Primary cutaneous CD30+ large-cell lymphoma presenting as a scrotal plaque
Published Web Location
https://doi.org/10.5070/D31fm128cqMain Content
Primary cutaneous CD30+ large-cell lymphoma presenting as a scrotal plaque
Sapna Modi MD1, Deborah J Yang MD2, Norman Sule MD3, Ted Rosen MD2
Dermatology Online Journal 15 (12): 9
1. Baylor College of Medicine, Houston, Texas2. Department of Dermatology, Baylor College of Medicine and Michael E DeBakey Veterans Affairs Medical Center, Houston, Texas. vampireted@aol.com
3. Department of Pathology, Baylor College of Medicine, Houston, Texas
Abstract
Primary cutaneous CD30+ large-cell lymphoma (CD30+ LCL) is a rare subtype of cutaneous T cell lymphoma. We report a patient with a one-year history of an enlarging scrotal plaque. Skin biopsy stained positive for CD30, and after thorough systemic evaluation, a diagnosis of primary cutaneous CD30+ LCL was made. Most commonly CD30+ LCL presents on the extremities and trunk. We present the first case to our knowledge of a patient initially presenting with a solitary scrotal nodule. When primary cutaneous CD30+ LCL is suspected, it is important to conduct a thorough systemic evaluation for systemic LCL and immunohistological studies for CD30- types. Ruling these conditions out will prevent unnecessarily aggressive treatment for primary cutaneous CD30+ LCL, an indolent disease with a favorable prognosis.
Introduction
Primary cutaneous CD30+ LCL is a rare type of cutaneous T cell lymphoma (CTCL) that tends to regress spontaneously. While this neoplasm presents in many ways, it most commonly appears as unifocal or multifocal nodules, occasionally with ulceration, on the trunk and extremities [1, 2, 3, 4]. In 2003, Lan et al. reported an unusual case of primary cutaneous CD30+ LCL presenting initially as ulcerative papules on the hands followed by an ulcerating nodule on the scrotum [3]. We report the first case of a patient initially presenting with a solitary scrotal nodule.
Case report
 |  |
| Figure 1 | Figure 2 |
|---|---|
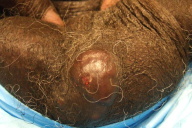
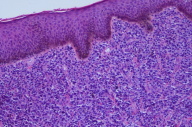
| Figure 1. Three centimeter nodule on left side of scrotum Figure 2. Extensive infiltration of the dermis by large anaplastic lymphocytic cells (H&E, x10) | |
 |
| Figure 3 |
|---|
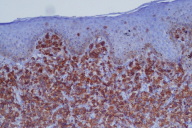
| Figure 3. Extensive infiltration of the dermis by large anaplastic lymphocytic cells with positive CD30 staining (CD30 stain, x10) |
A 63-year-old African American male with a past medical history of hypertension, coronary artery disease, and hyperlipidemia presented with a one-year history of a growing lesion on his scrotum. The patient denied pain, pruritus, or other associated symptoms. He also denied weight loss.
On physical examination, a 3 cm reddish-purple nodule was noted on the left side of the scrotum (Fig. 1). Biopsy of the scrotal lesion revealed extensive infiltration of the dermis by large anaplastic lymphocytic cells (Fig. 2) that diffusely expressed CD30 (Fig. 3) in addition to exhibiting bcl-2 positivity. The specimen was negative for CD20, CD3, CD5, CD10, CD45RO, bcl-6, and ALK antibodies. Computed tomography scans did not demonstrate systemic disease. A diagnosis of primary cutaneous CD30+ large-cell lymphoma was made, and the patient was then referred to oncology.
Discussion
The cutaneous T cell lymphomas (CTCL) include a diverse number of malignancies: mycosis fungoides (MF), Sézary syndrome, lymphomatoid papulosis (LyP), and anaplastic large-cell lymphoma (LCL) as well as several lesser-known entities. Clinical presentation, histology, and immunophenotype must be considered together in order to establish the specific subtype and ensure appropriate treatment and management [2, 4]. Recently recognized as a distinct condition, primary cutaneous CD30+ LCL accounts for approximately 10 percent of all CTCL cases [2, 5]. Diagnosis is based on the following criteria: (a) predominance (greater than 75%) of large clusters of CD30+ blast cells in the initial skin biopsy; (b) no clinical evidence of LyP; (c) no prior or concurrent LyP, MF, or other cutaneous lymphoma; and (d) no extracutaneous localization at presentation [4, 6].
Primary cutaneous CD30+ LCL is a rare malignancy arising de novo in the skin and most commonly affects men between the ages of 45 and 60. Clinically, this condition tends to appear as a solitary nodule or tumor, often with ulceration, although 20 percent are multifocal at presentation. These lesions tend to be firm and rapidly growing and favor the trunk and extremities [1, 2]. A study of 47 patients with primary cutaneous CD30+ LCL by Beljaards et al. showed that all 47 patients had lesions on the trunk, face, arms, or legs [4]. No patients had lesions located in the genital region. Recently Lan et al. described a patient who initially presented with spontaneously regressing papules on both hands, followed later by a scrotal nodule [3]. Our patient is the first case reported who initially presented with a solitary scrotal nodule without involvement of the more common locations.
Biopsy typically reveals clusters of infiltrating large CD30+ cells. Sparse inflammatory cells are often seen at the periphery of these tumor-cell clusters. Multinucleated cells, such as Reed-Sternberg cells, may also been seen in the biopsy specimen [1, 3].
