Skin metastases from lung cancer
Published Web Location
https://doi.org/10.5070/D39r83m6wjMain Content
Skin metastases from lung cancer
Todd W Mollet1, Carlos A Garcia MD2, Glenn Koester MD3
Dermatology Online Journal 15 (5): 1
1. College of Medicine, University of Oklahoma. todd-mollet@ouhsc.edu2. Department of Dermatology, University of Oklahoma. carlos-garcia@ouhsc.edu
3. Dermatology private practice, Edmond, Oklahoma. edmondderm@cs.com
Abstract
Cutaneous metastases from the lung are rare but must be ruled out in patients with suspicious skin lesions and history of smoking or lung cancer. All histological types of lung cancer may metastasize to the skin and clinical lesions are variable. The percentage of patients with lung cancer that develop cutaneous metastases ranges from 1 to 12 percent. In 20-60 percent of cases the skin lesions present before or synchronously with the diagnosis of the primary tumor. Skin lesions are often described as nodular, mobile or fixed, hard or flexible, single or multiple, and painless. Histologically, cutaneous metastases from the lung are frequently moderately or poorly differentiated. IHC markers that may be useful in these cases are anti-thyroid transcription factor (TTF) and CK7/20. Treatment of solitary cutaneous metastases usually includes surgery alone or combined with chemotherapy, and/or radiation. If multiple cutaneous lesions or internal metastases exist, chemotherapy is the primary option. Cutaneous metastases and their primaries in the lung are usually incurable and suggest an unfortunate prognosis. Poor prognostic indicators include non-resectable or small-cell primary tumors, multiple cutaneous metastases, or other distant metastases. Mean survival is usually about 5-6 months.
Introduction
Lung cancer is the second most common type of malignancy and the leading cause of death from cancer [1]. Its incidence is decreasing in men but increasing in women, and the most common age group is 55 to 65 years old. Prognosis of lung cancer is quite poor with an overall 5-year survival rate of about 15 percent. The most common histologic type is adenocarcinoma, followed by squamous cell carcinoma, small cell carcinoma, large cell carcinoma, and bronchial carcinoid. Frequent metastatic sites for lung cancer include hilar nodes, adrenal glands, liver, brain, and bone [2]. Cutaneous metastases from the lung are rare but must be ruled out in patients with suspicious skin lesions and history of smoking or lung cancer. All histological types of lung cancer may metastasize to the skin and clinical lesions are variable.
Several articles have discussed features of cutaneous metastases from lung cancer including incidence, location, gross presentation, histology, immunohistochemistry, ultrastructural characteristics, diagnosis, surgery, chemotherapy, radiation, and prognosis. To the best of our knowledge, however, there are no publications covering all these aspects in one review. Our purpose is to summarize current knowledge on this topic in a brief but comprehensive paper for future reference.
Methods
We performed a Medline search using PubMed, and performed a manual search of pertinent references and textbooks (Table 1). Keywords and combination of terms included "lung cancer, cutaneous, skin, metastases, tumor, carcinoma, histology, immunohistochemistry, cytokeratin, and TTF." We selected 45 articles that discussed cutaneous metastases from internal cancers including lung cancer. We included 22 retrospective studies, 16 case reports, 6 reviews, and 1 prospective study. There were no randomized controlled trials or case-control studies available.
Results
Incidence of Cutaneous Metastasis from Lung Cancer
Malignancies from the lung, breast, melanoma, oral cavity, colon, kidney, ovary, and stomach account for 80 percent to 90 percent of all cutaneous metastases in adults [3, 4, 5], but there are conflicting data regarding the etiology of skin metastases according to gender.
Previous literature shows that in men with skin metastases, the lung is the most common primary site (24%), followed by colorectal cancer (19%), melanoma (13%), and the oral cavity (12%). In women, the lung has been reported as the fourth most common primary site (4%) after the breast (69%), large intestine (9%), melanoma (5%), and ovary (4%). [4] In a more recent study, however, skin metastases in men most often arose from melanoma (32.3%), lung (11.8%), colorectal (11%), oral cavity (8.7%), and an unknown site (8.7%). In women, the culprits were the breast (70.7%), melanoma (12%), ovary (3.3%), unknown site (3%), oral cavity (2.3%) and lung (2%) [6].
