Mucous membrane plasmacytosis: A case report and review of the literature
Published Web Location
https://doi.org/10.5070/D30bn022pzMain Content
Mucous membrane plasmacytosis: A case report and review of the literature
Rakesh Bharti MD1, and Denise R Smith MA2
Dermatology Online Journal 9 (5): 15
From the BDC Research Centre1, Amritsar, Punjab, INDIA, and University of California Davis School of Medicine2
Abstract
Mucous membrane plasmacytosis is a rare, idiopathic condition consisting of a dense plasma-cell infiltrate of the mucous membranes. Zoon first described a plasma-cell infiltrate occurring on the glans penis and plasmacytosis involving other body orifices have been reported under a variety of different terms. White et al. simplified the terminology by suggesting all plasma-cell infiltrates of the mucous membranes of body orifices be termed plasma-cell orificial mucositis. The differential diagnoses and treatment for mucous membrane plasmacytosis are summarized. A middle-aged female with an unusual plasma-cell proliferation disorder of the upper aerodigestive tract is reported for its rarity.
Introduction
Mucous membrane plasmacytosis of the upper aerodigestive tract is a rare, benign disorder in which the mucous membranes are infiltrated by plasma cells. There is no known relation to recognizable dermatoses or neoplasms. The phenomenon of plasma-cell infiltrate was first described by Zoon [1] in 1952 when he described balantitis plasma cellularis. Since then, plasma-cell infiltrates have been found on the vulva, buccal mucosa, palate, nasal aperture, gingiva, lips, tongue, epiglottis, larynx, and other orificial surfaces. These conditions have been described with a variety of different terms. During the late 1960s and early 1970s, cases of plasma-cell infiltrates of the lips, gums, and tongue were described primarily in the dental literature under the names atypical gingivostomatitis [2, 3], idiopathic gingivostomatitis [4], and allergic gingivostomatitis [5]. The lesions were thought to be a result of a reaction to chewing gum, dentifrices, and other foreign substances [2, 4, 5, 6, 7, 8, 9, 10], although extensive allergy testing has been inconclusive [3, 11]. Sherman [12] and Luders [13] simplified the nomenclature in 1960 and 1973, respectively, by grouping the infiltrates by anatomy under the titles plasmacytosis circumorificialis and plasmacytosis mucosae. However, additional terms have been used in the literature to describe plasma-cell infiltrates of the aerodigestive tract, such as plasma cell-gingivitis [6, 7, 11, 14], plasmacytosis of the gingival [9], and plasma-cell cheilitis [15, 16]. In 1986, White et al. [17] grouped all plasma-cell infiltrates of the aerodigestive tract under the name plasma-cell orificial mucositis because of the fact that all the cases reported had clinical and histologic findings that were indistinguishable from one another, eliminating the need for separate names for each anatomic area. Since that time, plasma-cell orificial mucositis, or variants of the name such as mucous membrane plasmacytosis and plasma-cell mucositis, have been used to describe benign plasmacytic lesions of the aerodigestive tract in the majority of cases reported [18, 19, 20, 21, 22, 23].
Clinically, the lesions of mucous membrane plasmacytosis may present with a variety of symptoms and morphologies depending on the area of the aerodigestive tract in which it occurs. When appearing on the lips and gums, patients complain of sore, swollen lips and gums. Examination reveals a well-circumscribed, soft, slightly elevated, edematous mass in which the surface is red and glistening but not ulcerated. When occurring in other parts of the aerodigestive tract, such as the larynx, pharynx, palate, buccal mucosa, and trachea, patients complain of oral pain, dysphonia, dysphasia and difficulty breathing. On examination the lesions typically appear cobblestone- or wart-like[18]. Several cases have been reported in which patients demonstrate simultaneous plasma-cell infiltrate lesions of the gingiva and the pharynx or larynx [17, 18, 24, 25].
