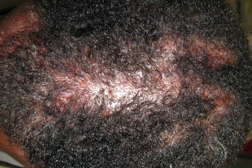
Sarcoidosis-induced alopecia
Published Web Location
https://doi.org/10.5070/D30fp92370Main Content
Sarcoidosis-induced alopecia
Nancy S House1 MD, John P Welsh2 MD, Joseph C English III3 MD
Dermatology Online Journal 18 (8): 4
1. Valley Dermatopathology and Department of Dermatology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania2. Division of Dermatology, Temple University School of Medicine, Philadelphia, Pennsylvania
3. Department of Dermatology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania
Abstract
Cutaneous sarcoidosis of the scalp may induce scarring alopecia, which clinically resembles other forms of primary cicatricial alopecia. Differentiation via histologic evaluation is necessary because sarcoidosis demonstrates classical non-caseating granulomas. Review of the literature reveals that sarcoidosis-induced alopecia occurs more commonly in black females age 23 to 78, with the majority of patients having coexisting facial sarcoidosis with pulmonary and lymph node involvement. Given the strong association between sarcoidal alopecia and systemic sarcoidosis, evaluation of the patient is indicated if alopecia is the initial presenting manifestation.
Introduction
The clinical differential diagnosis to be considered for primary cicatricial alopecia includes entities such as lichen planopilaris, discoid lupus erythematosus, central centrifugal cicatricial alopecia, Brocq alopecia, folliculitis decalvans, dissecting cellulitis of the scalp, and acne keloidalis. Disease entities that can cause secondary cicatricial alopecia include: immunobullous disorders, radiation, epidermal nevi, alopecia neoplastica, and sarcoidosis. We herein report two cases of alopecia clinically resembling lichen planopilaris and folliculitis decalvans but which demonstrated sarcoidal granulomas in patients with known systemic sarcoidosis. In addition, the literature concerning sarcoidosis-induced alopecia is reviewed.
Case reports
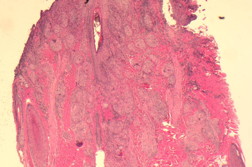
 |
| Figure 1 |
|---|
| Figure 1. Transversely-embedded scalp biopsy shows decreased follicular density, fibrosis, and sarcoidal granulomas. (H&E x100) |
Case 1. A 59-year-old African-American female presented complaining of alopecia. Physical examination revealed multiple sclerotic, scaling, scarred alopecic plaques on the scalp. The patient initially declined scalp biopsy and was treated with topical steroids. She returned nine months later with marked progression of alopecia and scarring. Clinically, a concern for a primary scarring alopecia such as lichen planopilaris or lupus erythematosus was noted by the dermatologist.
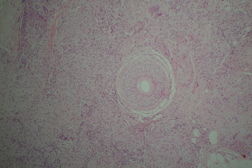
A punch biopsy taken from the crown of the scalp was embedded transversely and showed well-circumscribed granulomas associated with giant cells and mild lymphocytic infiltrates. Dermal fibrosis was noted and there was a decrease in the number of hair follicles. Foreign material was not identified in the routine sections but, upon polarization, minute fragments of refractile material were identified. Fite and GMS staining were negative for organisms.
Further evaluation of the patient revealed a distant history of pulmonary sarcoidosis and inspection of non-scalp skin revealed a small papule on the alar crease suggestive of papular sarcoidosis.
Case 2. A 50-year-old African-American male presented with alopecia of four years duration. It began as a pruritic plaque with papules on the vertex. He was initially managed with topical and intralesional corticosteroids. Following the development of pustules, he was diagnosed clinically as having folliculitis decalvans and was treated sequentially and unsuccessfully with several antibiotics including doxycycline, keflex, rifampin, and clindamycin; limited improvement was noted. The patient was then referred to the University of Pittsburgh where he was noted to have a reddish-brown alopecic plaque on the vertex with extension onto the frontal and temporal hairlines.
A biopsy revealed non-caseating granulomas consistent with sarcoidosis. Systemic evaluation revealed no pulmonary, ocular, cardiac, or other systemic involvement.
Discussion
Sarcoidosis is a multi system disorder of unknown etiology characterized by formation of non-caseating epithelioid granulomas. Organs most commonly affected include lungs, lymph nodes, eyes, and skin. Cutaneous involvement, seen in 25 percent of cases, usually occurs at the onset of disease and can be classified as specific or non-specific. Specific lesions show granulomas histologically and include maculopapules, nodules, plaques, subcutaneous nodules, infiltration of scars and lupus pernio. Non-specific lesions, which are not related to underlying granulomatous disease, include erythema nodosum, calcinosis cutis, prurigo, erythema multiforme, and nail changes [1, 2, 3, 4].
A review of Pubmed search engine of the English language literature reveals 39 published cases of sarcoidosis-induced alopecia [4-32]. The age range occurred between 23 and 78 years with a predominance of females (31 of the 35 in which gender is specified). Of the 35 cases in which race is mentioned, 20 patients were African or African-American. Cutaneous involvement in addition to the scalp is common, with 19 of 25 cases in which this is addressed revealing additional cutaneous lesions, the head and neck being the most common sites. Most patients with alopecia related to sarcoidosis have systemic involvement; of the 35 reports supplying this information, 30 report systemic disease, most commonly pulmonary and lymph node involvement. Therefore, careful and complete skin examination is indicated along with a work up for systemic sarcoidosis.