The percentage of patients with lung cancer that develop cutaneous metastases ranges from 1 to 12 percent [3, 7-12]. In a large series, skin was only the 13th most common site for metastases from the lung [7], but the diagnosis should be entertained in any patient with a prior history of lung malignancy or tobacco abuse [9]. Lung cancer is the fastest among internal neoplasms to metastasize to the skin after initial diagnosis (mean time 5.75 months) [13]. In 20-60 percent of cases the skin lesions present before or synchronously with the diagnosis of the primary tumor [4, 12, 14].
Cutaneous Manifestations of Lung Malignancy
Clinical Presentation
Internal malignancies generally disseminate to a site close to the primary tumor but they are capable of metastasizing anywhere on the cutaneous surface (Table 2) [15]. Lung, melanoma, and breast malignancies are the cancers most likely to metastasize to remote cutaneous sites [6]. Lung cancers usually involve the anterior chest, abdomen, and head/neck [10, 14, 15, 16]. In a study from Taiwan, skin metastases from the lung were the third most common malignancy of the scalp behind primary basal cell carcinoma and squamous cell carcinoma [17]. Another study from Japan showed that the most common site was the back [8]. Less common locations include the shoulder, flank, lower extremity, and upper extremity [10, 14, 15]. Rare sites include the gingiva, scrotum, perianal skin, lip, nose, burn scars, fingers, and toes [14, 18-23]. In some cases, cutaneous metastases may also arise at incision sites [3, 6].
 |  |
| Figure 1 | Figure 2 |
|---|---|
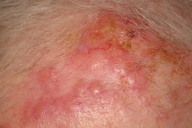
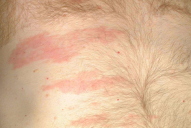
| Figure 1. Erythematous nodules on forehead from lung metastases Figure 2. Zosteriform plaque from lung cancer | |
 |
| Figure 3 |
|---|
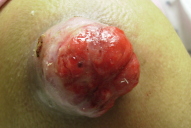
| Figure 3. Fungating lung metastases to the shoulder |
Cutaneous metastases from lung cancer do not have a characteristic presentation [10]. However, they are often described as nodular, mobile or fixed, hard or flexible, single or multiple, and painless [10, 24]. In a study of 126 patients, their colors varied from flesh-colored, red, pink, purple, or bluish black. In this same study, the sizes varied from 2 mm to 6 cm in diameter [10]. Less commonly, these lesions present as papular, plaque-like, ulcerated, vascular, zosteriform, erysipelas-like, and on the scalp as scarring alopecia [10, 25-30]. The zosteriform metastases are rare, often present on the chest or abdomen, may be painful, and are believed to be a result of penetration of the dorsal root ganglion [27, 30]. The erysipelas-like metastases may mimic true infections locally. These lesions result from traumatic seeding after chest wall procedures [26, 28], or from simple lymphatic invasion [14]. We have seen cases presenting as erythematous nodules on the forehead, a large fungating tumor on the shoulder, and zosteriform plaques on the thorax (Figs. 1-3).
Histological Presentation
Cutaneous metastases from the lung are frequently moderately or poorly differentiated [9, 24]. In fact, undifferentiated cutaneous metastases most often originate from the lung in men, breast in women, or melanoma [3, 15]. They typically invade the lymphovascular system and are usually limited to the dermis and subcutaneous layer [31]. The most common type is adenocarcinoma (ACC), followed by squamous-cell carcinoma or small-cell carcinoma, and then large-cell carcinoma (LCC) [8, 10, 14, 24, 31]. Some studies demonstrate adenocarcinoma to have the highest incidence and LCC to have the lowest incidence [10, 29]. However, two Japanese studies found large-cell carcinoma to have the highest incidence with about 10 percent of LCCs of the lung metastasizing to the skin [8, 11]. Other types of lung cancer rarely metastasizing to the skin include mesothelioma, bronchial carcinoids, bronchiolar carcinoma, mucoepidermoid carcinoma, pulmonary sarcoma, intravascular bronchioalveolar tumor, well-differentiated fetal adenocarcinoma, pleural epithelioid haemangioendothelioma, and adenoid cystic carcinoma [4, 11, 18, 32-38].