Histologically, the majority of lesions are described as having an acanthotic epidermis with narrow and elongated rete ridges, spongiosis and a dense subepithelial cellular infiltrate composed largely of mature plasma cells, but also including a few polymorphonuclear leukocytes and lymphocytes. Occasional Russell bodies (accumulation of immunoglobulin within cytoplasm of plasma cells) were occasionally noted [18]. The plasma cells do not show anaplasia or prominent nucleoli. In addition, there are reports of plasma-cell orificial mucositis that show epidermal atrophy [13] and eroded epidermis [18, 26, 27]. Immunoperoxidase staining show mixed population of kappa and lambda light chains and various heavy chains, demonstrating the polytypic and benign nature of the condition.
Case report
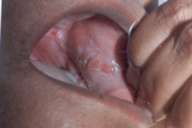
A 42-year-old married woman presented to the Bharti Derma Care and Research Centre in Amritsar, Punjab with a 1-year history of oral pain and dysphagia associated with ulcers in the mouth, mainly involving the buccal mucosa and palate. She had a cobblestone appearance of the palate and bilateral buccal mucosa. (Fig. 1) The patient had previously been treated with topical glycerin and vitamin-B-complex tablets with no improvement. The patient did not have systemic illness. The initial clinical impression included lichen planus, cicatricial pemphigoid, and deep fungal infection.
All the laboratory tests, including CBC, LFT, RF, Montoux, ESR, STS, LE cell, ANA and anti-DNA, were within normal limits. Absence of hyphae ruled out fungal infection.
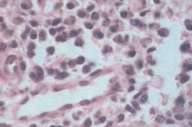
A biopsy was taken from left buccal mucosa. The initial report on hematoxylin-eosin (H & E) staining interpreted the findings as normal, intact, stratified squamous epithelium, with an underlying inflammatory infiltrate consisting mainly of lymphocytes and plasma cells, with several macrophages. No evidence of suprabasal bulla, acantholysis or increased mitotic activity was noted. Subsequently, a consultant pathologist noted that the submucosa contained dense, diffuse sheets of infiltrate, with many plasma cells mixed with of neutrophils, eosinophils, and lymphocytes (Fig. 2). Many Russell bodies were also observed and the overlying epithelium was ulcerated.
Cicatricial pemphigoid, lichen planus, and malignancy were ruled out by the histologic appearance. Serologic testing for syphilis was negative. Protein electrophoresis and immunohistochemistry were not performed for lack of resources. The diagnosis of mucous membrane plasmacytosis is most consistent with the clinical and histologic data. Treatments used included topical and systemic antifungals and corticosteroids, which provided symptomatic relief but did not cause a regression of the disease. The patient is still undergoing treatment with local and systemic corticosteroids.
|
|
| Figure 1 | Figure 2 |
|---|---|
| Ulcer on the buccal mucosa (Fig. 1). High power micrograph showing sheets of plasma cells (Fig. 2). | |
Differential Diagnosis
We report a case of oral lesions in the upper aerodigestive tract, which, on histology, demonstrate a dense plasma-cell infiltrate. Several other disorders may present with lesions that appear similar clinically or histologically including fungal infection, carcinoma, syphilis, lichen planus, cicatricial pemphigoid, allergic or contact mucositis, sarcoidosis, cheilitis granulomatosa, plasma-cell granuloma, plasmoacanthoma, rhinoscleroma, Rosai-Dorfman disease, Melkerson-Rosenthal syndrome, and extramedullary plasmacytoma. Several of these diagnoses can be ruled out by histology or further testing for an infectious process (Table 1).
Fungal infection can mimic mucous membrane plasmacytosis but is usually distinguished by absence of fungal hyphae on microscopic examination, negative KOH preparation, no growth on culture, or a lack of response to treatment with nystatin. Histologic examination of the tissue can exclude carcinoma from the differential diagnosis.
