Pyogenic granuloma-like Kaposi sarcoma: A diagnostic pitfall
Published Web Location
https://doi.org/10.5070/D396z7v7hwMain Content
Pyogenic granuloma-like Kaposi sarcoma: A diagnostic pitfall
Pauline L Scott MD1, Kiran Motaparthi MD1, Bhuvaneswari Krishnan MBBS2, Sylvia Hsu MD1
Dermatology Online Journal 18 (3): 4
Departments of Dermatology1 and Pathology2, Baylor College of Medicine, Houston, TexasAbstract
Pyogenic granuloma-like Kaposi sarcoma (PG-like KS) is a clinicopathologic variant of Kaposi sarcoma (KS), a vascular tumor caused by human herpesvirus-8 (HHV-8). PG-like KS is a challenging entity to diagnose because its clinical and histological features encompass both pyogenic granuloma (PG) and KS characteristics. Immunhistochemical staining with HHV-8 latent nuclear antigen-1 (LNA-1) has been shown to exhibit high sensitivity and specificity for diagnosing KS. Therefore, the integration of clinical features and context, histopathogical findings, and immunohistochemical analysis is important in obtaining the correct diagnosis of PG-like KS. We report a case of PG-like KS in an HIV-positive man.
Introduction
PG-like KS is a variant of KS that has been infrequently reported in both HIV- positive and HIV-negative patients [1, 2, 3]. PG-like KS can exhibit clinical and histologic features of both pyogenic granuloma (PG) and KS [4]. It is a vascular neoplasm caused by human herpesvirus-8 (HHV-8), also referred to as Kaposi sarcoma-associated herpesvirus (KSHV), which is detectable in more than 95 percent of lesions [5, 6]. We report a case of PG-like KS of the foot in a patient with a history of HIV/AIDS.
Case report
 |  |
| Figure 1 | Figure 2 |
|---|---|
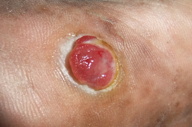
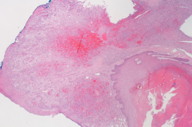
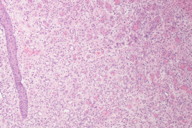
| Figure 1. A 2.5 cm x 2 cm circumscribed, friable, exophytic nodule on the left plantar foot Figure 2. Low power view: exophytic nodule with epidermal collarette and hemorrhage | |
 |  |
| Figure 3 | Figure 4 |
|---|---|
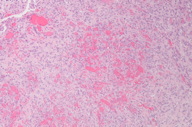
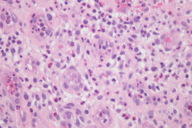
| Figure 3. Low power view: lobular proliferation of capillary-sized vessels Figure 4. High power view: lobular proliferation of capillary-sized vessels with neutrophil-dense infiltrate | |
A 61-year-old man presented with a 5-month history of a protuberant growth on his left foot. With the exception of recurrent bleeding with minimal trauma, he was asymptomatic. Past medical history was significant for HIV/AIDS with a CD4 count of 92. Physical examination disclosed a 2 cm x 2.5 cm non-tender, friable, exophytic nodule on his left plantar foot (Figure 1). The clinical findings were suggestive of a PG, but given the history of HIV/AIDS, KS was suspected.
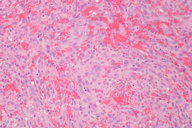
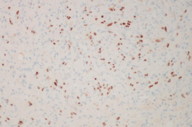
A shave biopsy was then performed for histological and immunohistochemical evaluation to assist in establishing the correct diagnosis. Histological examination revealed an exophytic nodule (Figure 2) composed of a lobular proliferation of capillary-sized vessels and a neutrophil-dense infiltrate (Figures 3 and 4) consistent with features of PG, but also demonstrated a spindle cell neoplasm with abundant irregular vascular spaces and hemorrhage, suggestive of KS (Figures 5 and 6). Immunohistochemistry with latent nuclear antigen-1 (LNA-1), a monoclonal antibody to HHV-8, exhibited diffuse nuclear staining (Figures 7 and 8). Based on the clinicopathologic correlation and supportive immunohistochemistry, the diagnosis of KS was confirmed.
 |  |
| Figure 7 | Figure 8 |
|---|---|
| Figure 7. Low power view: diffuse nuclear staining with LNA-1 Figure 8. High power view: diffuse nuclear staining with LNA-1 | |
Discussion
KS is a low-grade vascular malignancy that has been categorized into 4 epidemiological forms: 1) classic KS, most often affecting elderly men of Middle Eastern, Eastern European, and Mediterranean ancestry; 2) endemic (African) KS; 3) iatrogenic KS in association with immunosuppression; 4) HIV/AIDS-associated KS [7]. Classic KS usually presents with slow-growing bluish red-brown macules/patches that can progress to plaques and nodules. These lesions are usually found on the distal extremities [1]. Endemic (African) KS has many different clinical appearances, including a form that mimics classic KS, a more aggressive disseminated form in adults, and a form found in children with lymph node involvement [7]. Iatrogenic KS is associated with multiple cutaneous lesions on the distal extremities and follows a benign course [7, 8]. HIV/AIDS-associated KS presents with swiftly-developing disseminated disease with oral, skin, lymph node, and visceral involvement [1, 7].
PG-like KS has been reported in patients with both HIV-positive and HIV-negative status [1, 2, 3]. In one report, the PG-like KS was initially mistaken for a PG [1]. In another report, the PG-like KS was found on the hands of 3 patients, a location where PGs are commonly found [3].
