Generalized B-cell non Hodgkin's lymphoma in association with leukocytoclastic vasculitis and disseminated intravascular coagulation
Published Web Location
https://doi.org/10.5070/D30pc7q9drMain Content
Generalized B-cell non Hodgkin lymphoma in association with leukocytoclastic vasculitis and disseminated intravascular coagulation
Carla Cardinali MD1, Simone Santini MD2, Angelo di Leo MD2, Augusto Giannini MD3, and Lo Scocco Giovanni MD1
Dermatology Online Journal 12 (3): 15
1. U.O. Dermatologia, USL 4, Piazza Ospedale, 5- 59100 Prato Italia. cardinalic@yahoo.it2. U.O. Oncologia Medica, USL 4, Piazza Ospedale, 5- 59100 Prato Italia
3. U.O. Anatomia patologica, USL 4, Piazza Ospedale, 5- 59100 Prato Italia
Abstract
A 66-year-old woman was referred a 4-month history of asthenia, weight loss, productive cough, increasing dyspnea, epigastric pain, and night sweats. A B-cell non-Hodgkin lymphoma was discovered in association with a severe leukocytoclastic vasculitis and disseminated intravascular coagulation. The patient was treated successfully with the combination of cyclophosphamide, vincristine, prednisone, and rituximab.
Leukocytoclastic vasculitis (LCV) is an immune-complex related vascular disorder that combines segmental inflammation with necrosis of the blood vessels. It may occur in association with an underlying chronic disease, may be precipitated by infections or drugs, or may develop for unknown reasons. It can be very rarely associated with lymphoproliferative disorders and solid tumors. Lymphoproliferative disorders associated with LCV include Hodgkin disease, lymphosarcoma, adult T-cell leukemia, mycosis fungoides, and myelofibrosis.
Clinical synopsis
A 66-year-old woman was referred with a 4-month history of asthenia, considerable weight loss, productive cough, and increasing dyspnea, epigastric pain, and night sweats. For such symptoms she was admitted in 2004 to a medicine unit where the presence of a small, B-cell non-Hodgkin lymphoma (NHL) was discovered on the basis of a laterocervical lymph-node biopsy; widespread pulmonary intersticial involvement was diagnosed radiologically as well. Immunostaining with various monoclonal antibodies revealed the small cells to be CD20+, CD79a+, CD5+, Ki67+ : 5 percent; CD3-; CD10-; Bcl6-, cyclin D1-. Enlarged lymph nodes were detectable with computerized tomography scan of the neck, chest, and abdomen in association with bilateral pleural effusion and splenomegaly.
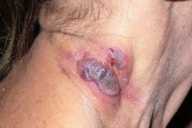
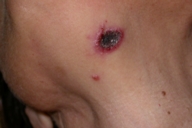
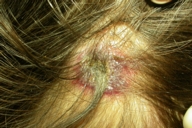
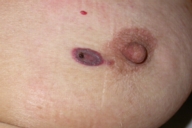
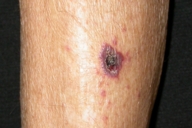
The patient became febrile and was transferred to an oncology unit. During the second hospitalization, muscle atrophy and oral mucositis were evident as well. During this period, the patient also developed papulo-nodular, hemorrhagic-necrotizing lesions with ulcerative evolution on laterocervical, nuchal, submental, thoracic regions as well as on extremities (Figs. 1-5).