Mucous membrane lesions are present in one-third of patients with secondary syphilis, and histology can show superficial and deep perivascular infiltrate of plasma cells, lymphocytes and macrophages distributed in a bandlike pattern in the dermis, accompanied by psoriasiform epidermal hyperplasia and hyperkeratosis [28]. However, negative serologic tests for syphilis and absence of spirochetes on a silver stain of tissue sections can rule out secondary syphilis as the cause for the mucous membrane lesions.
When lichen planus occurs on mucous membranes, these often painful lesions are usually white plaques, typically with a lacelike pattern. Other presentations of mucous membrane lichen planus include atrophy, erosions or ulcers, and bullae. Microscopically, lichen planus is characterized by a superficial, bandlike, lymphocytic infiltrate and occasionally hyperkeratosis, but no significant accumulation of plasma cells.
Cicatricial pemphigoid (benign mucosal pemphigoid) can clinically resemble mucous membrane plasmacytosis. The oral mucosa of patients with cicatricial pemphigoid is almost always involved, including the nasal mucosa, nasopharynx, larynx, oropharynx, hypopharynx, and esophagus. Oral lesions are characterized by evanescent vesicles that quickly rupture and leave erosions and ulcers, as opposed to the raised and cobblestone-appearing lesions of mucous membrane plasmacytosis. Cicatricial pemphigoid typically affects middle-aged to elderly women whereas mucous membrane plasmacytosis tends to affect elderly men. Histologically, there is subepithelial disruption or ulceration but no psoriasiform changes or plasmacytosis as expected with mucous membrane plasmacytosis [18, 28].
Allergic or irritant contact dermatitis can initially be ruled out by history and by distribution of the lesions. Dentures or foreign objects can be removed to rule out contact dermatitis and patch testing can be done to detect any other contact or allergic factors. Histologically, lesions of allergic contact dermatitis reveal spongiosis with infiltrate of monocytes and histiocytes in the dermis. Primary irritant dermatitis presents with superficial vesicles containing polymorphonuclear leukocytes.
The chronic, granulomatous disease sarcoidosis can affect mucous membranes, including the palate, tongue, buccal mucosa or posterior pharynx. In the oral cavity, the lesions are papules that may coalesce to form a flat plaque [28]. Microscopic examination reveals large, epithelioid histiocytes with a few giant cells and lymphocytes, and occasional asteroid bodies with no evidence of a plasma-cell infiltrate.
Mucous membrane plasmacytosis can clinically resemble cheilitis granulomatosa; however, histopathology shows evidence of a granulomatous infiltrate rather than a plasma-cell infiltrate.
Plasma-cell granulomas tend to locate in the oral cavity, primarily on the periodontal tissue. These lesions are often single; whereas, the lesions of mucous membrane plasmacytosis tend to be multiple. On histologic evaluation, plasma cells are prominent but are intermixed with abundant other cellular elements and usually surrounded by connective-tissue septae, distinguishing it from mucous membrane plasmacytosis [29].
Plasmoacanthoma is described as a discrete, verrucous tumor with hyperkeratosis, pronounced acanthosis of the epidermis and plasmacytic infiltrate in the dermis on histologic examination. Plasmoacanthoma is considered a separate disease entity from plasma-cell orificial mucositis; however, in 1995 van de Kerhof and van Baar [23] described the simultaneous occurrence of these conditions in a patient and suggested that both conditions are part of one single disease entity.
Rhinoscleroma is a chronic, inflammatory, granulomatous disease of the upper aerodigestive tract, associated with Klebsiella rhinoscleromatis infection, which can have a clinical presentation similar to that of mucous membrane plasmacytosis. Rhinoscleroma affects both sexes equally, most commonly during the third and fourth decades in persons from Russia, Central Europe, Egypt, Pakistan, Africa, and South and Central America. Histology reveals a granulomatous tumor consisting primarily of plasma cells, an occasional Russell body, a few spindle cells and hypertrophic collagenous tissue. Warthin-Starry silver stain can allow for visualization of the bacilli of rhinoscleroma within the foamy macrophages (Mikulicz's cells). Culture and serologic tests can also be performed to rule out rhinoscleroma [28].
