Invasive primary ductal apocrine adenocarcinoma of axilla: A case report with immunohistochemical profiling and a review of literature
Published Web Location
https://doi.org/10.5070/D33zc2p6qnMain Content
Invasive Primary Ductal Apocrine Adenocarcinoma of Axilla: a case report with immunohistochemical profiling and a review of
literature
Brian Pucevich MD1, Steven Catinchi-Jaime MD2, Jonhan Ho MD3, Drazen M Jukic MD PhD4
Dermatology Online Journal 14 (6): 5
1. Department of Internal Medicine, Allegheny General Hospital, Pittsburgh, PA2. Department of Pathology, Conemaugh Valley Memorial Medical Center, Johnstown, PA
3. Department of Dermatology, University of Pittsburgh, Pittsburgh, PA
4. Departments of Dermatology and Pathology, University of Pittsburgh, Pittsburgh, PA
Abstract
Primary cutaneous ductal apocrine adenocarcinoma (PCDAA) is a rare malignant cutaneous neoplasm usually arising in areas of high apocrine gland density. There have been a total of 40 cases of primary apocrine adenocarcinoma reported in the literature; many are indolent and slowly- developing, but some are rapidly progressive. Primary cutaneous ductal apocrine adenocarcinoma has distinctive histologic and immunohistochemical characteristics and the treatment of choice for PCDAA is wide local excision with clear margins. Sentinel lymph node biopsy (SLNB) has also been used to determine prognosis and management. We present a case of a 92-year-old female with a 1-year history of a subcutaneous tumor of the right axilla, histologically consistent with a diagnosis of PCDAA arising in an accessory nipple. To our knowledge this is the first reported case of apocrine adenocarcinoma to develop in an accessory nipple.
Introduction
Primary cutaneous ductal apocrine adenocarcinomas are extremely rare malignant cutaneous neoplasms. Most of the neoplasms involve the axilla [1-18], but have been described arising on the scalp [8, 19], forehead [20], eyelid [21], upper lip [22], submandibular skin [23], chest [24, 25], pubic skin [26], nipple [24], arm [24], wrist [27], and finger [28]. Time course and evolution of these malignant neoplasms are as varied as their size. Most of these neoplasms are relatively indolent and present as slowly enlarging nodules [8, 12]. However, some are rapidly progressive and extremely aggressive showing development over weeks to months [19, 22]. Most of these carcinomas present as erythematous nodules or tumors. The standard of care for these lesions consists of total local excision with clear margins, with or without lymph node dissection [12]. The literature has also explored the role of sentinel lymph node biopsy in the determination of management and prognosis of these neoplasms [29].
For this case report, we present a case of a 92-year-old female with a right axillary mass of one year's duration that was eventually diagnosed as primary cutaneous apocrine adenocarcinoma arising in accessory mammary tissue (an accessory nipple).
Case Report
The patient, a 92-year-old female, was referred from her primary care physician to a dermatologist for evaluation of a right axillary mass. The mass had been present for over a year but had become increasingly bothersome due to increasing size over the past 8 months. Examination revealed a large erythematous, slightly tender 4 x 2 cm, multilobular subcutaneous mass of the right axilla without ulceration. No other lesions were observed in the contralateral axilla or either breast. Ultrasound examination of the right axilla revealed a 3.8 x 1.6 x 2.5 cm bilobed solid mass. Mammogram revealed no significant lesions in either breast, consistent with the physical exam. The patient was referred to a general surgeon and the mass was excised and sent to general pathology for further examination. Repeated ultrasound examinations showed no residual mass of the right axilla post biopsy. Initial histopathologic examination by a general pathologist yielded a diagnosis of moderately differentiated adenocarcinoma of the skin and subcutaneous tissue of the right axilla. The case was sent to our dermatopathology unit as a referral.