Of great importance in the diagnosis of primary cutaneous LCL is determining CD30 positivity. Previous studies have shown that CD30 negative LCLs have a worse prognosis than CD30+ LCLs [4, 8] and should be treated more aggressively. A European multicenter study of 47 patients, however, showed that morphologic subtype of LCL (i.e., anaplastic vs. non-anaplastic types, including immunoblastic and large pleomorphic) has no bearing on prognosis. Based on this study, the authors recommended that "anaplastic" be dropped from the nomenclature [1, 4].
CD30+ lesions exist on a continuum from malignant CD30+ LCL to benign LyP. The tendency of primary cutaneous CD30+ LCL to regress spontaneously about 25 percent of the time suggests that this condition may be related to LyP, an indolent type of CTCL characterized by recurrent crops of pruritic papulonodular lesions that usually spontaneously regress. Histopathology of LyP reveals numerous inflammatory cells with few (less than 25%) CD30+ cells. This picture may be indistinguishable from some primary cutaneous CD30+ LCLs, making the clinical presentation critical in the final diagnosis [2, 3].
Primary cutaneous CD30+ LCL is also histologically similar to lymphonodal CD30+ anaplastic LCL. A diagnosis of systemic LCL is made if there is extracutaneous involvement or lymph node involvement beyond one regional lymph node. Systemic LCL shows a bimodal age distribution with a predominance in younger patients and is associated with the Epstein-Barr virus. Approximately 50 percent of cases show the t(2;5) translocation (ALK-positive), which aids in diagnosis [3, 7]. Unlike primary cutaneous CD30+ LCL, which may remain localized in the skin for prolonged periods of time, this systemic counterpart is an aggressive disease with rapid spread to other organs, including the skin. Given the worse prognosis of this latter condition, it is imperative that metastatic systemic LCL be excluded as a cause of a cutaneous nodule with CD30+ cells. Systemic work-up will also assist in differentiating primary cutaneous CD30+ LCLs from those occurring secondarily in patients with existing MF or other CTCL subtypes [4]. Secondary cutaneous CD30+ LCL has a worse prognosis, and therefore must not be overlooked in the evaluation of CD30+ LCLs [3].
Various therapeutic options may exist for primary cutaneous CD30+ LCLs depending on the extent of disease. Local radiotherapy and surgical excision are effective with limited disease, while systemic chemotherapy should be reserved for patients with generalized disease or progression to systemic LCL [1, 2]. Patients with solitary or localized lesions may require no treatment as spontaneous regression may occur in up to 42 percent of CD30+ LCLs. We referred our patient to an oncologist who will treat his solitary lesion with local radiotherapy.
This case documents an unusual presentation of primary cutaneous CD30+ LCL as a growing scrotal lesion. The cases of Lan et al. and our own demonstrate that the scrotum, although unusual, can be a site of primary cutaneous CD30+ LCL [1]. When primary cutaneous CD30+ LCL is suspected, it is important to conduct a thorough systemic evaluation for systemic LCL and immunohistological studies for CD30- types. Ruling out these conditions will prevent unnecessarily aggressive treatment for primary cutaneous CD30+ LCL, an indolent disease with a favorable prognosis.
References
1. Freitas IC, Vieira R, Afonso A, Pacheco A, Nunes P. Primary cutaneous CD30-positive large cell lymphoma. J Eur Acad Dermatol Venereol. 2000;14; 51-53. [PubMed]2. Parlette EC, Tabor CA. CD30+, large T-cell lymphoma: diagnostic distinction and management. Dermatol Online J. 2006;12; 8. [PubMed]
3. Lan CC, Yu HS, Cheng ST, Chen GS. Relapsing ulcerative papules over bilateral hands and scrotum in an Asian man: an atypical manifestation of primary cutaneous CD30-positive lymphoma. J Dermatol. 2003;30; 230-235. [PubMed]
4. Beljaards RC, Kaudewitz P, Berti E, Gianotti R, Neumann C, Rosso R, et al. Primary cutaneous CD30-positive large cell lymphoma: definition of a new type of cutaneous lymphoma with a favorable prognosis. A European Multicenter Study of 47 patients. Cancer. 1993;71; 2097-2104. [PubMed]
5. Fung MA, Murphy MJ, Hoss DM, Grant-Kels JM. Practical evaluation and management of cutaneous lymphoma. J Am Acad Dermatol. 2002;46; 325-357; quiz, 358-360. [PubMed]
6. Willemze R, Jaffe ES, Burg G, Cerroni L, Berti E, Swerdlow SH, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105; 3768-3785. [PubMed]
7. Bekkenk MW, Geelen FA, van Voorst Vader PC, Heule F, Geerts ML, van Vloten WA, et al. Primary and secondary cutaneous CD30(+) lymphoproliferative disorders: a report from the Dutch Cutaneous Lymphoma Group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95; 3653-3661. [PubMed]
8. Beljaards RC, Meijer CJ, Scheffer E, Toonstra J, van Vloten WA, van der Putte SC, et al. Prognostic significance of CD30 (Ki-1/Ber-H2) expression in primary cutaneous large-cell lymphomas of T-cell origin. A clinicopathologic and immunohistochemical study in 20 patients. Am J Pathol. 1989;135; 1169-1178. [PubMed]
© 2009 Dermatology Online Journal

