Fatal Henoch-Schonlein Purpura in an adult related to bowel perforation: Report and review of the literature
Published Web Location
https://doi.org/10.5070/D32f50n7g6Main Content
Fatal Henoch-Schonlein Purpura in an adult related to bowel perforation: Report and review of the literature
Una Miniter BS, Yoon-Soo Cindy Bae-Harboe MD, Jennifer G Powers MD, Shannon M Campbell DO, Lynne J Goldberg MD
Dermatology Online Journal 18 (10): 9
Boston University Boston, MassachusettsAbstract
BACKGROUND: Henoch-Schonlein purpura is an idiopathic, IgA associated, systemic small-vessel vasculitis characterized by the clinical tetrad of palpable purpura, arthralgias, renal dysfunction, and abdominal pain. Whereas Henoch-Schonlein is an overwhelmingly pediatric disease, its rare diagnosis in adults carries a much higher morbidity and mortality. OBSERVATIONS: We describe a 52-year-old man with biopsy proven Henoch-Schonlein who expired from bowel perforation. CONCLUSIONS: Severe gastrointestinal complications and death from gastrointestinal involvement by Henoch-Schonlein purpura is rare. The authors surmise that multiple co-morbidities may have contributed to our patient’s demise.
Introduction
Henoch-Schonlein purpura (HSP) is a small vessel vasculitis characterized clinically by palpable purpura, arthralgias, renal dysfunction, and abdominal pain, and histologically by perivascular deposition of IgA around dermal blood vessels. In over 90 percent of cases, the disease is found in children less than 10 years old with a peak occurrence at 3-5 years [1, 2]. Whereas the etiology of HSP is unknown, it is speculated to be triggered by infection, particularly in children, with Group A beta-hemolytic strep present in 50 percent of patients in one study [3]. Infectious precipitants in adults include GI pathogens such as various bacterial and viral infections, malignancies (particularly solid tumors of the lung and prostate and hematologic malignancies), and medications including angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists (losartan), antibiotics (clarithromycin), and nonsteroidal anti-inflammatory drugs [2, 4, 5, 6]. Adults with HSP have a higher incidence of complications and death.
Case report
A 52-year-old man with a past medical history of cirrhosis, intravenous drug use (IVDU), alcohol abuse, obesity, and hypertension was admitted for osteomyelitis of the right fourth toe. On hospital day two, the dermatology service was consulted for the sudden onset of a purpuric rash. The eruption began on the right arm where a blood pressure cuff was located and subsequently spread to involve his arms, legs, face, and trunk.
 |  |
| Figure 1 | Figure 2 |
|---|
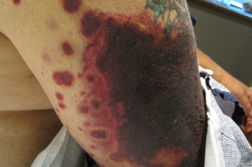
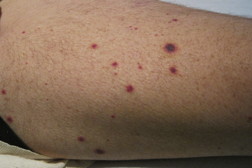
His examination was significant for multiple purple-to-black patches with erythematous borders on the extremities, bilateral cheeks, and nasal tip (Figure 1), as well as scattered, discrete purpuric macules and papules on the right lateral abdomen and extremities (Figure 2). There were several ecchymoses at the distal portion of his toes bilaterally and multiple 0.3 cm to 1.5 cm purpuric papules and plaques covering the plantar surface of his left foot. Several petechiae and erosions were observed on the hard palate.
 |  |
| Figure 3 | Figure 4 |
|---|
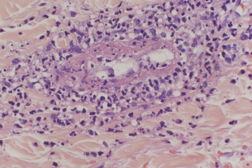
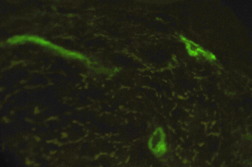
Two 4 mm punch biopsies were taken from the right thigh and sent for light microscopy, tissue culture, and direct immunofluorescence (DIF). The biopsy revealed epidermal necrosis, a superficial and deep perivascular and interstitial neutrophilic infiltrate, leukocytoclasia, extravasated erythrocytes, fibrinoid necrosis of blood vessel walls, and focal luminal occlusion of dermal blood vessels, diagnostic of small vessel vasculitis (Figure 3). Direct immunofluorescence revealed granular deposition of IgA and C3 around papillary dermal blood vessels, consistent with HSP (Figure 4).