Histopathology
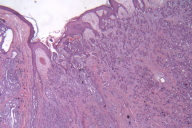
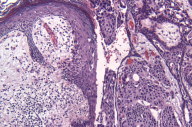
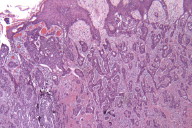
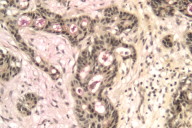
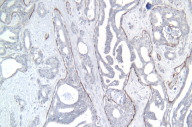
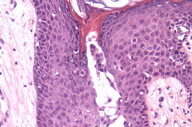
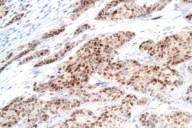
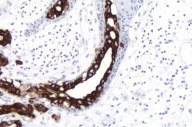
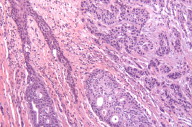
Histological exam of hematoxylin and eosin (H/E) stained biopsy material revealed an extremely interesting constellation of findings. There was a centrally located invagination of the epidermis that extended into a large duct- like structure (Figs. 1 and 2), coupled with 2 or 3 similar areas around it. There was a large, centrally located focus of malignant cells with clear ductal differentiation and some apocrine features. These cells exhibited obvious apocrine differentiation - decapitation secretion with prominent nucleoli. Numerous microcalcifications were also observed, as seen in the Fig. 3. The carcinoma exhibited glandular morphology reminiscent of the pattern sometimes exhibited by adenoid cystic carcinomas, but more poorly differentiated areas were also present (Fig. 3). Staining with mucicarmine stain revealed ductal (central luminal) positivity (Fig. 4). Centrally, the neoplastic tissue appeared more reminiscent of a nipple adenoma with a primarily papillary glandular morphology - further confirmed by a calponin stain that outlined the myoepithelial layer around each duct (Fig. 5). At the epidermal opening of the central duct in the epidermis, small infiltrating glands and single neoplastic cells with pagetoid spread were present. If this had been a shave biopsy, extramammary Paget disease would have been a primary consideration (Figs. 6 and 7).
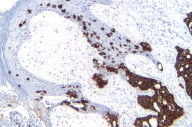
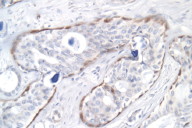
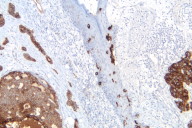
Immunohistochemical interpretation revealed positivity for GCDFP-15, EMA, mammaglobin, CK 7, AE1/AE3, and CAM 5.2. CD 34, CK 20 and S-100 were negative. The basal/myoepithelial cells stained with CK903, p63, calponin and somewhat with AMA. As previously mentioned calponin revealed a preserved myoepithelial layer around most of the residual ductal structures and most strikingly around the largest central duct (Figs. 5 and 8). The outside report indicated 3 + positivity for ER and PR and negativity for Her2/neu. An androgen receptor stain done at our facility was strongly positive (Fig. 9), as seen in carcinomas with apocrine derivation/differentiation. Notably, CK 7 not only outlined the central ductal structure, but also outlined areas of intraepidermal pagetoid spread (Figure 10) and an EMA stain showed identical findings (Figure 11), which can be compared with figure 12.
The differential diagnosis includes a primary cutaneous ductal apocrine adenocarcinoma as well as a primary mammary carcinoma arising in a supernumerary nipple. Because the mass had no connection to the breast as was confirmed by physical, mammographic, and ultrasound examination, and the lesion was intimately associated with a central duct opening to the epidermis, an axillary extension of a mammary carcinoma was not likely. Similarly, these features make the diagnosis of a metastatic process less likely.
Although one could argue that this indeed is a primary mammary carcinoma (arising in a vestigial breast - accessory nipple), we did feel that the evidence correlated better with reported cases of primary cutaneous apocrine adenocarcinomas.
