Persistent cutaneous hyperpigmentation due to hydroxychloroquinone one year after therapy discontinuation
Published Web Location
https://doi.org/10.5070/D32t07q0nwMain Content
Persistent cutaneous hyperpigmentation due to hydroxychloroquinone one year after therapy discontinuation
L Katie Morrison MD1,2, James J Nordlund MD1, Michael P Heffernan MD1
Dermatology Online Journal 15 (12): 15
1. Center for Clinical Studies, Houston, Texas2. Wright State Dermatology, Dayton, Ohio
Abstract
Hydroxychloroquine (HCQ) was initially indicated for the treatment of malaria, but more recently its anti-inflammatory and immune modulating properties have been utilized for treatment of multiple dermatologic and rheumatologic diseases. Mucocutaneous bluish-gray dyschromia is a rare side effect with HCQ and little information exists regarding its duration after drug discontinuation. The few existing case reports primarily describe small focal areas of discoloration. More extensive dyschromia has very rarely been reported with HCQ. We report a case of HCQ-induced dychromia diffusely involving the extremities that showed minimal resolution one year after treatment discontinuation.
Introduction
 |  |
| Figure 1 | Figure 2 |
|---|
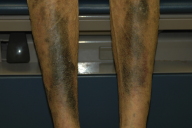
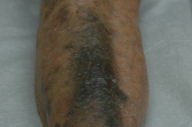
Hydroxychloroquine (HCQ) was initially indicated for the treatment of malaria. More recently its anti-inflammatory and immune modulating activities have been utilized in treating other diseases, such as lupus erythematosus, rheumatoid arthritis, Lyme arthritis, and Sjogren syndrome. Mucocutaneous bluish-gray discoloration is an uncommon side effect of HCQ treatment and little information exists regarding the duration of the dyschromia. The few case reports primarily describe small focal areas of discoloration, such as of the gingiva, hard palate, nails, temple, hands, or shins [1, 2, 3]. More diffuse bluish-gray hyperpigmentation has uncommonly been reported with HCQ. Also notable in this case is the persistence of the dyschromia one year after discontinuation of the medication.
Case report
A 67-year-old female with a history of subacute cutaneous lupus erythematosus began treatment with HCQ 200 mg twice daily. Other medications included thalidomide and topical clobetasol. Several months later, bluish-gray, non-pruritic, macular discoloration developed on the shins, forearms, and hands. Hydroxychloroquine was discontinued and 12 months later the dyschromia persisted with minimal resolution. The patient had no evidence of kidney or liver disease.
Discussion
Many of the potential side effects of HCQ pertain to pigmentation abnormalities, including hyperpigmentation, blue-gray mucocutaneous dyschromia, photodynamic reactions, whitening of the hair, and pigmented corneal deposits. The few reports of bluish pigmentation with HCQ described resolution within a few months or did not specifiy the duration of pigmentation after discontinuation of the drug [4, 5, 6].
Hydroxychloroquine is converted to an inactive metabolite in the liver and excreted in urine (25% in the active form) and bile. The plasma half-life is 32-50 days. Although some of the pigmentary effects are known to be dose dependent, in most cases dyschromia does not appear to be related to cumulative doses [7].
The biochemical basis of the dyschromia is unknown. Chloroquinone (CQ) has been demonstrated to bind to melanin, which is also the presumed etiology of pigment effects with HCQ. Iron has been found in two biopsy specimens of this blue-gray dyschromia [1, 4]. However, its presence was not demonstrated in another case [3].
The anti-malarials are commonly used to treat a wide variety of rheumatologic, dermatologic, and autoimmune diseases. It is important for clinicians to be aware of this less known pigmentary side effect of HCQ, which can occur in the absence of liver or kidney dysfunction; it is also important to remember the possibility of persistence years after drug discontinuation.
References
1. Veraldi, S., R. Schianchi-Veraldi, and G. Scarabelli, Pigmentation of the gums following hydroxychloroquine therapy. Cutis, 1992. 49(4): p. 281-2. [PubMed]2. Melikoglu, M.A., et al., Hydroxychloroquine-induced hyperpigmentation: a case report. J Clin Pharm Ther, 2008. 33(6): p. 699-701. [PubMed]
3. Puri, P.K., et al., Hydroxychloroquine-induced hyperpigmentation: the staining pattern. J Cutan Pathol, 2008. 35(12): p. 1134-7. [PubMed]
4. Rood, M.J., M.H. Vermeer, and T.W. Huizinga, Hyperpigmentation of the skin due to hydroxychloroquine. Scand J Rheumatol, 2008. 37(2): p. 158. [PubMed]
5. True, D.G., et al., Clinical images: Hydroxychloroquine-associated mucocutaneous hyperpigmentation. Arthritis Rheum, 2002. 46(6): p. 1698. [PubMed]
6. Amichai, B., A. Gat, and M.H. Grunwald, Cutaneous hyperpigmentation during therapy with hydroxychloroquine. J Clin Rheumatol, 2007. 13(2): p. 113. [PubMed]
7. Reynaert, S., J. Setterfield, and M.M. Black, Hydroxychloroquine-induced pigmentation in two patients with systemic lupus erythematosus. J Eur Acad Dermatol Venereol, 2006. 20(4): p. 487-8. [PubMed]
© 2009 Dermatology Online Journal

