Multiple pilomatricomas: Case presentation and review of the literature
Published Web Location
https://doi.org/10.5070/D31td0145cMain Content
Multiple pilomatricomas: Case presentation and review of the literature
Joel Juin Li Chan1, Hong Liang Tey MBBS, FAMS2
Dermatology Online Journal 16 (4): 2
1. School of Medicine, University of Melbourne, Australia2. Associate Consultant Dermatologist, National Skin Centre, Singapore. teyhongliang111@yahoo.com
Abstract
Pilomatricomas are the most common superficial pediatric tumor and current literature demonstrates that they can be diagnosed clinically. Excisional biopsy is recommended for diagnosis and curative treatment. Because the recurrence rate after excision is low, pilomatrical carcinoma should be considered in the case of local recurrence. Multiple pilomatricomas, on the other hand, are rare. They have been associated with various syndromes, of which myotonic dystrophy has been the most described. We present a patient with multiple pilomatricomas in an otherwise well 13-year-old girl and our management of her in relation to current literature.
Introduction
First described by Malherbe and Chenantais in 1880 as a benign neoplasm of sebaceous gland origin, our understanding of pilomatricoma has evolved significantly [1]. It is now understood that pilomatricomas are calcifying epitheliomas, which may differentiate towards the hair matrix, hair cortex, follicular infundibulum, outer root sheath, and hair bulge [2]. Etiology has been linked to mutations such as β catenin and bcl-2 [3, 4]. Epidemiology shows bimodal peaks in presentation, with up to 60 percent of cases occurring in the first two decades and a secondary peak in the sixth decade [5, 6, 7]. Although pilomatricomas represent only 0.12 percent of all cutaneous tumors, it is relatively common in children and represents the most common benign superficial tumor in this age group [5, 6, 7]. Multiple pilomatricomas in a single patient, on the other hand, is rare. A strong association between multiple pilomatricomas and myotonic dystrophy exists [8]. Currently disagreement exists with regards to accurate preoperative diagnosis; reported rates range from 0.01 percent to 82 percent [5, 6, 9, 10]. Various studies and case reports have demonstrated misdiagnoses with respect to clinical diagnosis, fine needle aspiration, ultrasound, positron emission tomography (PET), and computed tomography (CT) [5, 11, 12]. We report a case of an otherwise well adolescent teenager with multiple pilomatricomas and review the literature to date.
Case report
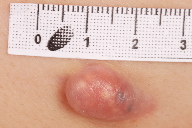
A 13-year-old girl presented to the National Skin Centre (NSC), Singapore with a 6-month history of a slowly growing mass on the right trunk, approximately at the mid axillary line. On examination, there was an asymptomatic, soft, erythematous nodule (14 x 10 mm) with obvious yellow-to-white central areas that were hard to palpation, suggesting calcium deposition. Pain was elicited on palpation and the tenting sign was positive. No systemic signs of illness, including lymphadenopathy or neurological signs, were present. The patient gave no history of recent trauma, radiation, infection, or surgery. The patient had presented before with similar lesions in April 2009 on the back and in April 2006 on the nape, both of which were excised and demonstrated classical features of pilomatricoma on histopathology.
Prior to presentation at NSC, she had had two pilomatricomas excised from her left arm and left leg in another hospital in 2004. No recurrences have occurred since. Other than a personal and family history of eczema, further history was unremarkable.
The patient and parents were keen for excision of the new nodule because the patient experienced pain when pressure was applied to the nodule. The patient is currently being followed.