Discussion
Apocrine adenocarcinoma is a rare malignant neoplasm arising in areas of high apocrine sweat gland density. To date, there have been 40 cases of apocrine adenocarcinoma reported in the literature. The first of these reports was published in 1944 [27]. Apocrine adenocarcinoma usually develops de novo (30 of the reported cases), but has also been observed to arise in association with other benign tumors, such as apocrine adenoma [13, 14, 15], apocrine hyperplasia [7, 9, 13], cylindroma [8], and nevus sebaceus [19]. Patients reported growth periods of between several weeks [19] to 40 years [6] before seeking medical care. Most neoplasms present as painless, solitary, nodules, 2-3 cm in size; there is a 2.6:1 male to female predominance in affected individuals. Most patients did not report any additional symptoms, but when present, they included pain [28], restriction of range of motion [12], ulceration [7, 13], and purulent discharge [7, 21].
As many as 17 of the 40 cases (42.5%) had lymph node metastases at presentation, which supports the use of SNLB in primary ductal apocrine carcinoma. Other metastases have been reported in the lungs [7, 16, 24], bones [19], brain [16], and parotid gland [23]. Additionally, cutaneous apocrine adenocarcinoma has a high incidence of local recurrence as seen in 11 of the 40 index cases (27.5%). Nine of these 11 cases (81.8%) of recurrence also exhibited metastatic disease which indicates that local recurrence commonly occurred in individuals with the most advanced disease. The relatively slow progression of tumor growth in apocrine adenocarcinoma and the relative lack of associated symptoms such as pain, swelling, or ulceration make clinical diagnosis of these tumors almost impossible. Biopsy and proper histologic examination are required to reach the proper diagnosis.
Histologic examination of cutaneous apocrine adenocarcinoma reveals characteristics common to the apocrine glands from which they arise. Typically, the carcinoma is composed of large cells with eosinophilic cytoplasm, hyperchromatic nuclei, mitotic figures, and apocrine decapitation secretion. Erratic ductal structures formed by malignant cells are commonly dilated. Glandular appearance of these neoplasms varies and includes papillary, cord-like, solid, or complex glandular patterns. Although 1 glandular type often predominates, multiple glandular morphologies can be present in the same neoplasm. Vascular invasion correlates with a high incidence of lymph node metastases [21]. Due to mucin production, PAS positive, diastase resistant material is often identified in the tumor cells with or without intracytoplasmic iron staining granules. Our case exhibited intracellular and extracellular mucin deposits. Immunohistochemical examination of these tumors commonly stains positively for GCDFP-15 (Gross cystic disease fluid protein), lysozyme and/or CD15.
Paties et al. proposed 3 criteria for histology-based diagnosis [8], including decapitation secretion, PAS-positive diastase resistant material identified in the cells and or lumen, and GCDFP-15 immunopositivity. Katagiri and Ansai [11] proposed this third criteria be modified to require that 2 of 3 antigens, GCDFP-15, lysozyme, and CD-15, be positive because glands of eccrine origin are occasionally GCDFP-15 positive. More recently, the mammaglobin immunohistochemical stain is commonly utilized instead or addition to the GCDFP. These criteria help to differentiate this neoplasm from eccrine and basal cell neoplasms, but not from metastatic neoplasms. An additional thing to keep in mind is that most of the published criteria deal with the differentiation of apocrine versus eccrine and basal cell neoplasms, rather than the differentiation of primary versus metastatic neoplasm.
