Punch biopsies of melanoma: a diagnostic peril
Published Web Location
https://doi.org/10.5070/D34vb8x54sMain Content
Punch biopsies of melanoma: A diagnostic peril
Giovanni D Lorusso MD, Deba P Sarma MD, and Syeda F Sarwar MD
Dermatology Online Journal 11 (1): 7
Department of Pathology, Veterans Affairs Medical Center and Louisiana State University Health Sciences Center. glorus@lsuhsc.edu
Abstract
Excisional or incisional biopsies of melanoma are used to determine depth of tumor invasion and to plan subsequent treatment. Accurate determination of depth of melanoma invasion is critical for treatment decisions and prognosis. Incisional or punch biopsies can be perilous for histopathologic determination of invasion, and both over- and underestimation of invasion can occur when using incisional methods. Likewise, histologic factors can lead to over- and underestimation of invasion. Prognosis and treatment of melanoma are primarily determined by depth of tumor invasion. We discuss several scenarios that can lead to over- and underestimation of depth of invasion in incisional biopsy specimens. We therefore discourage incisional or punch biopsies of suspected melanoma and recommend that depth of invasion not be reported on these types of specimens.
Introduction
Pigmented skin lesions are important clinical entities both because of their frequency and because of their potential for malignancy. For suspected melanoma, some authors oppose the performance of incisional or punch biopsies out of concerns for metastatic spread [1, 2]. Although other authors dismiss these concerns [3, 4], we present another reason to discourage melanoma punch biopsy: accurate measurement of invasion is sometimes impossible on these specimens. The goal of a biopsy is both diagnosis of the lesion and, if melanoma, determining the depth of invasion. Determination of the depth of invasion of a melanoma is an important factor for treatment decisions and prognosis.
Breslow [5] (1970) identified tumor thickness as the single strongest prognostic factor for melanoma. He noted that tumors thinner than 0.76 mm rarely metastasized. Although other cutoff points have been proposed [6, 7], the rational for measuring melanoma invasion is Breslow's observation that deeper lesions have a higher propensity for metastasis than thinner lesions. Deeply invasive lesions are treated more aggressively.
Invasion should be measured from the top of the granular layer to the deepest point of the tumor [8]. A shave or punch biopsy may not include the full depth of the lesion. In this case a pathologist can state only that tumor extends to the deep margin of the specimen, and cannot accurately measure depth of invasion [9]. Wound healing following these incisional biopsies may disrupt the tumor nodule and render accurate staging impossible, even in the subsequent excision [10].
Report of our experience
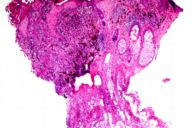
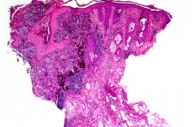
|
| Figure 1 |
|---|
| Original punch biopsy. Tumor thickness was measured as 1.5 mm in this biopsy. (H&E stain. Original magnification: 200x). |
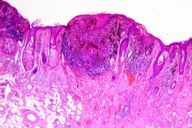
The patient was a 56-year-old man who presented to our hospital with a pathology report from an outside hospital. The report described a punch biopsy diagnosed as malignant melanoma of the earlobe with a 1.5-mm maximum depth of invasion. Excision of this lesion was subsequently performed. Maximum tumor invasion in the excision was measured as 0.8 mm. We requested the original biopsy slides to explain this discrepancy. We discovered that the original depth of invasion was measured in an area where the tumor descended along a hair follicle. The follicle was not readily evident in the level originally measured (Fig. 1) but was apparent in deeper levels (Fig. 2). Maximum invasion present in the excised lesion was 0.8 mm (Fig. 3).
Discussion
When considering depth of tumor invasion, melanoma cells extending along the epithelium of skin appendages are not measured [11]. Overstaging of the depth of invasion may occur in punch biopsies when tumor extension along appendages is present, but when the appendage is not appreciated. In some cases deeper sectioning reveals the appendage. The appendage may, however, not be present in a small biopsy. When melanocytes extending from the deepest point of the lesion into the deep dermis are accompanied by any squamoid proliferation, a nearby appendage should be suspected. If deeper sections fail to reveal the appendage, cytokeratin stain may confirm its presence [12]. If a skin appendage is present or suspected, the depth of invasion should not be reported. Overstaging results if tumor extension along an appendage is not recognized as such.
For a depth-of-invasion measurement to be valid, the deepest point of tumor invasion must be measured. Sampling error inherent in incisional or punch biopsies may mean that the deepest invasion is not contained in the sample. Gross examination cannot determine the deepest part of the lesion; yet this portion is critical to sample. Thus, sampling error may lead to understaging the lesion. Underestimation of invasion can lead to inadequate therapy; overestimation of invasion can lead to unnecessarily aggressive surgical treatment.
Summary
Excisional biopsy of suspected melanoma is encouraged whenever possible. Punch and incisional biopsies are not optimal specimens for suspicious pigmented lesions. Underestimation and overestimation of invasion are problems inherent to the nature of incisional and punch biopsies. Although the diagnosis is usually possible, attempting to evaluate the depth of invasion of these samples is treacherous and may lead to overestimation or underestimation of invasion. We discourage incisional or punch biopsies of suspicious pigmented lesions. We therefore propose that depth of melanoma invasion not be reported on punch or incisional biopsies.
References
1. Ironside P, Pitt TTE, Rank BK. Malignant melanoma: Some aspects of pathology and prognosis. Aust N Z Surg.47:70-75.1997.2. Rampen FHJ, van der Esch EP. Biopsy and survival of malignant melanoma. J Am Acad Dermatol1.2:385-388,1985.
3. Lederman JS, Sober AJ. Does biopsy type influence survival in clinical stage I cutaneous melanoma? J Am Acad Dermatol.13:983-987,1985.
4. Epstein E, Bragg K, Linden G. Biopsy and prognosis of malignant melanoma. JAMA. 208:1369-1371,1969.
5. Breslow A. Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann Surg.172:902-908,1970.
6. Day CL, Lew RA, Mihm, et al. The natural breakpoints for primary tumor thickness in clinical Stage I melanoma. N Engl J Med.305:1155,1981.
7. Buttner P, Garbe C, Bertz J, et al. Primary cutaneous melanoma: Optimized cutoff points of tumor thickness and importance of Clark's level for prognostic classification. Cancer.75:2499-2506,1995.
8. Elder DE, Murphy GF. Melanotic Tumors of the skin. Atlas of Tumor Pathology. Third series, Fascicle 2. Washington, DC; Armed Forces Institute of Pathology;1991:157.
9. Brown M. Staging and prognosis of melanoma. Semin Cutan Med Surg.16(2):113-121, 1997.
10. Kanzler MH, Mraz-Gernhard S. Treatment of primary cutaneous melanoma. JAMA.285:1819-1821,2001.
11. Elder D, Elenitsas R, Jaworsky C, Johnson B, eds. Lever's Histopathology of the skin. 8th ed. Philadelphia, PA: Lippincott-Raven; 1997: 672.
12. Breslow A. Prognostic factors in the treatment of cutaneous melanoma. J Cutan Pathol.6:208-212,1979.
© 2005 Dermatology Online Journal