Discussion and literature review
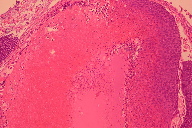
Pilomatricomas represent 70 percent of all adnexal tumors in the first two decades of life and most reports show a female preponderance [7, 9, 13]. Predilection sites for pilomatricoma presentation are the head and neck (>50%), with the upper limbs being the next most common site [6, 7, 9]. There is no current literature describing presentation on a non-hair bearing site, which makes sense because of the tumor’s differentiation towards the hair matrix. Features that can obscure the diagnosis on presentation include telangiectasia, hyperkeratosis, hemosiderin deposition, and erosion [5]. The history is often of an asymptomatic, slowly growing subcutaneous nodule with epidermal fixation and mobility over underlying structures [13]. ‘Tent’ Sign is produced by stretching the overlying skin to show the multifaceted components of calcification. The classical histology is said to be defined by the presence of ghost or shadow cells and basophilic cells; however, calcification is also common [5, 6]. Differential diagnoses to consider with pilomatricomas include sebaceous cyst, dermoid cyst, branchial cyst, preauricular cyst, ossifying hematoma, chondroma, lymphadenopathy, foreign body reaction, fibroxanthoma, giant cell tumor, and osteoma cutis.
Attempts have been made to diagnose pilomatricoma using imaging techniques. The classic CT radiological appearance has been described to be that of a non-contrast enhancing, sharply demarcated subcutaneous nodule [11]. Ultrasound has demonstrated accuracy rates between 28.9 and 46 percent and the following have been found to be possible discriminative ultrasonographic criteria from other subcutaneous tumors: heterogeneous echotexture, internal echogenic foci in scattered-dot pattern, and a hypoechoic rim or posterior shadowing [14]. Misdiagnoses with PET/CT have been reported [11].
Attempts have also been made to use fine needle aspiration (FNA) to diagnose pilomatricoma. Kumar and Verma found that the presence of basaloid cells, ghost cells, and foreign body giant cells are essential for a confident diagnosis of pilomatricoma. Additional supporting features included calcification, naked nuclei, and nucleated squamous cells. Differentials on FNA cytology were squamous cell carcinoma, giant cell lesions, and epidermal inclusion cysts [12]. There were, however, numerous reports of misdiagnosis using FNA [11, 12] and biopsy remains the definitive means of diagnosis.
Although our understanding of any possible transition of pilomatricoma to pilomatrical carcinoma is limited; the current consensus is to treat by surgical removal primarily for cosmesis and to prevent the possibility of malignant transformation (despite a lack of evidence in this aspect). There are few reports in the literature of malignant transformation and, of those tumors that have, most are in elderly patients. Hence, a greater degree of suspicion is required in this group of patients [5]. Comprehensive management of patients should involve physical examination for lymphadenopathy. Histological features suggestive of malignancy include focal necrosis, invasive nests with irregular/infiltrative borders, large vesicular nuclei, prominent nucleoli, and multiple mitotic figures. Recurrence rates after excision are low and pilomatrical carcinoma should be considered if lesions recur [5, 6, 9, 10].
The occurrence of multiple pilomatricomas has been associated with the development of myotonic dystrophy (MD), which can occur before or after the onset of lesions. Myotonic dystrophy (Steinert Disease) is described to be an autosomal dominant condition with variable phenotypic penetrance. Hence, immediate family may not always demonstrate a family history of the condition. Cigliano et al. reported 1 out of 2 of their patients with multiple pilomatricomas had MD whereas Julian et al. reported the occurrence of MD in 1 out of 4 of their patients [5, 8, 9]. Myotonic dystrophy usually presents during the teenage years or adulthood, but the onset of pilomatricomas has been extremely variable, ranging from many years before to many years after the onset of symptomatic MD [8]. There have also been various reports of other syndromes being associated with multiple pilomatricomas, including Turner syndrome, Gardner syndrome, and sarcoidosis [15].
Of interest to dermatologists are the low rates of preoperative diagnosis, demonstrated to be as low as 0.01 percent by Lan et al. [5, 6]. In the study by Julian et al., whereas only 21 percent of cases had a correct preoperative diagnosis, 88.89 percent of these correct diagnoses were made by dermatologists [5]. Given the predilection sites for head and neck, and the higher incidence in children and adolescents, the majority of presentations are to otolaryngologists or pediatricians. The input of dermatologists into such cases may be of significant benefit.