The 4 epidemiological KS forms have typically exhibited a histological continuum evolving from patch to plaque and eventually to a nodular stage. Recent observations have led to a new grouping of histologic KS variants: 1) KS variants associated with disease evolution; 2) older literature KS variants; 3) recent KS variants; 4) KS variants related to therapy outcomes [4]. PG-like KS falls into the “recent KS variants” group and can be very difficult to distinguish from PG given overlapping histologic features, such as epidermal collarette resulting from nodular prominence, ulceration and inflammation, and lobular proliferation of capillaries [4].
HHV-8 is the causative agent in all forms of KS and immunohistochemical staining with HHV-8 LNA-1 antibody has been found to be both highly sensitive and specific for KS in several studies [7,9-13]. HHV-8 LNA-1 staining has advantages over the earlier molecular techniques, such as reverse transcriptase polymerase chain reaction (RT-PCR), in that it is cheaper, faster, and more widely available [9]. Consequently, HHV-8 immunostaining is a valuable tool to aid in the diagnosis of KS variants that demonstrate atypical clinical or histologic features. Given that cases of PG simulating KS have been documented prior to the use of HHV-8 immunostaining, these earlier cases could potentially represent PG-like KSs [14].
Immunohistochemical analysis with antibodies to smooth muscle cells (SMA) and Factor VIII (FVIII), commonly seen in PG pericytes and mature endothelial cells, respectively, can also be used to aid in distinguishing between PG and KS. In KS, pericytes and mature endothelial cells are not observed [3]. Endothelial markers, including CD31 and CD34 and the lymphatic marker D2-40 stain spindle cells of nodular KS lesions and can also be helpful [3, 15]. However, simultaneous expression of both PG and KS markers may occur. For example, one reported case of PG-like KS demonstrated expression of SMA, FVIII, CD31, and CD34. HHV-8 immunostaining confirmed the diagnosis [3]. Thus, HHV-8 immunostaining is advisable when components of both KS and PG are present in the same lesion.
In conclusion, KS may mimic PG clinically and histologically, and thus, PG-like KS represents a diagnostic pitfall. Based on the appropriate context, the clinical impression should take precedence over the histology. If the diagnosis of KS is suspected, immunohistochemistry with HHV-8 LNA-1 should be utilized to confirm the correct diagnosis.
References
1. Wyatt ME, Finlayson CJ, Moore-Gillon V. Kaposi’s sarcoma masquerading as pyogenic granuloma of the nasal mucosa. J Laryngol Otol. 1998 Mar;112(3):280-2. [PubMed]2. Ryan P, Aarons S, Murray D, Markham T, Oi’Sullivan S, Lyons F, Lee G, Fitzgibbon J. Human herpesvirus 8 (HHV-8) detected in two patients with Kaposi’s sarcoma-like pyogenic granuloma. J Clin Pathol. 2002 Aug;55(8):619-22. [PubMed]
3. Cabibi D, Cacciatore M, Viviano E, Guarnotta C, Aragona F. 'Pyogenic granuloma-like Kaposi's sarcoma' on the hands: immunohistochemistry and human herpesvirus-8 detection. J Eur Acad Dermatol Venereol. 2009 May;23(5):587-9. [PubMed]
4. Grayson W, Pantanowitz L. Histological variants of cutaneous Kaposi sarcoma. Diagn Pathol. 2008 Jul;3:31. [PubMed]
5. Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science. 1994 Dec;266(5192):1865-9. [PubMed]
6. Antman K, Chang Y. Kaposii’s sarcoma. N Engl J Med. 2000 Apr;342(14):1027-38. [PubMed]
7. Hbid O, Belloul L, Fajali N, Ismaili N, Duprez R, Tanguy M, Benomar H, Tahri EH, Gessain A, Huerre M. Kaposii’s sarcoma in Morocco: a pathological study with immunostaining for human herpesvirus-8 LNA-1. Pathology. 2005 Aug;37(4):288-95. [PubMed]
8. Jakob L, Metzler G, Chen KM, Garbe C. Non-AIDS associated Kaposii’s sarcoma: clinical features and treatment outcome. PLoS One. 2011 Apr;6(4):e18397. [PubMed]
9. Schwartz EJ, Dorfman RF, Kohler S. Human herpesvirus-8 latent nuclear antigen-1 expression in endemic Kaposi sarcoma: an immunohistochemical study of 16 cases. Am J Surg Pathol. 2003 Dec;27(12):1546-50. [PubMed]
10. Robin YM, Guillou L, Michels JJ, Coindre JM. Human herpesvirus 8 immunostaining: a sensitive and specific method for diagnosing Kaposi sarcoma in paraffin-embedded sections. Am J Clin Pathol. 2004 Mar;121(3):330-4. [PubMed]
11. Urquhart JL, Uzieblo A, Kohler S. Detection of HHV-8 in pyogenic granuloma-like Kaposi sarcoma. Am J Dermatopathol. 2006 Aug;28(4):317-21. [PubMed]
12. Patel RM, Goldblum JR, Hsi ED. Immunohistochemical detection of human herpes virus-8 latent nuclear antigen-1 is useful in the diagnosis of Kaposi sarcoma. Mod Pathol. 2004 Apr;17(4):456-60. [PubMed]
13. Cheuk W, Wong KO, Wong CS, Dinkel JE, Ben-Dor D, Chan JK. Immunostaining for human herpesvirus 8 latent nuclear antigen-1 helps distinguish Kaposi sarcoma from its mimickers. Am J Clin Pathol. 2004 Mar;121(3):335-42. [PubMed]
14. Fukunaga M. Kaposii’s sarcoma-like pyogenic granuloma. Histopathology. 2000 Aug;37(2):192-3. [PubMed]
15. Mesri EA, Cesarman E, Boshoff C. Kaposii’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010 Oct;10(10):707-19. [PubMed]
© 2012 Dermatology Online Journal