Treatment of choice for primary cutaneous ductal apocrine carcinoma is wide local excision. Radiotherapy [5, 6] and chemotherapy [18] have been used as adjunctive treatments along with excision but have showed little benefit in mortality or morbidity in patients with either local or metastatic disease. There is only one case report of metastatic apocrine adenocarcinoma remission with chemotherapy [30]. Adjuvant radiotherapy may have a role in situations where the size of the tumor or involvement with certain vascular, neurologic or other anatomic structures would preclude total excision with clear margins [12]. Consideration should be given to SLNB biopsy and local lymph node dissection because up to 42 percent of the 40 cases in the literature search had lymph node metastasis on presentation. This rate of metastasis is similar to the 50 percent observed by Chamberlain [12] and the 44 percent Cooper [31] found in their respective reviews. Naturally, prophylactic nodal dissection does not reduce the incidence of local recurrence [20, 27]. Furthermore, SLNB has shown benefit in detecting subclinical lymph node metastasis in other sweat gland neoplasms [32]. Similarly, SLNB has been used to determine the need for local lymph node dissection in patients with apocrine adenocarcinoma [29]. However, we were unable to find clear evidence of SLN biopsy improving morbidity or mortality in the literature.
To our knowledge, this is the first case of primary cutaneous ductal apocrine carcinoma arising in vestigial mammary tissue (accessory nipple). We further characterize the immunohistochemical phenotype with a more comprehensive set of antibodies than previously published in the literature. Review of the literature shows one previous case arising in a nipple [24], but not an accessory nipple. The presence of the accessory nipple was not clinically suspected. It is reasonable to expect cutaneous apocrine adenocarcinoma to occur anywhere along the embryonic milk line and not only where apocrine glands are located in high density. This has been postulated to correlate with sites of occurrence of extramammary Paget disease in some individuals [33]. Also, as our case demonstrated, the appearance of an accessory nipple can be subtle and sometimes is only discovered upon careful histological examination. Therefore, cutaneous apocrine carcinomas may not be included in the initial differential diagnosis. Apocrine neoplasms can be expected to occur with higher frequency in accessory nipples due to a higher density of apocrine glands in locations where one would not expect to find such glands. Ultimately, this case illustrates the necessity of taking a biopsy of a sufficient size to permit the determination of the correct diagnosis.
References
1. Elliott, GB, Ramsey DW. Sweat gland carcinoma. Annals of Surgery 1956;144:99-106.2. Saigal RK, Khanna SC, Chandler J. Apocrine gland carcinoma in axilla: Report of a case. Indian Journal of Dermatology. 1971;37:177-180.
3. Futrell JW, Krueger GR, Chretien PB, Ketcham AS. Multiple primary sweat glan carcinomas. Cancer 1971;28:686-691.
4. Kipkie GF, Haust MD. Carcinoma of apocrine glands. Archives of Dermatology 1958;78:440-445.
5. Kuno Y, Numata T, Kanzaki T. Adenocarcinoma with signet ring cells of the axilla showing apocrine features: A case report. The American Journal of Dermatopathology. 1999;21:37-41.
6. Maynard JD. A case of carcinoma of an axilary apocrine gland. British Journal of Surgery. 1966;53:239-240.
7. Yoshida A, Kodama, Y, Hatanaka S, et al. Apocrine adenocarcinoma of the bilateral axillae. Acta Pathologica Japonica. 1991;41:927-932.
8. Paties C, Taccagni GL, Papotti M. Apocrine carcinoma of the skin: A clinicopathologic, imunohistochemical and ultrastructural study. Cancer 1993;71:375-381.
9. Nishikawa Y, Todusashi Y, Saito Y. A case of hamartomatous apocrine gland hyperplasia of both axillae. American Journal of Surgical Pathology. 1994;18:832-836.
10. Zehr KJ, Rubin M, Ratner L. Apocrine adenocarcinoma presenting as a large ulcerated axillary mass. Dermatol Surg. 1997;23:585-587.
11. Katagiri Y, Ansai S. Two cases of cutaneous apocrine ductal carcinoma of the axilla. Dermatology. 1999;199:332-337.
12. Chamberlain RS, Huber K, White JC, Travaglino-Parda R. Apocrine gland carcinoma of the axilla: Review of the literature and recommendations for treatment. American Journal of Clinical Oncology. 1999;22:131-135.
13. Miyamoto T, Hagari Y, Inoue S, Watanabe T, Yoshino T. Axillary apocrine carcinoma with benign apocrine tumours: a case report involving a pathological and immunohistochemical study and review of the literature. Journal of Clinincal Pathology. 2005;58:757-761.