Metastatic ACCs from the lung are usually moderately differentiated [5, 31]. They sometimes show well-differentiated glandular structures or intracytoplasmic mucin [4]. In these cases, gastrointestinal, ovarian, kidney, and breast primaries should be ruled-out [3]. Metastatic squamous-cell carcinomas from the lung are often moderately or poorly differentiated [4]. In these cases, upper GI primaries should be ruled out [3]. Small-cell carcinomas are generally anaplastic and have hyperchromatic nuclei with little cytoplasm [4]. Clinical information, immunohistochemistry, and electron microscopy detecting dense-core granules can help distinguish small-cell carcinoma from other histologically similar cancers [32, 39]. Grossly, these tumors may prefer to metastasize to the back [6, 40]. LCCs simply present as undifferentiated tumors with large cells [4]. Mesotheliomas most often metastasize to the skin by direct invasion or by traumatic seeding but may rarely metastasize to distant sites [35, 36]. These tumors may resemble angioendothelioma malignancies [32]. In some cases, mesothelioma may be diagnosed over adenocarcinoma with electron microscopy [32]. Cutaneous bronchial carcinoid metastases usually show a trabecular pattern [32, 34]. Another clue to diagnosing these patients may be carcinoid syndrome [40].
Immunohistochemistry
Immunohistochemistry (IHC) may be useful when the primary site of a cutaneous metastasis is unknown and a shorter differential is desired [31]. Although not extensively studied in cutaneous metastases from the lung, markers that may be useful in these cases are anti-thyroid transcription factor (TTF) and CK7/20. Anti-TTF is both sensitive and specific for primary adenocarcinomas, bronchioalveolar carcinomas, and small-cell carcinomas when a thyroid primary is ruled-out [41]. Azoulay et al. found anti-TTF reactivity in their 1 case of lung cancer metastasizing to the skin [42]. A CK7+/20- pattern is sensitive but not specific for primary adenocarcinomas and bronchioalveolar carcinomas (Table 3) [41, 43, 44]. This pattern was found in 1 out of 1 cutaneous metastases from lung cancer by Saeed et al. and 4 out of 4 by Azoulay et al. [31, 42]. However in another study, only 5 out of 8 followed this pattern [45]. This is consistent with a review study where CK7/20 staining patterns in distant metastases deviated from those in primary lung tumors (Table 4) [46]. Given this data, IHC staining for cutaneous metastases from the lung is likely less sensitive than hoped and should be reserved for cases where clinical and histological information is inconclusive [42].
Diagnosis
Diagnosis is most often based on clinical information and histology, although as mentioned previously, immunohistochemistry and electron microscopy may be helpful [39, 42]. If the histology of the primary and metastatic lesions is similar, the diagnosis is confirmed [47]. If there is no known primary site, one must determine then if the lesion is primary or secondary [47]. Also, the histological subtype should be used to narrow the differential diagnosis [3, 32]. A primary site is investigated by history, physical exam, and multiple screening methods including CBC, CMP, chest x-ray, mammogram, US, CT, and MRI [47]. With the use of these imaging tools, Coslett et al found that 87.5 percent of patients (7/8) and Terashima et al found that 64.7 percent of patients (22/34) had primary tumors in the upper lobes of the lungs [9, 11]. Coslett et al and Molina et al propose that lung malignancies in the upper lobes have a greater tendency to metastasize to the skin [9, 48].
Treatment
Treatment of solitary cutaneous metastases usually includes surgery alone or combined with chemotherapy, and/or radiation [49, 50]. In one study, Ambrogi et al treated 2 patients with surgery and chemotherapy. One patient remained alive at 74 months and the other patient expired at 8 months [49]. In another study by the same authors, 6 patients underwent surgical treatment. Four of these were also treated with chemotherapy and 2 received chemo-radiation. On average, these patients survived 12.5 months after diagnosis [50]. It was proposed that surgery may increase survival in this subset of patients [48, 49].
If multiple cutaneous lesions or internal metastases exist, chemotherapy is the primary option [48]. During chemotherapy, these lesions can be used to monitor the response of the entire malignancy [10]. Unfortunately, studies reveal survival rates of only 6.5 to 8 months after chemotherapy alone [8, 9, 11]. This may partially be due to low perfusion of the skin [24]. Specific agents used include cisplatin, cyclophosphamide, adriamycin, mitomycin, interferon-B, etoposide, vindesine, and carboplatin [8]. Nevertheless, Kamble et al used cisplatin and VP-16 or adriamycin, cyclophosphamide, and vincristine regimens without adequate responses in any patients [24].
