Follicular acneiform eruption induced by Bevacizumab
Published Web Location
https://doi.org/10.5070/D31683r8wtMain Content
Follicular acneiform eruption induced by bevacizumab
Maria Gavrilova MD, Jose M Martin MD, Alejandro Martin-Gorgojo MD, Esperanza Jorda-Cuevas MD PhD
Dermatology Online Journal 18 (9): 15
Department of Dermatology, Clinical University Hospital of Valencia, Valencia, SpainAbstract
Bevacizumab is a recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF). This antibody, in combination with other antineoplastic agents, is currently used to treat various neoplasms, including colorectal, lung, breast, kidney cancer, and glioblastoma. It is also being used as an off-label intravitreal agent in the treatment of proliferative (neovascular) eye diseases. We report the development of a skin rash with two different patterns in a patient with a hemangiopericytoma of the meninges, a rare aggressive sarcoma, who was treated with repeated intravenous injections of bevacizumab.
Bevacizumab is a recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF). This antibody, in combination with other antineoplastic agents, is currently used to treat various neoplasms, including colorectal, lung, breast, kidney cancer, and glioblastoma. It is also being used as an off-label intravitreal agent in the treatment of proliferative (neovascular) eye diseases.
We report the development of a skin rash with two different patterns in a patient with a hemangiopericytoma of the meninges, a rare aggressive sarcoma, who was treated with repeated intravenous injections of bevacizumab.
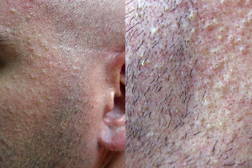 | 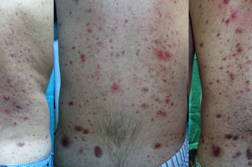 |
| Figure 1 | Figure 2 |
|---|
A 37-year-old man, with the diagnosis of a meningeal hemangiopericytoma that was treated 10 years before with surgical resection and radiation therapy, presented with bone, liver, and lung metastases. Chemotherapy cycles with oral temozolomide 150 mg/m² on days 1-7 and days 15-21 and intravenous bevacizumab 5 mg/kg on days 8 and 22, repeated at 28-day intervals, were started. After two months of this treatment, the patient developed violaceous nodules and papules on the lumbar skin. In one week, the lesions transformed into an eruption, which affected all the skin of the back and consisted of papular and papular-pustular lesions (Figure 1). At the same time, numerous monomorphic translucent papules with a follicular distribution on the face could also be seen (Figure 2). A biopsy specimen of the lesions on the back revealed a lymphocytic superficial infiltrate without vascular proliferation. The treatment with topical (erythromycin) and systemic antibiotics (oral doxycycline 100 mg daily) produced only slight improvement.
Several cutaneous adverse effects have been reported with bevacizumab therapy: mucosal bleeding, stomatitis, disturbed wound healing [1], papuloerythematous rash, and cutaneous thrombosis. Bevacizumab-associated acneiform eruption was reported in the treatment of colon cancer and glioblastoma (systemic administration)[2, 3] and macular degeneration (intravitreal administration [4, 5]. The eruption in these patients was similar to that characterized in patients treated with epidermal growth factor receptor (EGFR) inhibitors and was also dose dependent. However, in comparison to these drugs, bevacizumab usually causes less frequent and less severe cutaneous side effects. VEGF plays a role not only in tumor neovascularization. It has been shown that VEGF is a growth factor for follicular cells, keratinocytes, and endothelial cells [6]. This may explain the variety of follicular-pustular eruptions, similar to the EGFR inhibitor-associated eruptions. EGFR inhibitor-induced cutaneous side effects are associated with an effective antineoplastic activity of the drug. Such correlation has not yet been confirmed for bevacizumab. Although there have been reports relating rash development and a positive drug response [7], further research is needed to support this observation.
In conclusion, advances in oncological therapy have been accompanied by a new spectrum of cutaneous side effects. Dermatologists should be aware of the cutaneous reactions of these drugs and evaluate correctly the skin rash in these patients. We present an extensive acneiform reaction that developed in a patient with a meningeal hemangiopericytoma treated with bevacizumab. Taking into consideration a lack of reports of the cutaneous side effects after administration of bevacizumab, as well as others anti-angiogenic drugs, further investigations into associated eruptions and management are needed.
References
1. Scappaticci FA, Fehrenbacher L, Cartwright T, et al. Surgical wound healing complications in metastatic colorectal cancer patients treated with bevacizumab. J Surg Oncol. 2005;91:173-180. [PubMed]2. Gotlib V, Khaled S, Lapko I, et al. Skin rash secondary to bevacizumab in a patient with advanced colorectal cancer and relation to response. Anticancer Drugs. 2006;17:1227-1229 [PubMed]
3. Keenan BP, Abuav R. Acneiform eruption in a patient receiving bevacizumab for glioblastoma multiforme. Arch Dermatol. 2010;146:577. [PubMed]
4. Amselem L, Diaz-Llopis M, Garcia-Delpech S, et al. Papulopustular eruption after intravitreal bevacizumab (Avastin). Acta Ophthalmol. 2009;87:110-111. [PubMed]
5. Ladas ID, Moschos MM, Papakostas TD, et al. Skin rash associated with intravitreal bevacizumab in a patient with macular choroidal neovascularization. Clin Ophthalmol. 2009;3:129-31. [PubMed]
6. Man XY, Yang XH, Cai SQ, et al. Expression and localization of vascular endothelial growth factor and vascular endothelial growth factor receptor-2 in human epidermal appendages: a comparison study by immunofluorescence. Clin Exp Dermatol. 2009;34:396-401. [PubMed]
7. Saif MW, Longo WL, Israel G. Correlation between rash and a positive drug response associated with bevacizumab in a patient with advanced colorectal cancer. Clin Colorectal Cancer. 2008;7:144-148. [PubMed]
© 2012 Dermatology Online Journal