 |  |
| Figure 1 | Figure 2 |
|---|---|
| Figure 1. Papulo-nodular and hemorrhagic-necrotizing lesions with ulcerative evolution on the right laterocervical area | |
Figure 2. Ulcerative-necrotizing lesion on the submental region | |
 |  |
| Figure 3 | Figure 4 |
|---|---|
| Figure 3. Scaling and crusted lesion in the nuchal region | |
Figure 4. Papulonodular lesion with purpuric effusion and central depression on the breast | |
 |
| Figure 5 |
|---|
| Figure 5. Hemorrhagic-necrotizing lesion on the extensor surface of the left leg |
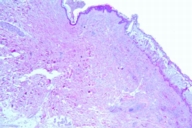
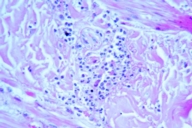
Cultures for fungi and bacteria from the ulcerative skin areas were negative. Histopathological examination of a skin biopsy, taken from a nodulo-ulcerative lesion, revealed intraepidermal blister formation and a neutrophilic, angiocentric, segmental inflammation with endothelial cell injury and fibrinoid necrosis of the blood vessel walls in the dermis (Figs. 6, 7). Neither immunoglobulins nor complement components were found in the perivascular areas, by direct immunofluorescence, on lesional skin as well as on perilesional areas.
Oral candidiasis from Candida albicans and Candida glabrata was found. Sero-haematological investigation revealed an elevated erythrocyte sedimentation rate, lymphopenia with increased and activated T-cell population, inversion of TCD4+/TCD8+ ratio with a strong reduction of TCD4+ lymphocytes and depletion of B-lineage lymphocytes.
A high titer of serum CA-19.9 antigen was found and the levels of serum α-fetoprotein, carcino-embryonic antigen and CA-15.3 antigen were normal. Increased levels of serum angiotensin-converting enzyme (ACE) were also detected. Calcemia and calciuria were within normal limits.
Screening for rheumatoid factor, antinuclear antibody, anti-extractable nuclear antigens, anti-neutrophil cytoplasmic and antiphospholipid antibodies were negative. Cryoglobulins were not detected. Complement component C3 and C4 and circulating immunocomplexes serum levels were normal. Serological screening for cytomegalovirus, Epstein-Barr virus, herpes simplex virus, varicella zoster virus, coxsackie virus, echovirus, adenovirus, Coxiella burneti, Mycoplasma pneumoniae was negative. Hepatitis B, hepatitis C, and HIV markers were negative. Disseminated intravascular coagulation (DIC) was recognized on the basis of coagulation factor reduction and activation of the fibrinolytic system with low fibrinogen (177, normal range: 200-400 mg/dL) and increased D-dimer levels (1000, normal range: 0-270 ng/mL).
Mild pulmonary hypertension was noted on the echocardiogram. Standard chest x-ray showed a widespread pulmonary interstitial involvement that was possibly the result of a lymphomatous lymphangitis. Bone marrow biopsy did not reveal neoplastic infiltration.
On the basis of the clinical and histopathological features, the diagnosis of leukocytoclastic vasculitis in association with generalized B-cell NHL was made.
In the following days, the status of our patient rapidly worsened with the development of respiratory failure (Eastern Cooperative Oncology Group performance status of 2 at admission to 3-4 during hospitalization). Serological tests for disseminated bacterial, mycotic, and viral infections were negative. Based on histological finding of NHL and positive immunohistochemistry for CD20 on cell surfaces, after informed consent, the patient underwent treatment with rituximab at standard dose of 375 mg/m² /week for 4-weekly courses; she could not be treated with aggressive chemotherapy for her immunodeficiency because of low hemoglobin levels and low platelet count.
The patient quickly recovered from respiratory failure a few days after the first course with rituximab. The platelet count, hemoglobin levels, and D-dimer levels also rapidly improved, so she underwent combined treatment with rituximab and chemotherapy (R-CVP: cyclophosphamide 750mg/m² IV on day 1; vincristine 1.4 mg/m² iv on day 1; prednisone 100 mg po days 1-5, rituximab 375mg/m² on day 1, repeated every 3 weeks).
Oral candidiasis was treated with nystatin suspension and subsequently with fluconazole and caspofungin. The cutaneous lesions were managed by local antibiotics and resolved with hyperpigmented, atrophic scars.