Radiation has been used alone and or in combination with chemotherapy, and/or surgery. In a study by Coslett et al., 1 patient survived just 2 months after diagnosis of the cutaneous metastasis with chest radiation alone and 3 other patients survived a mean of 1.8 months with chemoradiation [9]. Hidaka et al irradiated the skin lesion in 2 patients with 1 patient surviving 5 months and the other surviving 1 year and 7 months [8]. Thus, radiation is usually not effective except in rare cases. However, irradiation of the metastatic lesion may be palliative if the lesion is painful or bleeding [24].
Prognosis
Cutaneous metastases and their primaries in the lung are usually incurable and suggest an unfortunate prognosis [6]. Poor prognostic indicators include non-resectable or small-cell primary tumors, multiple cutaneous metastases, or other distant metastases [50]. Unfortunately, skin metastases usually present with other internal metastases [8, 9, 11]. Patients initially presenting with cutaneous metastases live approximately 3-4 months less than patients who develop a skin metastasis later in their disease process [8, 11, 31]. Mean survival is usually about 5-6 months after the diagnosis of a cutaneous metastasis, although some patients may live longer than a year [8, 9, 11].
Conclusion
Cutaneous metastases from the lung, although uncommon, are a very real problem in our patients. In men and women with suspicious skin lesions, especially in those who have a smoking or lung cancer history, cutaneous metastasis from the lung should be in the differential diagnosis. They most often present as nodules on the chest wall, abdomen, or head/neck, but they may also present as many other forms and on any cutaneous surface. The metastases are usually moderately-to poorly-differentiated adenocarcinomas, squamous-cell carcinomas, small-cell carcinomas, or large-cell carcinomas. The diagnosis can frequently be made using clinical information and histology from a skin biopsy, but may require immunohistochemistry and/or electron microscopy. Unfortunately, cutaneous metastases from the lung frequently indicate a very poor prognosis. However, treatment should not be withheld given documented cases of prolonged survival with treatment.
References
1. Rolz-Cruz G, Kim CC. Tumor invasion of the skin. Dermatol Clin. 2008 Jan; 26:89-102, viii. [PubMed]2. Goljan EF. Rapid Review Pathology. 2nd ed. St. Louis: Mosby; 2006. 321-4.
3. Rosen T. Cutaneous Metastases. Med Clin North Am. 1980 Sep; 65: 885-900. [PubMed]
4. Brownstein MH, Helwig EB. Metastatic tumors of skin. Cancer. 1972 May; 29:1298-1307. [PubMed]
5. Connor DH, Taylor HB, Helwig EB. Cutaneous metastases of renal cell carcinoma. Arch Pathol. 1963 Sep; 76:339-46. [PubMed]
6. Lookingbill DP, Spangler N, Helm KF. Cutaneous metastases in patients with metastatic carcinoma: a retrospective study of 4020 patients. J Am Acad Dermatol. 1993 Aug; 29:228-36. [PubMed]
7. Ask-Upmark E. Clinical aspects of tumor metastases. Nord Med. 1956 Oct 4; 56(40):1433-40. [PubMed]
8. Hidaka T, Ishii Y, Kitamura S. Clinical features of skin metastases from lung cancer. Intern Med. 1996; 35:459-62. [PubMed]
9. Coslett LM, Katlic MR. Lung cancer with skin metastasis. Chest. 1990; 97: 757-9. [PubMed]
10. Dreizen S, Dhingra H, Chiuten D, Umsawasdi T, Valdivieso M. Cutaneous and subcutaneous metastases of lung cancer. Postgrad Med. 1986 Dec; 80: 111-6. [PubMed]
11. Terashima T, Kanaqawa M. Lung cancer with skin metastasis. Chest. 1994 Nov; 106: 1448-50. [PubMed]
12. Lookingbill DP, Spangler N, Sexton FM. Skin involvement as the presenting sign of internal carcinoma. A retrospective study of 7316 cancer patients. J Am Acad Dermatol. 1990 Jan; 22: 19-26. [PubMed]
13. Marcoval J, Moreno A, Peyrí J. Cutaneous infiltration by cancer. J Am Acad Dermatol. 2007 Oct; 57:577-80. Epub 2007 Mar 26. [PubMed]
14. Perng DW, Chen CH, Lee YC, Perng RP. Cutaneous metastases of lung cancer: an ominous prognostic sign. Chin Med J. 1996; 57:343-7. [PubMed]
15. Brownstein MH, Helwig EB. Patterns of cutaneous metastasis. Arch Dermatol. 1972 Jun; 105:862-8. [PubMed]
16. Neel V, Sober A. Metastatic tumors to the skin. In: Kufe D, Bast R, Frei E, Holland J, Gansler T, Pollock R, Weichselbaum R, editors. Cancer Medicine. Danbury: B. C. Decker Incorporated; 2003. Section 32, Subsection 123.
