Persistent erythematous plaque after minor trauma in an immunocompromised woman
Published Web Location
https://doi.org/10.5070/D39dx5j4txMain Content
Persistent erythematous plaque after minor trauma in an immunocompromised woman
Rana Mays MD, Rachel Gordon MD, Janice M Wilson BS, Whitney J LaPolla MD, Karan K Sra MD, Vandana Madkan MD, Stephen K Tyring
MD PhD
Dermatology Online Journal 18 (4): 2
Center for Clinical Studies, Webster, TexasAbstract
Scedosporium apiospermum is a ubiquitous soil fungus with a worldwide distribution. It can cause a wide range of clinical disease, from cutaneous and subcutaneous infections, to pneumonia, brain abscess, and life threatening systemic illness. The diagnosis of cutaneous disease is with biopsy and culture. We discuss the case of an elderly immunocompromised woman who presented with a persistent erythematous plaque on the elbow after minor trauma. A biopsy revealed Scedosporium apiospermum. Treatment usually requires surgical resection in conjunction with antifungal therapy.
Case presentation
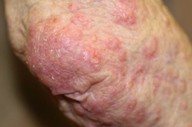
An 80-year-old woman presented with “red bumps” on the right elbow. This condition had worsened in the past three weeks. The patient had scraped her elbow during a mild fall 6 months prior. She presented approximately 2 weeks later with complaints of scaly red nodules and associated tenderness (Figure 1). Past medical history was significant for rheumatoid arthritis, for which she had been taking abatacept (Orencia®) for approximately 4 months. Other medical history included hypertension, hypothyroidism, hyperlipidemia, coronary artery disease, and osteoporosis. After the initial onset of the skin lesions, the patient’s abatacept was stopped and she was placed on oral prednisone 10 mg daily. No other new medications or changes to prior medications were noted.
 |  |
| Figure 1 | Figure 2 |
|---|---|
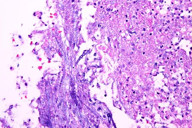
| Figure 1. Erythematous scaly plaque with satellite papules on the posterior right elbow Figure 2. High power view (x40) showing fungal elements in the inflammatory debris. | |
Physical examination revealed an otherwise healthy-appearing elderly female in no apparent distress. She was afebrile, with normal vital signs. Examination of the right elbow revealed multiple 2 mm x 2 mm subcutaneous nodules and minute pustules on an erythematous base. A 4 mm punch biopsy was performed. Laboratory evaluation was notable for a mildly elevated white blood cell count (11 thousand/uL, normal = 3.8-10.8 thousand/uL) and absolute neutrophil count (8008 cells/uL, normal = 1500-7800 cells/uL). Sodium was slightly low (132 mm/L, normal = 135-146 mmols/L) and AST was mildly elevated (39 U/L, normal = 10-35 U/L).
Histopathology of the skin biopsy revealed areas of suppurative granulomatous inflammatory reaction in the dermis with abundant fungus on routine staining (Figure 2). Septate hyphae were relatively localized to the inflammatory process. It was noted that these findings could be consistent with Aspergillus.
Two weeks later, repeat physical examination was notable for worsening of the erythematous papules and emergence of satellite pustules. Shotty right axillary lymphadenopathy was also noted. Repeat complete blood cell count demonstrated worsening leukocytosis and a left shift.
At that time, the patient was started on terbinafine 250 mg once daily for 30 days and a repeat biopsy was obtained and sent for aerobic and fungal cultures. The skin lesions and axillary lymphadenopathy slowly improved with treatment; the patient was continued on another 30 days of terbinafine for a total of 60 days of treatment. One month later, despite the improvement in the size and erythema of the lesions, residual pink plaques with satellite clusters remained on the right elbow. A 3 mm punch biopsy was performed and repeat pathology confirmed continued evidence of fungal hyphae. Cultures of the specimen grew Scedosporium apiospermum. After completion of the terbinafine, the patient underwent surgical debridement of the remaining lesions and responded well. The remaining skin lesions resolved post-operatively.
Discussion
Scedosporium apiospermum (formerly Monosporium apiospermum) is the anamorph or asexual form of Pseudallescheria boydii (formerly Allescheria boydii and Petriellidium boydii), together comprising two forms of a filamentous mold that is ubiquitous worldwide in soil and water. Environmental isolations have been procured from sewage sludge, polluted streams, and manure of poultry and cattle [1, 2]. The fungus was first described as a causative agent of pale-grain eumycetoma (maduromycetoma or Madura foot), a chronic cutaneous and subcutaneous infection endemic to tropical areas [1].
Scedosporium is mainly known for subcutaneous infections following minor trauma to the skin [3]. The organism can also colonize the external ear canals, paranasal sinuses, and bronchi [2, 4]. Recently, more invasive forms of Scedosporium have emerged; it is becoming recognized as a potent etiology of severe infection in immunocompromised individuals [2]. Mycetoma is one of the classic manifestations of Scedosporium apiospermum. It begins as a small, hard, painless nodule that softens with time and may ulcerate and drain. Another manifestation is mycotic keratitis and non-mycetoma-like cutaneous and subcutaneous infections [5]. A review of the literature revealed 65 such cases, with presentations varying from ecchymoses, plaques, pustules, and bullae to lymphocutaneous syndromes in sporotrichoid patterns. The most frequently reported dermatologic manifestations were subcutaneous nodules and ulcers; the most common sites were upper and lower extremities. Soft tissue and wound infections were also reported. Infection most often resulted from direct inoculation; frequently, but not always, a history of trauma to the infected area was given [4].