Conclusions
LCV is an inflammatory necrotizing condition of dermal vessels, presenting with variable clinical symptoms. In most cases it becomes manifest as palpable purpura and hemorrhagic-necrotizing lesions. Bullous, nodular, and urticarial presentations are also seen, and sometimes livedo reticularis-like and pyoderma gangrenosum-like manifestations occur. Common etiological factors include bacterial and viral antigens, a variety of collagen vascular diseases (systemic lupus erythematous, Sjogren syndrome, rheumatoid arthritis, Behcet disease) hyperglobulinemic states, cryoglobulinemia, bowel bypass syndrome, ulcerative colitis, cystic fibrosis, and primary biliary cirrhosis [1, 2, 3, 4, 5, 6, 7]. The relationship between DIC and cutaneous vasculitis has been also suggested [8].
Despite extensive investigation, no association with viral infections, connective tissue disorders or other chronic diseases was found in our patient in whom the appearance of LCV followed the development of a generalized B-cell NHL lymphoma. The association between vasculitis and lymphoproliferative diseases is relatively uncommon; LCV is reported in association with Hodgkin disease, lymphosarcoma, adult T-cell leukemia, mycosis fungoides, myelofibrosis, and acute and chronic myelogenous leukemia [7, 9, 10, 11].
Urticarial vasculitis and Churg-Strauss cutaneous extravascular necrotizing granuloma have been reported in the literature as paraneoplastic presentations of B-cell NHL lymphoma [12, 13] but no report exists about the concomitant necrotizing cutaneous vasculitis and this tumor.
Even if the exact pathogenesis of the association with lymphoma is unclear, it is possible that immune defects, occurring in case of malignancy, give rise to complement fixation in vessel walls with subsequent development of vasculitis.
The presence of lymphoma was probably responsible for the initiation of coagulation cascade too, as suggested by the existence of DIC. In fact, hemostatic abnormalities and thrombotic disorders are often found in patients with malignant lymphoma. One of the mechanisms of DIC, in this case, may involve elevated cytokine production by lymphoma cells, which can stimulate the expression of leukocyte tissue factor in blood cells [14].
In addition to the existence of DIC, our patient presented splenomegaly, increased serum ACE levels, mediastinal lymphoadenopathy, and pulmonary infiltrates, but none of diagnostic tests evidenced sarcoidosis. Sarcoidosis-lymphoma syndrome is a well known entity so that, in case of lymphoma, a possible sarcoidosis should be considered in case of elevated ACE levels, hypercalciuria, and bilateral hilar adenopathy on chest x-ray and chest CT [15, 16].
Interestingly, intraepidermal blister formation was found on histopathological examination of lesional skin. Blistering may occur as a secondary event associated with skin viral infections such as herpes simplex and varicella zoster [17, 18]. In our patient, however, these infections were excluded and skin blistering seemed to be related to tissue injury and fluid accumulation within intraepidermal layers. Moreover, no acquired autoimmune blistering disease could be demonstrated to explain the histopathological findings, as also suggested by negative direct immunofluorescence.
Also of interest was the remission of cutaneous manifestations after the combination of cyclophosphamide, vincristine, prednisone, and rituximab. Rituximab, the chimeric anti-CD20 antibody, is widely used in the clinical treatment of patients with NHL. This drug sensitizes NHL B-cell lines to drug-induced apoptosis via down-regulation of Bcl-x(L) expression and its binding to the B-cell surface antigen CD20 [19].
