK, Rich P, Pandya A, Gross K, Andres K, Ginkel A, and ML. Owens. Imiquimod 5% cream for the treatment of superficial basal cell carcinoma: a double-blind, randomized, vehicle-controlled study. J Am Acad Dermatol. 2002 Sep;47(3):390-8. PubMed
12. Sterry W, Rtzicka T, Herrera E, Takwale A, Bichel J, Andres K, Ding L, and MR Thissen. Imiquimod 5% cream for the treatment of superficial and nodular basal cell carcinoma: randomized studies comparing low-frequency dosing with and without occlusion. Br J Dermatol. 2002 Dec;147(6):1227-36. PubMed.
13. Shumack S, Robinson J, Kossard S, Golitz L, Greenway H, Schroeter A, Andres K, Amies M, Owens M. Efficacy of topical 5% imiquimod cream for the treatment of nodular basal cell carcinoma: comparison of dosing regimens. Arch Dermatol. 2002 Sep;138(9):1165-71. PubMed
14. Eklind J, Maschke J, Lindbrink P, Hengge UR, and U. Tartler. Imiquimod to treat different cancers of the epidermis. Dermatol Surg. 2003 Aug;29(8):890-6; discussion 896. PubMed
15. Mackenzie-Wood A, Kossard S, de Launey J, Wilkinson B, and ML. Owens. Imiquimod 5% cream in the treatment of Bowen's disease. J Am Acad Dermatol. 2001 Mar;44(3):462-70. PubMed
16. Wu JK, Siller G, Whitehead K. Treatment of Bowen's disease and basal cell carcinoma of the nose with imiquimod 5% cream. Australas J Dermatol. 2003 May;44(2):123-5. PubMed
17. Schroeder TL, and RD. Sengelmann. Squamous cell carcinoma in situ of the penis successfully treated with imiquimod 5% cream. J Am Acad Dermatol. 2002 Apr;46(4):545-8. PubMed
18. Ahmed I, and J. Berth-Jones. Imiquimod: a novel treatment for lentigo maligna. Br J Dermatol. 2000 Oct;143(4):843-5. PubMed
19. Chapman MS, Spencer SK, and JB. Brennick. Histologic resolution of melanoma in situ (lentigo maligna) with 5% imiquimod cream. Arch Dermatol. 2003 Jul;139(7):943-4. PubMed
20. Naylor MF, Crowson N, Kuwahara R, Teague K, Garcia C, Mackinnis C, Haque R, Odom C, Jankey C, Cornelison RL. Treatment of lentigo maligna with topical imiquimod. Br J Dermatol. 2003 Nov;149 Suppl 66:66-70. PubMed
21. Steinmann A, Funk JO, Schuler G, and P. von den Driesch. Topical imiquimod treatment of a cutaneous melanoma metastasis. J Am Acad Dermatol. 2000 Sep;43(3):555-6. PubMed
22. Wolf IH, Smolle J, Binder B, Cerroni L, Richtig E, Kerl H. Topical imiquimod in the treatment of metastatic melanoma to skin. Arch Dermatol. 2003 Mar;139(3):273-6. PubMed
23. Ugurel S, Wagner A, Pfohler C, Tilgen W, and U. Reinhold. Topical imiquimod eradicates skin metastases of malignant melanoma but fails to prevent rapid lymphogenous metastatic spread. Br J Dermatol. 2002 Sep;147(3):621-4. PubMed
24. Drummer R, Urosevic M, Kempf W, Kzakov D, and G. Burg. Imiquimod induces complete clearance of a PUVA-resistant plaque in mycosis fungoides. Dermatology. 2003;207(1):116-8. PubMed
25. Dendorfer M, Oppel T, Wollenberg A, and JC. Prinz. TTopical treatment with imiquimod may induce regression of facial keratoacanthoma. Eur J Dermatol. 2003 Jan-Feb;13(1):80-2. PubMed
26. Zamponga JC, Flowers FP, Roth WI, and AM. Hassenein. Treatment of primary limited cutaneous extramammary Paget's disease with topical imiquimod monotherapy: two case reports. J Am Acad Dermatol. 2002 Oct;47(4 Suppl):S229-35. PubMed
27. Qian Z, Zeitoun NC, Shieh S, Helm T, and AR. Oseroff. Successful treatment of extramammary Paget's disease with imiquimod. J Drugs Dermatol. 2003 Jan;2(1):73-6. PubMed
© 2004 Dermatology Online Journal

