Psoriasis and hepatitis C treated with anti-TNF alpha therapy (etanercept)
Published Web Location
https://doi.org/10.5070/D33627s2b6Main Content
Psoriasis and hepatitis C treated with anti-TNF alpha therapy (etanercept)
Roberto Cecchi MD, Laura Bartoli MD
Dermatology Online Journal 12 (7): 4
Department of Dermatology, Spedali Riuniti, V. Matteotti 1, 51100 Pistoia, Italy. r.cecchi@usl3.toscana.it
Abstract
The treatment of severe psoriasis in patients with concomitant hepatitis-C virus infection is quite difficult because several systemic anti-psoriatic drugs are contraindicated owing to their liver toxicity. Recent observations in the literature suggest that etanercept is an effective and safe therapy for patients in this setting. We present a 45-year-old man with extensive plaque psoriasis and concurrent hepatitis C, successfully treated with 12-months of etanercept monotherapy. Regular monitoring of hepatic enzymes and viral load during the treatment disclosed no worsening of baseline values. This case confirms that etanercept may be a safe option for the therapy of patients with psoriasis and hepatitis-C virus infection.
The management of patients with psoriasis and concomitant hepatitis-C virus (HCV) infection is often difficult because treatments for hepatitis C, such as interferon-α and ribavirin, may trigger or exacerbate psoriasis and psoriatic arthritis. In addition, most systemic therapies for psoriasis, including immunosuppressants (cyclosporine) and other potentially hepatotoxic drugs (acitretin and methotrexate), are relatively contraindicated in HCV infection.
Recent data suggest that etanercept has an excellent safety profile for the treatment of severe psoriasis with psoriatic arthritis and concomitant hepatitis C virus (HCV) infection [1, 2]. We present an additional case of psoriasis and hepatitis C successfully and safely treated with etanercept monotherapy for 12 months.
Clinical synopsis
A 45-year-old man suffered from psoriasis vulgaris for 20 years and HCV (genotype 1) infection since 1996. In March 2005, a liver biopsy documented mild chronic hepatitis with no liver fibrosis. The patient had never received therapy for HCV infection. His psoriasis had been previously treated with different topical therapies including corticosteroids, salicylic acid, tar, dithranol, and vitamin-D analogues. Moreover, systemic medications with retinoids (acitretin), photochemotherapy, and narrow band ultraviolet B (UVB) gave only transient and unsatisfactory results. In 2002 he had begun treatment with oral cyclosporine, which was suspended after 3 months because of liver-enzyme elevation and arterial hypertension.
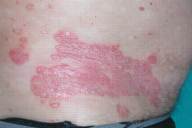
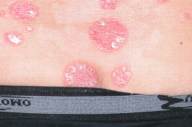
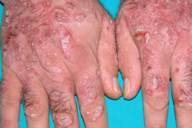
In September 2005 the patient had an exacerbation of his psoriasis with development of extensive psoriatic plaques on the trunk, buttocks, scalp, and limbs (Figs. 1A,1B, and 1C). His Psoriasis Area and Severity Index (PASI) score was 20. A tuberculin skin test was negative and a chest radiograph was within normal ranges.
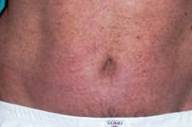
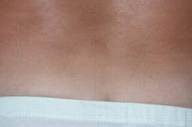
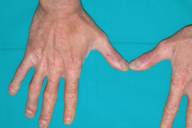
Given the severity of skin involvement and the contraindications for the use of other systemic drugs because of HCV infection, etanercept (Enbrel®) was administered at a dose of 50 mg subcutaneously twice weekly. A gradual improvement of psoriasis was seen within 2 weeks. After 3 months of therapy the PASI score had fallen to 6, and etanercept was reduced to 25 mg twice weekly (Figs. 2A, 2B, and 2C). The treatment was well-tolerated with no adverse effects. Serologic examinations were repeated at regular intervals and revealed no worsening of liver-function tests or HCV viral load during the 12-month therapy (see Table 1).
|
|
| Figure 1A | Figure 2A |
|---|
|
|
| Figure 1B | Figure 2B |
|---|
|
|
| Figure 1C | Figure 2C |
|---|---|
| Before and after treatment with etanercept. 1A and 2A: trunk. 1B and 2B: back. 1C and 2C: hands. | |
Discussion
Etanercept is a recombinant fusion protein of human tumor necrosis factor-α (TNF-α) receptor and immunoglobulin G1, which binds TNF-α and TNF-β molecules, preventing them from linking with their receptors [3]. It is approved by the US Food and Drug Administration for the treatment of psoriatic arthritis and psoriasis and other rheumatologic diseases, including rheumatoid arthritis (RA), juvenile RA, and ankylosing spondylitis.