A study in 2008 by Zaballos et al. attempted to identify means of improving clinical diagnosis by dermatoscopy. The common findings on dermatoscopic examination of pilomatricomas included irregular white structures and the presence of polymorphous/atypical vessels (i.e., reddish homogenous areas, hairpin vessels, linear-irregular vessels, and dotted vessels). Coupled with the absence of other specific dermatoscopic criteria for other skin tumors, this could be used to improve clinical diagnosis [16].
In conclusion, pilomatricoma can be diagnosed clinically and excisional biopsy is recommended for diagnosis and curative treatment. Because the recurrence rate after excision is low, pilomatrical carcinoma should be considered in the case of local recurrence. Multiple pilomatricomas have been associated with various syndromes of which myotonic dystrophy has been the most described. Long term follow up is recommended for these patients.
References
1. Malherbe A, Chenantais J. Note sur l’epithelioma calcifie des glandes sebaces. Progres Medical. 1880;8:826-8.2. Kurokawa I, Yamanaka K-i, Senba Y, Sugisaki H, Tsubura A, Kimura T, et al. Pilomatricoma can differentiate not only towards hair matrix and hair cortex, but also follicular infundibulum, outer root sheath and hair bulge. Experimental dermatology. Exp Dermatol. 2009;18(8):734-7. [PubMed]
3. Hassanein A, Glanz S. β-catenin expression in benign and malignant pilomatrix neoplasms. Br J Dermatol 2004;150(3):511-6. [PubMed]
4. Farrier S, Morgan M. bcl-2 Expression in pilomatricoma. Am J Dermatopathol 1997;19(3):254-7. [PubMed]
5. Julian CG, Bowers PW. A clinical review of 209 pilomatricomas. J Am Acad Dermatol 1998;39(2 Part 1):191-5. [PubMed]
6. Lan MY, Lan MC, Ho CY, Li WY, Lin CZ. Pilomatricoma of the head and neck: a retrospective review of 179 cases. Arch Otolaryngol Head Neck Surg 2003;129(12):1327-30. [PubMed]
7. Moehlenbeck FW. Pilomatrixoma (calcifying epithelioma): a statistical study. Arch Dermatol 1973;108(4):532-4. [PubMed]
8. Geh JL, Moss AL. Multiple pilomatrixomata and myotonic dystrophy: a familial association. Br J Plast Surg 1999;52(2):143-5. [PubMed]
9. Cigliano B, Baltogiannis N, De Marco M, Faviou E, Settimi A, Tilemis S, et al. Pilomatricoma in childhood: a retrospective study from three European paediatric centres. Eur J Pediatr 2005;164(11):673-7. [PubMed]
10. Pirouzmanesh A, Reinisch JF, Gonzalez-Gomez I, Smith EM, Meara JG. Pilomatrixoma: a review of 346 cases. Plast Reconstr Surg 2003;112(7):1784-9. [PubMed]
11. Jung Y-S, Kang J-G, Park W-S, Ryu J. Pilomatricoma: Diagnostic pitfalls in PET/CT and fine-needle aspiration biopsy. Otolaryngol Head Neck Surg 2007;37(5):845-6. [PubMed]
12. Kumar N, Verma K. Fine needle aspiration (FNA) cytology of pilomatrixoma. Cytopathology. 1996;7(2):125-31. [PubMed]
13. Golpour M. Evaluation of characteristics of patients with pilomatricoma in Mazandaran province, 1996-2006. Pak J Biol Sci 2009;12(6):548-50. [PubMed]
14. Choo HJ, Lee SW, Lee YH, Lee JH, Oh M, Kim MH, et al. Pilomatricomas: the diagnostic value of ultrasound. Skeletal Radiology. 2009. [PubMed]
15. Cecen E, Ozguven AA, Uysal KM, Gunes D, Ozer E, Olgun N, et al. Pilomatricoma in children: a frequently misdiagnosed superficial tumor. Pediatr Hematol Oncol 2008;25(6):522-7. [PubMed]
© 2010 Dermatology Online Journal