17. Chiu CS, Lin CY, Kuo TT, Kuan YZ, Chen MJ, Ho HC, Yang LC, Chen CH, Shih IH, Hong HS, Chuang YH. Malignant cutaneous tumors of the scalp: a study of demographic characteristics and histologic distributions of 398 Taiwanese patients. J Am Acad Dermatol. 2007 Mar; 56:448-52. Epub 2006 Dec 1. [PubMed]
18. Rubinstein R, Baredes S, Caputo J, Galati L, Schwartz R. Cutaneous metastatic lung cancer: Literature review and report of a tumor on the nose from a large cell undifferentiated carcinoma. Ear Nose Throat J. 2000 Feb; 79:96-7, 100-1. [PubMed]
19. Peris K, Cerroni L, Paoloni M, Margiotta V, Chimenti S. Gingival metastasis as first sign of an undifferentiated carcinoma of the lung. J Dermatol Surg Oncol. 1994 Jun; 20: 407-9. [PubMed]
20. Balakrishnan C, Noorily MJ, Prasa JK, Wilson RF. Metastatic adenocarcinoma in a recent burn scar. Burns. 1994 Aug; 20; 371-2. [PubMed]
21. Garcia-Arpa M, Rodriguez-Vazquez M, Sanchez-Caminero P, Delgado M, Vera E, Romero G, Cortina P. Digital acrometastasis. Actas Dermosifiliogr. 2006 Jun; 97:334-6. [PubMed]
22. Nakamura H, Shimizu T, Kodama K, Shimizu H. Metastasis of lung cancer to the finger: a report of two cases. Int J Dermatol. 2005 Jan; 44:47-9. [PubMed]
23. Weitzner S. Cutaneous metastasis confined to the scrotum: report of two cases. Rocky Mt Med J. 1970; 67:40-2. [PubMed]
24. Kamble R, Kumar L, Kochupillai V, Sharma A, Sandoo MS, Mohanti BK. Cutaneous metastases of lung cancer. Postgrad Med J. 1995; 71: 741-3. [PubMed]
25. Ahmed I. Cutaneous Metastases. In: Bolognia J, Jorrizo J, Horn T, Mancini A, Mascaro J, Rapini R, Salasche S, Saurat J, Stingl G, editors. Dermatology. Elsevier Limited; 2003. 1953-1956.
26. Hazelrigg DE, Rudolph AH. Inflammatory metastatic carcinoma. Arch Dermatol. 1977 Jan; 113:69-70. [PubMed]
27. Matarasso SL, Rosen T. Zosteriform metastasis: case presentation and review of the literature. J Dermatol Surg Oncol. 1988 Jul; 14:774-8. [PubMed]
28. Homler HJ, Goetz CS, Weisenburger DD. Lymphangitic cutaneous metastases from lung cancer mimicking cellulitis. Carcinoma Erysipeloides. West J Med. 1986 May; 144:610-2. [PubMed]
29. Brownstein MH, Helwig EB. Spread of tumors to the skin. Arch Dermatol. 1973 Jan; 107:80-6. [PubMed]
30. Kikuchi Y, Matsuyama A, Nomura K. Zosteriform metastatic skin cancer: report of three cases and review of the literature. Dermatology. 2001; 202: 336-8. [PubMed]
31. Saeed S, Keehn CA, Morgan MB. Cutaneous metastasis: a clinical, pathological, and immunohistochemical appraisal. J Cutan Pathol. 2004 July; 31:419-30. [PubMed]
32. Schwartz RA. Histopathologic aspects of cutaneous metastatic disease. J Am Acad Dermatol. 1995 Oct; 33:649-57. [PubMed]
33. Yanagawa H, Hashimoto Y, Bando H, Takishita Y, Nagano T. Intravascular bronchioloalveolar tumor with skin metastases. Chest. 1994 Jun; 105: 1882-4. [PubMed]
34. Archer CB, MacDonald DM. Bronchial carcinoid presenting with cutaneous metastases. J R Soc Med. 1984; 77 Suppl 4:33-4. [PubMed]