Increased levels of circulating TNF-α and TNF receptors have been reported in patients with hepatitis C, although the exact role of this cytokine in the pathogenesis of HCV infection is still unclear [4, 5]. However, there are scarce data regarding the safety of biological drugs, including etanercept, in patients with hepatitis C [6]. Antagonists of TNF-α (etanercept and infliximab) have shown no adverse effect on liver function tests and viral load in a group of patients with RA and HCV infection during a short-term observation [7]. In addition, a recent case study has shown that etanercept is effective and safe as adjuvant treatment to interferon and ribavirin in the management of patients with hepatitis C [8].
To our knowledge, there are only four studies with seven reported cases in the current literature regarding the use of etanercept for the treatment of patients with psoriasis with psoriatic arthritis who also have HCV infection [1, 2, 9, 10]. In all these cases and in our patient, the drug has demonstrated a consistent efficacy with a rapid and significant clinical improvement of psoriasis, without worsening hepatitis or interfering with hepatitis therapy. In particular, etanercept does not appear to significantly affect liver-function parameters or HCV viral load during short-term administration.
Additional studies on larger series are necessary to better evaluate the long-term safety of this treatment in the setting of psoriasis and HCV infection.
References
1. Khanna M, Shirodkar MA, Gottlieb AB. Etanercept therapy in patients with autoimmunity and hepatitis C. J Dermatol Treat. 2003 Dec; 14(4): 229-32. PubMed2. Magliocco MA, Gottlieb AB. Etanercept therapy for patients with psoriatic arthritis and concurrent hepatitis C virus infection: report of 3 cases. J Am Acad Dermatol. 2004 Oct; 51(4): 580-4. PubMed
3. Mease PJ, Goffe BS, Metz J, VanderStoep A, Finck B, Burge DJ.Etanercept in the treatment of psoriatic arthritis and psoriasis: a randomised trial. Lancet. 2000 Jul 29; 356(9227): 385-90. PubMed
4. Nelson DR, Lim HL, Marousis CG, Fang JW, Davis GL, Shen L, Urdea MS, Kolberg JA, Lau JY. Activation of tumor necrosis factor-alpha system in chronic hepatitis C virus infection. Dig Dis Sci. 1997 Dec; 42(12): 2487-94. PubMed
5. Nasir A, Arora HS, Kaiser HE. Apoptosis and pathogenesis of viral hepatitis C: an update. In Vivo. 2000 Jan-Feb; 14(1): 297-300. PubMed
6. Calabrese LH, Zein N, Vassilopoulos D. Safety of antitumor necrosis factor (anti-TNF) therapy in patients with chronic viral infections: hepatitis C, hepatitis B, and HIV infection. Ann Rheum Dis. 2004 Nov; 63 Suppl 2:ii18-ii24. PubMed
7. Peterson JR, Hsu FC, Simkin PA, Wener MH. Effect of tumor necrosis factor alpha antagonists on serum transaminases and viraemia in patients with rheumatoid arthritis and chronic hepatitis C infection. Ann Rheum Dis.2003 Nov; 62(11): 1078-82. PubMed
8. Zein NN; Etanercept Study Group. Etanercept as an adjuvant to interferon and ribavirin in treatment-naïve patients with chronic hepatitis C virus infection: a phase 2 randomized, double-blind, placebo-controlled study. J Hepatol. 2005 Mar; 42(3): 315-22. PubMed
9. Rokhsar C, Rabhan N, Cohen SR. Etanercept monotherapy for a patient with psoriasis, psoriatic arthritis, and concomitant hepatitis C infection. J Am Acad Dermatol 2006 Feb; 54(2): 361-2. PubMed
10. De Simone C, Paradisi A, Capizzi R, Carbone A, Siciliano M, Amerio PL. Etanercept therapy in two patients with psoriasis and concomitant hepatitis C. J Am Acad Dermatol. 2006 Jun; 54(6): 1102-4. PubMed
© 2006 Dermatology Online Journal