Scarring alopecia secondary to sarcoidosis is common [5, 8, 10, 14, 15, 20, 25, 26, 30] although non-scarring alopecia has also been described [5, 9, 16] including a case of alopecia involving only the legs [28] and a case of total body alopecia [19]. Localized scalp involvement is common but rarely, diffuse alopecia can occur [12, 13]. Indurated plaques and papules may be present [5, 6, 12, 30] and erythema with scale has been described [14, 27, 31]. Scalp lesions clinically resembling discoid lupus erythematosus [6, 18, 32], lichen planopilaris [7], and necrobiosis lipoidica [15, 22] have been reported.
Response to therapy is poor and most often consists of topical and intralesional corticosteroids, systemic corticosteroids, and antimalarials. In the 5 case reports detailing some degree of improvement, systemic corticosteroids are most frequent [19, 22, 24], but one patient partially responded to intralesional corticosteroids [25] and another to methotrexate [26]. In some cases, treatment may halt progression of the process but does not result in hair regrowth [7, 8].
The histologic features of scarring alopecia related to sarcoidosis consist of non-caseating “naked” granulomas, similar to other forms of involvement by sarcoidosis [33]. These findings allow distinction from other forms of scarring alopecia such as lichen planopilaris, which shows lichenoid lymphocytic infiltrates with necrotic keratinocytes of follicles and sometimes of the surface epidermis. Discoid lupus erythematosus typically shows follicular plugging, vacuolar interface changes, superficial and deep perivascular and periadnexal lymphocytic infiltrates, and variable dermal mucin deposition. Other forms of lymphocyte-mediated primary cicatricial alopecia include central, centrifugal scarring alopecia, which may show granulomas in late stages. However, these are associated with ruptured follicles and hair fibers only. Other features include premature desquamation of the inner root sheath and perifollicular lymphocytic infiltrates. Neutrophil-mediated cicatricial alopecias, including folliculitis decalvans and dissecting cellulitis, show destructive suppurative folliculitis with the latter also demonstrating abscesses and sinus tracts [34, 35].
Similar to other forms of scarring alopecia, in sarcoidal alopecia, destruction of hair follicles and fibrosis may be seen depending on the stage of the lesion [5, 6]. Birefringent foreign material, as noted in Case 1, has been described in 22 to 77 percent of biopsies of cutaneous sarcoidosis [33, 36] and it has been postulated that foreign material may act as a nidus for granuloma formation in sarcoidosis patients. These findings of polarizing materials with non-caseating granulomas should not prevent the evaluation for systemic sarcoidosis. We were not able to elicit a history of trauma or other etiology for foreign material in our patient, but because the fragments were uniformly distributed throughout the tissue and all were <1 µm in diameter, we suggest that they more likely represent contamination.
Patients presenting with alopecia secondary to scalp involvement by sarcoidosis should be evaluated for other cutaneous manifestations and should undergo complete workup for systemic sarcoidosis because most reported cases have exhibited systemic disease, particularly thoracic involvement. Skin biopsies of sarcoidosis not infrequently demonstrate polarizable foreign material suggesting that foreign body may serve as a nidus for granuloma formation but should not prevent systemic evaluation.
References
1. English JC 3rd, Callen JP. Sarcoidosis. In : Callen JP et al, eds. Dermatologic Signs of Internal Disease. China, Saunders Elsevier, 2009: 287-95.2. Fernandez-Faith E, McDonnell J. Cutaneous sarcoidosis: differential diagnosis. Clin Dermatol. 2007;25:276-87. [PubMed]
3. English JC, Patel PJ, Greer KE. Sarcoidosis. J Am Acad Dermatol. 2001;44:725-43. [PubMed]
4. Mangas C, Fernandez-Figueras M, Fite E, et al. Clinical spectrum and histological analysis of 32 cases of specific cutaneous sarcoidosis. J Cutan Pathol. 2006;33:772-77. [PubMed]
5. Katta R, Nelson B, Chen D, Roenigk H. Sarcoidosis of the scalp: a case series and review of the literature. J Am Acad Dermatol. 2000;42:690-2. [PubMed]
