Primary diffuse large B-cell lymphoma of the skin
Published Web Location
https://doi.org/10.5070/D32pm0882mMain Content
Primary diffuse large B-cell lymphoma of the skin
Andreas C Lazaris MD, Aphrodite Nonni MD, Irene Pathiaki, Efstratios Patsouris MD
Dermatology Online Journal 12 (4): 16
Department of Pathology, Medical School, National and Kapodistrian University of Athens, Greece. alazaris@med.uoa.grAbstract
B-cell lymphomas of the skin represent a wide spectrum of disorders. We present the microscopic and immunohistochemical features of a primary cutaneous B-cell lymphoma and classify it either as a diffuse large B-cell lymphoma or an indolent follicle-center-cell lymphoma according to the updated REAL/WHO system and the EORTC classification respectively. The former system is widely recommended; nevertheless, the EORTC system is also worth considering when planning management, because most data on long-term outcomes are based on it and it is considered to offer remarkable prognostic information.
Clinical synopsis
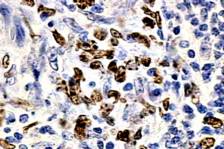 |
| Figure 3 |
|---|
| Figure 3. Strong Ki-67 immunoreactivity of many lymphoma cells. Large cell morphology is evident (immunoperoxidase stain, × 400). |
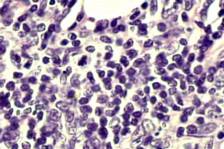
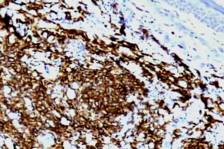
Diffuse large-cell lymphomas of B-cell lineage comprise a histogenetically heterogenous group. Some appear to begin as follicle-center-cell lymphomas, which can have varying proportions of large cells. Cutaneous lymphomas of B-cell lineage are frequent among extranodal B-cell lymphomas. Included within the category of cutaneous follicle-center-cell lymphomas (FCCLs) is reticulohistiocytoma of the back, so called Crosti lymphoma. The present case appeared as two purplish adjacent cutaneous nodules on the back of a middle-aged man, with a maximum diameters of 2.5 and 1.5 cm, respectively. Histologically, the nodules consisted of a heavy diffuse infiltrate focally extending to the subcutis, but not to the epidermis. The dense dermal lymphoid infiltrates were separated from the epidermis by a Grenz zone of uninvolved papillary dermis. Numerous centroblasts, immunoblasts, and large centrocytes (Fig. 1) as well as few reactive T cells were observed; no neoplastic follicles were seen. Mitotic figures were frequent. Immunohistochemically, neoplastic cells were CD20(L26)-positive (Fig. 2) and immunoglobulin (Ig)-negative. Proliferation marker Ki-67 was expressed in more than 70 percent of neoplastic cells (Fig. 3). CD3 and bcl-2 protein were negative. A weak cytoplasm and membrane expression of CD30 (an inducible marker of lymphocytic activation) was noticeable in few neoplastic cells. The patient's systemic workup was negative. The above findings were consistent with the diagnosis of a diffuse large B-cell lymphoma (DLBCL) [according to the updated Revised European-American classification of Lymphoid Neoplasms/World Health Organization (WHO) Classification (REAL/WHO classification)] [1] equivalent to the diagnosis of an indolent follicle-center-cell lymphoma (FCCL) [according to the European Organization for Research and Treatment of Cancer (EORTC) Classification [2], the latter classification applying only to primary cutaneous lymphomas. It is estimated that approximately 10 percent of systemic B-cell lymphomas involve the skin secondarily. This is usually a poor prognostic sign. Systemic dissemination was excluded for our patient after further clinical and laboratory examination (absence of lymphadenopathy, clear marrow); actually, the absence of bcl-2 protein overexpression had already supported the diagnosis of primary cutaneous disease. The patient has been treated with multi-agent chemotherapy (CHOP as chemoprophylaxis) and remains free of relapse after an 18-month followup.
Discussion
In contrast to their nodal counterparts, these predominantly diffuse large-cell variants of FCCL do not appear to behave in a particularly aggressive fashion with one exception; available data suggest that such lymphomas are biologically distinct from morphologically similar primary cutaneous tumors occurring on the lower legs, mainly of elderly females. This latter form of lymphoma displays relatively increased aggressiveness with a 5-year survival of about 50-60 percent. Despite large cell histology, patients with non-leg FCCL (EORTC) are expected to have an indolent course and an overall 5-year survival in excess of 90 percent [3]. Such good-prognosis patients should be treated as for WHO follicle center lymphoma, i.e., predominantly with radiotherapy [4]. However, the monoclonal antibody to CD20, rituximab, has been reported to give successful results in FCCL (EORTC) and DLBCL (REAL/WHO) of the skin. Aggressive treatment is certainly recommended in patients when large cell histology is accompanied by independent adverse prognostic factors (i.e., round cell morphology and location on the leg) [4]. Thus, the EORTC reserves the use of DLBCL to the specific aggressive clinical entity involving one or both legs of the elderly.
As far as the differential diagnosis of DLBCL is concerned [5], non-lymphoid malignancies need to be considered before the diagnosis of DLBCL is made. With regard to lymphoid lesions, the differential diagnosis of DLBCL versus cutaneous lymphoid hyperplasia is usually easy, particularly when patterns of Ig expression are taken into account (DLBCL: monotypic or Ig-negative B cells; cutaneous lymphoid hyperplasia: polytypic B cells). In addition, a rare variant of DLBCL that can pose difficulties is the so called T-cell-rich B-cell lymphoma; the neoplastic cells of this condition are large B lymphocytes, but their presence is obscured by an infiltrate of small reactive cells. Finally, the distinction between large-cell T- or B-cell lymphomas may not be of clinical importance. Most large cell lymphomas are readily typed, expressing either the T-cell antigen CD3 or such B-cell markers as CD20 (L26)—as in the present case—and/or CD79a.
References
1. Jaffe ES, Harris NL, Stein H, Vardiman JWE, eds. World Health Organization Clas-sification of Tumours. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon, Pa: IARC Press, 2001.2. Willemze R, Kerl H, Sterry W, Berti E, Cerroni L, Chimenti S, Diaz-Perez JL, Geerts ML, Goos M, Knobler R, Ralfkiaer E, Santucci M, Smith N, Wechsler J, van Vloten WA, Meijer CJ. EORTC classification for primary cutaneous lymphomas: a proposal from the cutaneous lymphoma Study Group of the European Organization for Research and Treatment of Cancer. Blood 1997;90:354-71.
3. Connors JM, His ED, Foss FM. Lymphoma of the skin. Hematology (Am Soc He-matol Educ Program) 2002;263-82.
4. Prince HM, Yap LM, Glum R, McCormack C. Primary cutaneous B-cell lympho-mas. Clin. Exp. Dermatol. 2002;28:8-12.
5. LeBoit E, McCalmont TH. Cutaneous lymphomas and leukemias. In: Lever's Histo-pathology of the Skin. Elder D, Elenitsas R, Jaworsky Chr, Johnson B Jr, eds. 8th edn. Lippincott, Philadelphia, 1997 p. 805-46.
© 2006 Dermatology Online Journal