Rosai-Dorfman disease (sinus histiocytosis with massive lymphadenopathy) can involve the head and neck [18 ]. Most patients who present with skin lesions are 40 years old or older at presentation. The lesions can be isolated or disseminated yellow-brown papules or nodules or macular erythema. The histopathologic features of Rosai-Dorfman disease are a superficial and deep, perivascular infiltrate of lymphocytes and plasma cells, in addition to a diffuse dermal infiltrate of large, foamy histiocytes. Additionally, fibrosis and lymphophagocytosis are characteristically present, features not common in mucous membrane plasmacytosis. The elements of epithelial psoriasiform changes and dyskeratosis typical of mucous membrane plasmacytosis are not seen in Rosai-Dorfman disease [28].
Melkerson-Rosenthal syndrome consists of a triad of recurring facial paralysis or paresis; soft, nonpitting edema of the lips; and scrotal tongue. Usually starting in adolescence, the first symptoms can be the swelling of the skin and mucous membranes of the face and mouth. A tuberculoid type of granuloma with lymphedema and a banal perivascular infiltrate are seen on microscopic examination [28].
On the basis of histology, extramedullary or primary cutaneous plasmacytoma needs to be considered. This tumor is found in the upper respiratory tract in approximately 80 percent of cases, especially in the nasal cavity and sinuses, nasopharynx, and larynx and can be sessile, polypoid or pedunculated [29]. Histologically, plasmacytomas are composed of a diffuse infiltrate of plasma cells in the dermis and subcutaneous tissue. There is minimal to prominent nuclear atypical of the plasma cells and on immunohistochemistry they are monoclonal, distinguishing plasmacytoma from plasmacytosis. Gene-rearrangement studies can be done if immunohistochemistry is inconclusive.
Therapy and prognosis
Although several treatment modalities have been tried, including corticosteroids (topical, intralesional, and systemic), antibiotics, destruction of the tissue (liquid nitrogen, CO2 laser and electrocoagulation), excision of the tissue, and radiation therapy, no treatment clearly stands out as consistently effective. (Table 2)
Corticosteroids have been used with the most frequency but with inconsistent results. A review of the literature by White et al. [17] in 1986 revealed that topical and intralesionally administered steroids were of no benefit and other studies have reported some symptomatic improvement but no true regression of the disease [10, 11, 15, 18, 23]. Additionally, the patient reported in this case report has not responded to topical or systemic corticosteroids. A few cases in which corticosteroids induced marked regression with residual scarring [24, 25], as well as total eradication of the disease have been reported [19, 20, 21].
Antibiotics have also been used to treat mucous membrane plasmacytosis. Treatment with oral nystatin was found to be ineffective in the patient described in this case report as well in another case report [17]. Ferrerio et al. [18] report on two patients treated with antibiotics (type unspecified) without effect. In one case, 2 percent fusidic acid was found to be an effective treatment [27] and ineffective in another [30]. Tamaki et al.[16] described two patients with plasma-cell cheilitis who responded to oral griseofulvin with complete remission of lesions.
Fogarty [31] reports on a case of gingival plasmacytosis that had progressed to involve the larynx and was treated with systemic chemotherapy (cyclophosphamide, vincristine, and prednisolone) with temporary improvement, but symptoms progressed after therapy was discontinued. Eventually, this patient was treated with low-dose radiation therapy, which subsequently caused symptomatic improvement, which was maintained at 12month followup.