35. Dutt PL, Baxter JW, O'Malley FP, Glick AD, Page DL. Distant cutaneous metastasis of pleural malignant mesothelioma. J Cutan Pathol. 1992 Dec; 19:490-5. [PubMed]
36. Gaudy-Marqueste C, Dales JP, Collet-Villette AM, Grob JJ, Astoul P, Richard MA. Cutaneous metastasis of pleural mesothelioma: two cases. Ann Dermatol Venereol. 2003 Apr; 130: 455-9. [PubMed]
37. Chao SC, Lee JY. Well-differentiated fetal adenocarcinoma presenting with cutaneous metastases. Br J Dermatol. 2004 Apr; 150:778-80. [PubMed]
38. Al-Shraim M, Mahboub B, Neligan PC, Chamberlain D, Ghazarian D. Primary pleural epithelioid haemangioendothelioma with metastases to the skin. A case report and literature review. J Clin Pathol. 2005 Jan; 58:107-9. [PubMed]
39. Fox JL, Berman B, Prioleau PG. Skin metastases from small-cell carcinoma of the lung. J Dermatol Surg Oncol. 1983 Jun; 9(6):451-4. [PubMed]
40. Schwartz R. Cutaneous metastatic disease. J Am Acad Dermatol. 1995 Aug; 33:161-82. [PubMed]
41. Jerome Marson V, Mazieres J, Groussard O, Garcia O, Berjaud J, Dahan M, Carles P, Daste G. Expression of TTF-1 and cytokeratins in primary and secondary epithelial lung tumours: correlation with histological type and grade. Histopathology. 2004 Aug;45(2):125-34. [PubMed]
42. Azoulay S, Adem C, Pelletier F, Barete S, Frances C, Capron, F. Skin metastases from unknown origin: role of immunohistochemistry in the evaluation of cutaneous metastases of carcinoma of unknown origin. J Cutan Pathol. 2005 Sep; 32: 561-6. [PubMed]
43. Koca R, Ustundag Y, Kargi E, Numanoglu G, Altinyazar HC. A case with widespread cutaneous metastases of unknown primary origin: Grave prognostic finding in cancer. Dermatol Online J. 2005 Mar 1; 11(1):16. [PubMed]
44. Rosai J. Special techniques in surgical pathology. In: Rosai and Ackerman's Surgical Pathology. Ninth edn., Vol. 1. Mosby, Philadelphia, 2004, p. 37-91.
45. Sariya D, Ruth K, Adams-McDonnell R, Cusack C, Xu X, Elenitsas R, Seykora J, Pasha T, Zhang P, Baldassano M, Lessin SR, Wu H. Clinicopathologic correlation of cutaneous metastases: experience from a cancer center. Arch Dermatol. 2007 May;143(5):613-20. [PubMed]
46. Tot T. Cytokeratins 20 and 7 as biomarkers: usefulness in discriminating primary from metastatic adenocarcinoma. Eur J Cancer. 2002 Apr;38(6):758-63. [PubMed]
47. Helm K, Lookingbill DP. Cancers Metastatic to the Skin. In: Miller S, Maloney M, editors. Cutaneous oncology. Blackwell Science; 1998. 964-971.
48. Molina Garrido MJ, Guillén Ponce C, Soto Martínez JL, Martínez Y Sevila C, Carrato Mena A. Cutaneous metastases of lung cancer. Clin Transl Oncol. 2006 May; 8:330-3. [PubMed]
49. Ambrogi V, Tonini G, Mineo TC. Prolonged survival after extracranial metastasectomy from synchronous resectable lung cancer. Ann Surg Oncol. 2001 Sep; 8:663-6. [PubMed]
50. Ambrogi V, Nofroni I, Tonini G, Mineo TC. Skin metastases in lung cancer: analysis of a 10-year experience. Oncol Rep. 2001 Jan-Feb; 8(1): 57-61. [PubMed]
© 2009 Dermatology Online Journal