During his hospital stay, the patient developed renal insufficiency (Cr 2.3 mg/dL), arthralgias, and multiple episodes of bloody diarrhea. He was also found to have positive IgG anti-cardiolipin antibodies, low levels of cryoglobulins (cryocrit <1%), elevated homocysteine levels (27.8 umol/L; nl 6-16 umol/L), low Protein S and Protein C activity (29% and 14%, respectively), and low C3 and C4 levels (C3 27 mg/dL; nl 79-160 mg/dL and C4 9; nl 17-48 mg/dL respectively). His stool culture was positive for Campylobacter. Upper endoscopy revealed purpuric raised lesions in the hypopharynx. His renal function improved on oral corticosteroids. On hospital day 17, dizziness and a significant drop in hemoglobin prompted an x-ray computed tomography (CT) of the abdomen, which revealed free air suggestive of a perforated viscus. Over the next 4 days, the patient underwent multiple resections of the jejunum and ileum, appendectomy, and end ileostomy. Patchy ischemia and foci of perforation of the jejunum and ileum, a shrunken, cirrhotic liver, extensive portal hypertension, caput medusae, and retroperitoneal varicosities were noted intraoperatively. One week later, the patient developed a fever, tachycardia, tachypnea, and progressively worsening hypotension and expired. The post-mortem examination of the small intestine revealed foci of small bowel perforation and invasive candidiasis. No changes to suggest vasculitis were noted.
Discussion
Generally, HSP is a benign, self-limiting process in children with an excellent prognosis. The major morbidity is related to renal involvement with progression to renal insufficiency occurring in approximately 5-15 percent of children [7-13]. Whereas adults are much less commonly affected, they display higher morbidity and mortality (up to 25%). This is often related to renal insufficiency, which is seen in about 30 percent of adult cases [13-17].
The reported incidence of GI involvement in patients with HSP ranges from up to 56 percent to 85 percent [18, 19, 20]. Symptoms include abdominal pain, nausea, vomiting, diarrhea, hematochezia, melena, and pancreatitis [1, 19, 20]. In a report by Saulsbury of 100 children with HSP, 63 percent had GI involvement, with half of these having GI bleeding and ten of these having gross bloody or melanotic stools [1]. In 19 (30%), the GI symptoms preceded the rash. In a review by Ebert, GI bleeding was seen in 18-52 percent of patients, and only occasionally required endoscopic or surgical therapy. In both reports, GI bleeding was always accompanied by abdominal pain [1, 4]. Seven of 16 patients (44%) with HSP reported by Esaki, et al had GI involvement, manifest in colicky pain, nausea, vomiting, and melena [20]. Chen reported GI involvement in 78 percent of 208 children with HSP, 5 percent of which had severe complications [21]. IgA-related enteropathy has also been reported in the absence of skin findings [22, 23].
GI symptoms are thought to occur from several mechanisms. One is vasculitis of the GI tract, leading to edema and hemorrhage of the bowel wall [1]. Whereas mesenteric vasculitis has a high risk of necrosis and massive GI bleeding, some feel it is uncommon [24]. Obstruction, presumably from ischemia, can occur, as can cholecystitis from vasculitis of the gallbladder [25, 26]. Low levels of Factor XIII are common and can cause abdominal pain and bleeding [27, 28]. Endoscopic biopsies have sometimes revealed GI vasculitis, particularly in the small intestine, although non-specific biopsy findings have also been reported and may even be more common [19, 20, 29-31]. Of 11 biopsies taken from 11 patients reported in an endoscopic study of HSP by Nishiyama et al, all showed non-specific findings [31].
Rare but serious complications of GI involvement by HSP in children include intussusception, bowel infarction, perforation, massive GI bleeding, protein-losing enteropathy, and pancreatitis [1, 21]. It had been said that in adults, surgical intervention for GI involvement by HSP is rarely required [18]. However, patients with severe GI complications and death have been reported [7, 23, 32, 33]. In 1990, Lawes and Wood reported a 41-year-old man with a purpuric eruption, arthralgias, abdominal pain, vomiting and GI bleeding who had 45 centimeters of infarcted bowel resected [18]. The authors state that early involvement and frequent assessments by the surgical team is crucial to avoid abdominal catastrophes. Chan et al reported a 57-year-old woman with biopsy proven HSP of the skin and kidney who died following resection of the distal ileum for gangrene because of small to medium vessel vasculitis (although no IgA was documented) despite corticosteroid therapy [32]. Lippl et al reported a 56-year-old man with biopsy proven HSP who developed abdominal pain, profuse GI bleeding from a submucosal jejuna artery, and worsening renal function who required resection of a 10 cm section of jejunum [30]. The histopathologic findings were felt to be “consistent with treated vasculitis.” In a series of 57 adult patients (16 and over), Tancrede-Bohin, et al reported two deaths, both in elderly patients from intestinal infarction [33]. In a retrospective study of 250 patients with HSP nephritis requiring biopsy, 13 (11%) had serious GI involvement leading to transfusion, surgery or death [7]. Lee et al reported a 60-year-old man who had several GI complications over time, including GI bleeding, ileal microperforation, small bowel obstruction and ileal stricture [29]. It was initially assumed that the microperforation was from sepsis, because his GI symptoms preceded his purpuric eruption. However, both skin and terminal ileal biopsies demonstrated leukocytoclastic vasculitis. Of 15 adult cases of HSP with GI involvement reported by Nakamura, 4 required surgical resection, all of the ileum, and 2 died [23]. Because both deaths were in elderly patients, they suggest that advanced age might be a risk factor for poor prognosis in HSP.