Several authors report on resorting to debulking procedures, either surgical excision, electrocoagulation, carbon dioxide laser, or cryotherapy, to reduce the mass effect of the mucous membrane plasmacytosis involving the pharynx and larynx [17, 18, 24, 30, 31]. These procedures resulted in improvement of symptoms; however, the procedures are a temporary measure as the lesions and symptoms recurred. In fact, a few patients have required a tracheostomy, owing to the progression of the mucous membrane plasmacytosis lesions causing subglottic strictures [18, 31].
Long-term prognosis for patients with mucous membrane plasmacytosis appears good. Ferreira [18] reports that the majority of the cases he followed (seven of nine) had no progression nor regression of their disease, and the long-term survival appeared good with the longest survivor living 16 years with the disease. However, a few studies have demonstrated progression of the disease from the oral mucosa to the larynx [17, 18, 21, 24, 25, 31], occasionally causing sufficient airway obstruction to require a tracheostomy [18, 31]. No studies to date report a progression of benign mucous membrane plasmacytosis to a malignancy of any type.
Conclusion
Mucous membrane plasmacytosis is a diagnosis of exclusion, distinguished primarily on the histologic finding of a marked submucosal plasma-cell infiltrate in addition to psoriasiform epithelial changes, after conditions such as infection and plasmacytoma have been eliminated. The etiology of this condition is unclear but is believed to be a nonspecific inflammatory response, in the form of a plasma-cell infiltrate, to an unknown exogenous agent. Attempts to induce plasma-cell infiltrations on mucosal and nonmucosal surfaces by allergic and irritant stimuli were not successful [15]. Roman [30] hypothesized that plasma-cell gingivitis may be associated with low levels of serum IgA and secretory IgA, which allows localized, repetitive, subclinical infections that could lead to the plasma-cell infiltrate. Aiba [26] points out that a plasma-cell infiltrate is a rare histologic feature in ordinary inflammatory dermatoses but is often found around such epidermal neoplasms as actinic keratosis, Bowen disease, squamous cell carcinoma, and syringocystadenoma papilliferum. The authors hypothesize that, although the inciting factor of the plasma-cell infiltrate is unknown, it is plausible that similar mechanisms are involved in both the mucosal and skin plasma-cell infiltrate conditions. The best treatment for mucous membrane plasmacytosis is also unclear, with inconsistent data from trials with corticosteroids, antibiotics, radiation, ablative therapy, and surgical excision. Mucous membrane plasmacytosis should be included in the differential diagnosis of swelling or lesions of the aerodigestive tract; typical histology can easily confirm the diagnosis.
References
1. Zoon JJ. Balanoposthite chronique circonscrite benign a plasmocytes (contra erythroplasie de Queyrat). Dermatologica 1952; 105: 1-7.2. Owings JR Jr. An atypical gingivostomatitis: a report of four cases. J Periodontol 1969; 40: 538-542.
3. Perry HO, Deffner NF, Sheridan PJ. Atypical gingivostomatitis. Nineteen cases. Arch Dermatol 1973; 107: 872-878.
4. Kerr DA, McClatchey KD, Regezi JA. Idiopathic gingivostomatitis. Cheilitis, glossitis, gingivitis syndrome; atypical gingivostomatitis, plasma-cell gingivitis, plasmacytosis of gingival. Oral Surg Oral Med Oral Pathol 1971; 32: 402-423.
5. Kerr DA, McClatchey KD, Regezi JA. Allergic gingivostomatitis (due to gum chewing). J Periodontol 1971; 42: 709-712.
6. Lubow RM, Cooley RL, Hartman KS, McDaniel RK. Plasma-cell gingivitis. Report of a case. J Periodontol 1984; 55: 235-241.
7. MacLeod RI, Ellis JR. Plasma cell gingivitis related to the use of herbal toothpaste Br Dent J. 1989; 166: 375-376.
8. Perry HO. Idiopathic gingivostomatitis. Dermatol Clin 1987; 5: 719-722.
9. Poswillo D. Plasmacytosis of the gingiva. Br J Oral Surg 1968; 5: 194-202.
10. Silverman S Jr, Lozada F. An epilogue to plasma-cell gingivostomatitis (allergic gingivostomatitis). Oral Surg Oral Med Oral Pathol 1977; 43: 211-217.