6. Henderson CL, Lafleur L, Sontheirmer RD. Sarcoidal alopecia as a mimic of discoid lupus erythematosus. J Am Acad Dermatol. 2008;59:143-5. [PubMed]
7. LaPlaca M, Vincenzi C, Misciali C, Tosti A. Scalp sarcoidosis with systemic involvement. J Am Acad Dermatol. 2008;59:S126-7. [PubMed]
8. Douri T, Chawaf AZ, Alrefaee BA. Cicatricial alopecia due to sarcoidosis. Dermatol Online J. 2003;9:16. [PubMed]
9. Akhdari N, Skalli HD, Lakhdar H. Erythematous lesions on the scalp. Arch Dermatol. 2004;140:1003-8. [PubMed]
10. Takahashi H, Mori M, Muraoka S. et al. Sarcoidosis presenting as a scarring alopecia: report of a rare cutaneous manifestation of systemic sarcoidosis. Dermatology. 1996;193(2):144-6. [PubMed]
11. Bleehen SS. Systemic sarcoidosis with scalp involvement. Proc R Soc Med. 1969;62:348-9. [PubMed]
12. Baker H. Atrophic alopecia due to granulomatous infiltration of the scalp in systemic sarcoidosis. Proc R Soc Med. 1965;58:243-4. [PubMed]
13. Bluefarb SM, Szymanski FJ, Rostenberg A. Sarcoidosis as a cause of patchy alpecia. Arch Dermatol. 1955;71:602-4. [PubMed]
14. Golitz LE, Shapiro L, Hurwitz E. Stritzler R. Cicatricial alopecia of sarcoidosis. Arch Dermatol. 1973;107-758-60. [PubMed]
15. Andersen KE. Systemic sarcoidosis with necrobiosis lipoidica-like scalp lesions. Acta Derm Venereol. (Stockh) 1977;57:367-9. [PubMed]
16. Greer KE, Harman LE, Allan KL. Unusual cutaneous manifestations of sarcoidosis. South Med J. 1977;70:666-8. [PubMed]
17. Harrison GN, Lipham M, Elguindi AS, Loebl DH. Acute sarcoidosis occurring during the course of systemic lupus erythematosus. South Med J. 1979;72:1387-8. [PubMed]
18. Jacyk WK. Sarcoisosis in the West African. A report of eight Nigerian patients with cutaneous lesions. Trop Geogr Med. 1984;36:231-6. [PubMed]
19. Smith SR, Kendall MJ, Kondratowicz GM. Sarcoidosis: a cause of steroid-responsive total alopecia. Postgrad Med J. 1986;62:205-7. [PubMed]
20. Bazex J, Dupin P, Giordano F. Sarcoidose cutanee et viscerale. Ann Dermatol Venereol. 1987;114:685-90. [PubMed]
21. Weiler V, Redtenbacher S, Bancher C, et al. Concurrence of sarcoidosis and aortitis: case report and review of the literature. Ann Rheum Dis. 2000;59:850-3. [PubMed]
22. Harman KE, Calonje E, Robson A, Black MM. Sarcoidosis presenting as a scarring alopecia resembling necrobiosis lipoidica. Clin Exp Dermatol. 2003;28:565-6. [PubMed]
23. Gutz VS, Gillissen A. Welche systemerkrankung steckt dahinter? MMW Fortschr Med. 2002;144:42-5. [PubMed]
24. Cho HR, Shah A, Hadi S. Systemic sarcoidosis presenting with alopecia of the scalp. Int J Dermatol. 2004;43:520-2. [PubMed]
25. Chong WS, Tan HH, Tan SH. Cutaneous sarcoidosis in Asians: a report of 25 patients from Singapore. Clin Exp Derm. 2005;30:120-4. [PubMed]
26. Mosam A, Morar N. Recalcitrant cutaneous sarcoidosis: an evidence-based sequential approach. J Dermatol Treat. 2004;15:353-9. [PubMed]
27. Costabel U, Guzman J, Baughman RP. Systemic evaluation of a potential cutaneous sarcoidosis patient. Clin Dermatol. 2007;25:303-11. [PubMed]
28. Rapp SE. An unusual cause of hair loss. Arch Dermatol. 2002;138:259-64. [PubMed]
29. Morrison JGL. Sarcoidosis in the Bantu: necrotizing and mutilating forms of the disease. Br J Dermatol. 1974;90:649-55. [PubMed]
30. Rudolph RI, Holzwanger JM, Heaton CL. Diffuse cicatricial alopecia of the scalp caused by sarcoidosis. Cutis. 1975;15:524-8.
31. Maurice PD, Goolamali SK. Sarcoidosis of the scalp presenting as scarring alopecia. Br J Dermatol. 1988;110(Suppl 33):116-7.
32. Sauter LS. Sarcoidosis simulating disseminated discoid lupus erythematosus. Arch Dermatol. 1966;94:670-2. [PubMed]
33. Ball NJ, Kho GT, Martinka M. The histologic spectrum of cutaneous sarcoidosis: a study of twenty-eight cases. J Cutan Pathol. 2004;31:160-8. [PubMed]
34. Olsen E, Bergfeld W, Cotsarelis G, et. al. Summary of North American Hair Research Society (NAHRS)-sponsored workshop in cicatricial alopecia. Duke University Medical Center, February 10 and 11, 2001. J Am Acad Dermatol. 2003;48:103-10. [PubMed]
35. Ross EK Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol. 2005;53:1-37. [PubMed]
36. Marcoval J, Mana J, Moreno A, et. Al. Foreign bodies in granulomatous cutaneous lesions of patients with systemic sarcoidosis. Arch Dermatol. 2001;137:427-30. [PubMed]
© 2012 Dermatology Online Journal