Although our patient’s skin biopsy findings were classic for HSP, his skin lesions were unusual in their location and large size. Skin lesions in HSP typically occur on the buttocks and lower extremities, and are rare on the face [7]. Lesions of HSP are indistinguishable from other small vessel vasculitides and typically consist of discrete or focally confluent purpuric papules. Our patient had such lesions, but in addition exhibited large, purpuric dusky plaques more suggestive of a thrombotic vasculopathy. It is likely that in addition to HSP, our patient was admitted in an acquired hypercoagulable state. He had several causes of hypercoagulability that might have contributed to atypical skin lesions, including obesity, diabetes, cirrhosis, recent surgery (toe amputation), chronic infection/sepsis (osteomyelitis), and the lab findings of hyperhomocysteinemia, Protein S and C deficiency, cryoglobulinemia, and anticardiolipin antibodies.
We report a 52-year-old male who died because of GI complications of HSP. Our patient was not elderly and did not have known renal disease at the time of presentation, which are known risk factors for severe GI disease and death associated with HSP. We suspect that numerous comorbidities contributed to his demise and suggest that multiple medical problems may put adult HSP patients at increased risk of severe GI complications including death.
References
1. Saulsbury FT. Henoch-Schonlein purpura in children: report of 100 patients and review of the literature. Medicine. 1999:78;395-409. [PubMed]2. Gonzalez LM, Janniger CK, Schwartz RA. Pediatric Henoch-Schonlein purpura. Int J Dermatol. 2009 Nov;48(11):1157-65. [PubMed]
3. Saulsbury FT. Henoch-Schonlein pupura in children. Report of 100 patients and review of the literature. Medicine (Baltimore). 1999 Nov; 78(6):395-409. [PubMed]
4. Ebert EC. Gastrointestinal manifestations of Henoch-Schonlein Purpura. Dig Dis Sci. 2008 Aug;53(8):2011-9. Epub 2008 Mar 20. [PubMed]
5. Fervenza FC. Henoch-Schonlein purpura nephritis. Int J Dermatol. 2003 Mar;42(3):170-7. [PubMed]
6. Borras-Blasco J, Enriquez R, Amoros F, et al. Henoch-Schonlein purpura associated with clarithromycin. Int J Clin Pharmacol Ther. 2003 May; 41(5): 213-6. [PubMed]
7. Pillebout E, Thervet E, Hill G, Alberti C, Vanhille P, Nochy D. Henoch-Schonlein Pupura in adults: outcome and prognostic factors. J Am Soc Nephrol. 2002 May; 13(5): 1271-8. [PubMed]
8. Linskey KR, Kroshinsky D, Mihm MC, Hoang MP. Immunoglobulin-A-associated small vessel vasculitis: A 10 year experience at the Massachusetts General Hospital. J Am Acad Dermatol. 2011 Jul 29: [Epub ahead of print]. [PubMed]
9. Meadow SR, Glasgow EF, White RH, Moncrieff MW, Cameron JS, Ogg CS: Schonlein-Henoch nephritis. Q J Med. 1972 Jul; 41(163): 241-58. [PubMed]
10. Niaudet P, Habib R. Schonlein-Henoch purpura nephritis: prognostic factors and therapy. Ann Med Interne. 1994;145(8): 577-80. [PubMed]
11. Yoshikawa N, White RH, Cameron AH. Prognostic significance of the glomerular changes in Henoch-Schoenlein nephritis. Clin Nephrol. 1981 Nov;16(5):223-9. [PubMed]
12. Goldstein AR, White RH, Akuse R, Chantler C. Long-term follow-up of childhood Henoch-Schonlein nephritis. Lancet. 1992 Feb 1;339(8788): 280-2. [PubMed]
13. Scharer K, Krmar R, Querfeld U, Ruder H, Waldherr R, Schaefer F. Clinical outcome of Schonlein-Henoch purpura nephritis in children. Pediatr Nephrol. 1999 Nov;13(9): 816-23. [PubMed]
14. Hung SP, Yang YH, Lin YT, Wang LC, Lee JH, Chiang BL. Clinical manifestations and outcomes of Henoch-Schonlein purpura: a comparison between adults and children. Pediatr Neonatol. 2009 Aug;50(4):162-8. [PubMed]