11. Sollecito TP, Greenberg MS. Plasma cell gingivitis. Report of two cases. Oral Surg Oral Med Oral Pathol 1992; 73: 690-693.
12. Schuermann H. Plasmacytosis circumorificialis. Dtsch Zahnarztl 1960; 15: 601-610.
13. Luders G. Plasmocytosis mucosae: Ein oft verkanntes neues Krankheitsbild. Munch ed Wochenschr 1972; 114: 8-12.
14. Palmer RM, Eveson JW. Plasma-cell gingivitis. Oral Surg Oral Med Oral Pathol 1981; 51: 187-189.
15. Baughman RD, Berger P, Pringle WM. Plasma cell cheilitis. Arch Dermatol 1974; 110: 725-6.
16. Tamaki K, Osada A, Tsukamoto K, Ohtake N, Furue M Treatment of plasma cell cheilitis with griseofulvin. J Am Acad Dermatol. 1994; 30: 789-790.
17. White JW Jr, Olsen KD, Banks PM. Plasma cell orificial mucositis. Report of a case and review of the literature. Arch Dermatol 1986; 122: 1321-1324.
18. Ferreiro JA, Egorshin EV, Olsen KD, Banks PM, Weiland LH. Mucous membrane plasmacytosis of the upper aerodigestive tract. A clinic pathologic study. Am J Surg Pathol 1994; 18: 1048-1053.
19. Jones SK, Kennedy CT. Response of plasma cell orificial mucositis to topically applied steroids. Arch Dermatol 1988; 124: 1871-1872.
20. Kaur C, Thami GP, Sarkar R, Kanwar AJ. Plasma cell mucositis. J Eur Acad Dermatol Venereol 2001; 15: 566-567.
21. Khan NA, McKerrow WS, Palmer TJ. Mucous membrane plasmacytosis of the upper aerodigestive tract. A case report with effective treatment. J Laryngol Otol 1997; 111: 293-295.
22. Noorily AD. Plasma cell orificial mucositis. Otolaryngol Head Neck Surg 1997; 116: 416-417.
23. Van de Kerkhof PC, van Baar HM. Co-occurrence of plasma cell orificial mucositis and plasmoacanthoma. Report of a case and review of the literature. Dermatology 1995; 191: 53-55.
24. Timms MS, Sloan P. Association of supraglottic and gingival idiopathic plasmacytosis. Oral Surg Oral Med Oral Pathol 1991; 71: 451-453.
25. Timms MS, Sloan P, Balzan AP. Idiopathic plasmacytosis of the oral and supraglottic mucosa. J Laryngol Otol 1988; 102: 646-648.
26. Aiba S, Tagami H. Immunoglobulin-producing cells in plasma cell orificial mucositis. J Cutan Pathol 1989; 1: 207-210.
27. Mahler V, Hornstein OP, Kiesewetter F. Plasma cell gingivitis: treatment with 2% fusidic acid. J Am Acad Dermatol 1996; 34: 145-146.
28. Odom, RB, James, WD, Berger, TG. Andrew's Diseases of the Skin Clinical Dermatology, 9th Edition. Philadelphia: W.B. Saunders Co., 2000.
29. Bataskis, JG. Plasma cell tumors of the head and neck. Ann Otol Rhinol Laryngol 1983; 92: 311-313.
30. Roman CC, Yuste CM, Gonzalez MA, Gonzalez AP, Lopez G. Plasma cell gingivitis. Cutis 2002; 69: 41-45.
31. Fogarty G, Turner H, Corry J. plasma-cell infiltration of the upper aerodigestive tract treated with radiation therapy. J Laryngol Otol 2001; 115: 928-930.
© 2003 Dermatology Online Journal