14. Isei T, Yasuda H, Haradaet A, et al. A case of axillary apocrine adenocarcinoma arising from apocrine adenoma in the right axilla. Skin Cancer. 1995;10:156-159.
15. Amo Y, Kawano N. A case of ductal apocrine carcinoma in the left axilla with tubular apocrine adenoma in the right axilla. Journal of Dermatology. 2003;30:72-75.
16. Neumann L, Sorensen JA. Apocrine carcinoma of the axillae. The Scandanavian Journal of Plastic and Reconstructive Surgery. 1989;68:157-158.
17. Cameselle-Teijeiro J, Alfonsin-Barreiro H, Allegue F, Caeiro M. Apocrine carcinoma with signet ring cells and histiocytoid features: A potentially confusing axillary tumor. Pathology Research and Practice. 1997;193:717-720.
18. Kycler W, Korski K, Laski P, et al. Metastatic apocrine adenocarcinoma of the axillary area. Reports of Practical Oncology and Radiotherapy. 2006;11:299-302.
19. Domingo j, Helwig EB. Malignant neoplasms associated with nevus sebaceous of Jadassohn. Journal of the American Academy of Dermatology. 1979;1:545-546.
20. Smith CCK. Metastsizing carcinoma of sweat glands. British Journal of Surgery. 1955;43:80-84.
21. Shintaku M, Tsuta K, Yoshida H, Tsubura A, Nakashima Y, Noda K. Apocrine adenocarcinoma of the eyelid with aggressive biological behavior. Pathology International. 2002;52:169-173.
22. Hayes MM, Matisic JP, Weir L. Apocrine carcinoma of the lip: A case report including immunohistochemical and ultrastructural study, discussion of differential diagnosis, and review of the literature. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology and Endodonics. 1996;82:193-199.
23. Francisco J, Alfaro S, Oliveira D, Tonon S, Dias E. Apocrine carcinoma in the parotid gland and in the submandibular region. Brazilian Journal of Otorhinolaryngology. 2005;71:224-227.
24. Stout AP, Cooley SGE. Carcinoma of sweat gland. Cancer. 1951;4:521-536.
25. Baes H, Suurmond D. Apocrine sweat gland carcinoma: A report of a case. British journal of Dermatology. 1970;83:483-486.
26. Yamamoto O, Haratake J, Hisoka M. A unique case of apocrine carcinoma on the male pubic skin: Histopathologic and ultrastructural observations. Journal of Cutaneous Pathology. 1993;20:378-383.
27. Horn RC. Malignant papillary cystadenoma of sweat glands with metastases to the regional lymph nodes. Surgery. 1944;16:348-355.
28. Dhawan SS, Nanda VS, Grekin S. Apocrine adenocarcinoma: Case report and review of the literature. The Journal of Dermatologic Surgery and Oncology. 1990;16:468-470.
29. Hernandez JM, Copeland EM. Infiltrating apocrine adenocarcinoma with extramammary pagetoid spread. American Surgeon. 2007;73:307-309.
30. Mezger J, Remberger K, Schalhorn A, et al. Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Medical Oncology and Tumor Pharmacotherapy. 1986;3:29-34.
31. Cooper PH. Carcinoma of sweat glands. Pathology Annual. 1987;22:83-124.
32. Bogner PN, Fullen DR, Lowe L, Paulino A, Biermann JS, Sondak VK, Su LD. Lymphatic mapping and sentinel lymph node biopsy in the detection of early metastasis from sweat gland carcinoma. Cancer. 2003;97:2285-9.
33. Urabe A, Matsukuma A, Shimizu N, Nishimura M, Wada H, Hori Y. Extramammary Paget's disease. Journal of Cutaneous Pathology. 1990;17:257-265.
© 2008 Dermatology Online Journal