15. Jithpratuck W, Eishenawy Y, Saleh H, Youngberg G, Chi DS, Krishnaswamy G. The clinical implications of adult-onset henoch-schonlein purpura. Clin Mol Allergy. 2011 May 27;9(1):9. [PubMed]
16. Fogazzi GB, Pasquali S, Moriggi M, et al. Long-term outcome of Schonlein-Henoch nephritis in the adult. Clin Nephrol. 1989 Feb; 31(2):60-6. [PubMed]
17. Phillip R, Lugmani R. Mortality in systemic vasculitis: a systematic review. Clin Exp Rheumatol. 2008 Sep-Oct;26(5 Suppl 51):S94-104. [PubMed]
18. Lawes D, Wood J. Acute abdomen in Henoch-Schonlein purpura. J R Soc Med. 2002 Oct;95(10):505-6. [PubMed]
19. Banerjee B, Rashid S, Singh E, Moore J. Endoscopic findings in Henoch-Schonlein purpura. Gastrointest Endosc. 1991 Sep-Oct;37(5):569-71. [PubMed]
20. Esaki M, Matsumoto T, Nakamura S, et al. GI involvement in Henoch-Schonlein purpura. Gastrointest Endosc. 2002 Dec;56(6):920-3. [PubMed]
21. Chen SY, Chang KC, Yu MC, Asueh S, Ou LS. Pulmonary hemorrhage associated with Henoch-Schonlein purpura in pediatric patients: case report and review of the literature. Semin Arthritis Rheum. 2011 Oct;41(2):305-12. [PubMed]
22. Kato S, Ebina K, Naganuma H, Sato S, Maisawa S, Nakagawa H. Intestinal IgA deposition in Henoch-Schonlein purpura with severe gastro-intestinal manifestations. Eur J Pediatr. 1996 Feb;155(2):91-5. [PubMed]
23. Nakamura S, Hisamatsu T, Kikuchi J, et al. A Case of IgA-related Enteropathy Complicated with Gastrointestinal Bleeding and Progressive IgA Nephropathy: A Possible Variant Henoch-Schonlein Purpura? Intern Med. 2010 Aug;49(16):1755-61. [PubMed]
24. Wang L, Huang FC, Ko SF, Cheng MT. Successful treatment of mesenteric vasculitis caused by Henoch-Schonlein purpura with methylprednisolone pulse therapy. Clin Rheumatol. 2003 May;22(2):140-2. [PubMed]
25. Kumon Y, Hisatake K, Chikamori M, Hara H, Numata Y, Ya- mano T, Ohno F. A case of vasculitic cholecystitis associated with Schonlein-Henoch purpura in an adult. Gastroenterol Jpn. 1988 Feb;23(1):68-72. [PubMed]
26. Hoffmann JC, Cremer P, Preiss JC, Ebhardt H, Kruschewski M, Buhr HJ, Zeitz M. Gallbladder involvement of Henoch- Schonlein purpura mimicking acute acalculous cholecystitis. Digestion. 2004;70(1):45-48. [PubMed]
27. Dalens B, Travade P, Labbe A, Bezou MJ. Diagnostic and prognostic value of fibrin stabilising factor in Schonlein-Henoch syndrome. Arch Dis Childhood. 1983 Jan;58(1):12-14. [PubMed]
28. Henriksson P, Hedner U. Nilsson IM. Factor XIII (fibrin stabilising factor) in Henoch-Schonlein’s purpura. Acta Pediatr Scand. 1977 May;66(3):273-277. [PubMed]
29. Lee HJ, Kim SM, Yun SR, et al. A Case of Henoch-Schonlein Purpura with Psoas Muscle Abscess and Full-blown Gastrointestinal Complications. Korean J Gastroenterol. 2007 Feb;49(2):114-8. [PubMed]
30. Lippl F, Huber W, Werner M, Nekarda H, Berger H, Weigert N. Life-threatening gastrointestinal bleeding due to a jejunal lesion of Henoch-Schonlein purpura. Endoscopy. 2001 Sep;33(9):811-813. [PubMed]
31. Nishiyama R, Nakajima N, Ogihara A, Oota S, Kobayashi S, Yokoyama K, Oonishi M, Miyamoto S, Akai Y, Watanabe T, Uno A, Mizuno S, Ootani T, Tanaka N, Moriyama M. Endoscope images of Schonlein-Henoch purpura. Digestion. 2008;77(3-4):236-41. Epub 2008 Aug 7. [PubMed]
32. Chan JC, Li PK, Lai FM, Lai KN. Fatal adult Henoch-Schönlein purpura due to small intestinal infarction. J Intern Med. 1992 Aug; 232(2):181-4. [PubMed]
33. Tripodi A, Anstee QM, Sogaard KK, Primignani M, Valla DC. Hypercoagulability in cirrhosis: causes and consequences. J Thromb Haemost. 2011 Sept;9(9):1713-23. [PubMed]
© 2012 Dermatology Online Journal