References
1. Witzens M, Moehler T, Neben K, Fruehauf S, Hartschuh W, Ho AD, Goldschmidt H. Development of leukocytoclastic vasculitis in a patient with multiple myeloma during treatment with thalidomide. Ann Hematol. 2004 Jul; 83 (7): 467-70. PubMed2. Bayer-Garner IB, Smoller BR. Leukocytoclastic (small vessel) vasculitis in multiple myeloma. Clin Exp Dermatology. 2003 Sep; 28 (5):521-24. PubMed
3. Marini A, Fenk R, Plettnberg H, Ruzicka T, Haas R, Hengge UR. Rare types of vasculitis as markers of plasmocytoma. Hautarzt. 2005 Jan; 19. PubMed
4. Ghinoi A, Mascia MT, Puccini R, Ferri C. Autoimmune and lymphoproliferative HCV-correlated manifestations: example of mixed cryoglobulinemia. G Ital Nefrol. 2004 May-Jun; 21(3): 225-37. PubMed
5. Schwartzenberg S, Levo Y, Averbuch M. Generalized vasculitis, thrombocytopenia, and transient lymphoproliferative disorder caused by idiopathic mixed cryoglobulinemia. Am J Med Sci. 2003 Jul; 326(1): 47-50. PubMed
6. Navarro JF, Quereda C, Rivera M, Navarro FJ, Ortuno J. Anti-neutrophil cytoplasmic antibody-associated paraneoplastic vasculitis. Postgrad Med J. 1994 May; 70(823): 373-375. PubMed
7. Lotti TM, Comacchi C, Ghersetich I. Cutaneous necrotizing vasculitis. Relation to systemic disease. Adv Exp Med Biol. 1999; 455: 115-125. PubMed
8. Kamouchi M, Yoshinari M, Goto H, Ishitsuka T, Murai K, Tashiro K, Fujishima M. Disseminated intravascular coagulation in a patient with progressive systemic sclerosis associated with necrotizing angiitis and generalized lymphoadenopathy. Acta Haematol. 1991; 86 (4): 203-205. PubMed
9. Wooten MD, Jasin HE. Vasculitis and lymphoproliferative diseases. Semin Arthritis Rheum. 1996 Oct; 26(2): 564-74. PubMed
10. Levy-Clarke GA, Buggage RR, Shen D, Vaughn LO, Chan CC, Davis JL. Human T-cell lymphotropic virus type-1 associated t-cell leukemia/lymphoma masquerading as necrotizing retinal vasculitis. Ophthalmology. 2002 Sep; 109(9): 1717-22. PubMed
11. Granstein RD, Soter NA, Haynes HA. Necrotizing vasculitis within cutaneous lesions of mycosis fungoides. J Am Acad Dermatol. 1983 Jul; 9(1): 128-33. PubMed
12. Wilson D, McCluggage WG, Wright GD. Urticarial vasculitis: a paraneoplastic presentation of B-cell non-Hodkin's lymphoma. Rheumatology. 2002 Apr; 41(4): 476-77. PubMed
13. Calonje JE, Greaves MW. Cutaneous extravascular necrotizing granuloma (Churg-Strauss) as a paraneoplastic manifestation of non-Hodgkin's B-cell lymphoma. J R Soc Med. 1993 Sep; 86(9): 549-50. PubMed
14. Sase T, Wada H, Yamaguchi M, Ogawa S, Kamikura Y, Nishikawa M, Kaneko T, Abe Y, Nishioka J, Nobori T, Shiku H. Haemostatic abnormalities and thrombotic disorders in malignant lymphoma. Thromb Haemost. 2005 Jan; 93(1): 153-59. PubMed
15. Regdosz R, Mulliez P, Saout J, Creusy C, Crinquette J. Elevation of serum angiotensin converting enzyme levels in Hodgkin's lymphoma. Presse Med. 1985 Oct 5; 14(33): 1757. PubMed
16. Fuzibet JG, Cassuto JP, Gratecos N, Iordache A, Viot G, Dujardin P. Malignant lymphoma and sarcoidosis. Changes in the serum levels of angiotensin-converting enzyme. Presse Med. 1984 Oct 20; 13(37): 2270. PubMed
17. Diaz LA, Giudice GJ. End of the century overview of skin blisters. Arch Dermatol. 2000 Jan; 136(1): 106-12. PubMed
18. Cohen P, Guillevin L. Vasculitis associated with viral infections. Presse Med. 2004 Nov 6; 33(19 Pt 2):1371-84. PubMed
19. Jazirehi AR, Huerta-Yepez S, Cheng G, Bonavida B. Rituximab (chimeric anti-CD20 monoclonal antibody) inhibits the constitutive nuclear factor-{kappa}B signaling pathway in non-Hodgkin's lymphoma B-cell lines: role in sensitization to chemotherapeutic drug-induced apoptosis. Cancer Res. 2005 Jan 1; 65(1): 264-276. PubMed
© 2006 Dermatology Online Journal