On direct microscopy and staining with 20 percent KOH, the septated hyphae are indistinguishable from Aspergillus and Fusarium. Clinically, Scedosporium can also cause cavitary lesions very similar to Aspergillus. On histopathology the Scedosporium hyphae show slightly more irregular branching at acute angles. Culture of the specimen is recommended as the standard for diagnosis. Colonies are fast growing and grey-white in color [2].
Adults receiving treatment with corticosteroids and immunosuppressive therapy have a higher incidence of invasive disease. Those particularly affected include post organ transplantation patients and patients with leukemia, lymphoma, systemic lupus erythematous, and rheumatoid arthritis, as in the case presented here [2, 6]. The severity of invasive disease can range from mild. such as sinusitis and pneumonia, to more severe, such as osteomyelitis, keratitis, meningitis, brain abscesses, endophthalmitis, endocarditis, and disseminated systemic disease [2, 4, 7, 8].
Scedosporium apiospermum has been reported to have very high levels of anti-fungal resistance, most notoriously to amphotericin B [7]. Treatment of localized infections involves surgical resection of the tissue if possible, with accompanying antifungal therapy. In numerous reports, the combination of surgical excision with antifungal therapy was superior to antifungal therapy alone [7]. Recommendations resulting from in vitro studies and case reports differ, although many reports indicate voriconazole may be a particularly effective treatment, along with most other azoles. In one study, 11 of 21 patients with cutaneous and subcutaneous infections responded to itraconazole, although treatment failed in 3 cases [2, 6, 8, 9, 10]. Of these three cases, all resolved with voriconazole. For life threatening invasive disease such as systemic illness or brain abscess the use of miconazole and voriconazole have been reported [11, 12]. Additionally, combinations of azoles and terbinafine or amphotericin B have shown to be synergistically effective in vitro [4].
Conclusion
Scedosporium apiospermum is an endemic soil fungus that usually infects cutaneous and subcutaneous tissue after minor trauma or injury. It can clinically present as erythematous plaques, nodules, and pustules that can then form mycetomas. In immunocompromised individuals, Scedosporium apiospermum can manifest as osteomyelitis, pneumonia, brain abscess, endocarditis, and systemic disease. Diagnosis is made by culture of the clinical specimen and sometimes, histopathological studies. Treatment for subcutaneous and localized infection usually involves surgical resection and/or either itracolnizole or voriconazole. In immunocompromised patients with more invasive or systemic disease the use of miconazole or voriconazole has demonstrated good outcomes [2, 12].
References
1. de Hoog GS, Marvin-Sikkema FD, Lahpoor GA et al. Ecology and physiology of the emerging opportunistic fungi Pseudallescheria boydii and Scedosporium prolificans. Mycoses. 1994; 37: 71-8. [PubMed]2. Guarro J, Serda A, Kantarcioglu R et al. Scedosporium apiospermum: changing spectrum of a therapy-refractory opportunist. Medical Mycol 2006; 44: 295-327. [PubMed]
3. McGinnis MR. Mycetoma. Dermatol Clin 1996; 14: 97-104. [PubMed]
4. Cortez KJ, Roilides E, Quiroz-Telles F et al. Infections caused by Scedosporium spp. Clin Microbiol Rev 2008; 21: 157-97. [PubMed]
5. Rippon JW. Medical Mycology: The pathogenic fungi and the pathogenic actinomycetes. 3rd ed. Philadelphia: WB Saunders, 1998.
6. Husain S, Muñoz P, Forrest G, Alexander BD et al. Infections due to Scedosporium apiospermum and Scedosporium prolificans in transplant recipients: clinical characteristics and impact of antifungal agent therapy on outcome. Clin Infect Dis 2005; 40: 89-99. [PubMed]
7. Castiglioni B, Sutton DA, Rinaldi MG et al. Pseudallescheria boydii (Anamorph Scedosporium apiospermum). Infection in solid organ transplant recipients in a tertiary medical center and review of the literature. Medicine (Baltimore) 2002; 81: 333-48. [PubMed]
8. Lemerle E, Bastien M, Demolliens-Dreux G et al. Cutaneous Scedosporium infection revealed by bullous-necrotic purpura. Ann Dermatol Venereol 1998; 125:711-4. [PubMed]
9. Liu YF, Zhao XD, Ma CL, Li CX et al. Cutaneous infection by Scedosporium apiospermum and its successful treatment with itraconazole. Clin Exp Dermatol 1997; 22: 198-200. [PubMed]
10. Severo LC, Oliveira Fde M, Londero AT. Subcutaneous scedosporiosis. Report of two cases and review of the literature. Rev Inst Med Trop Sao Paulo, 1997; 39: 227-30. [PubMed]
11. Schiess RJ, Coscia MF, McClellan GA. Petriellidium boydii pachymeningitis treated with miconazole and ketoconazole. Neurosurgery 1984; 14: 220-4. [PubMed]
12. Steinbach WJ, Perfect JR. Scedosporium species infections and treatments. J Chemother 2003; 15 Suppl 2: 16-27. [PubMed]
© 2012 Dermatology Online Journal

