Satisfying patient expectations with soft-tissue augmentation
Published Web Location
https://doi.org/10.5070/D394t0r445Main Content
Satisfying patient expectations with soft-tissue augmentation
Stephen H Mandy MD FAAD
Dermatology Online Journal 15 (7): 1
Volunteer Professor of Dermatology, Department of Dermatology and Cutaneous Surgery, University of Miami, Miami Beach, Florida.
steve@mandymd.comAbstract
The popularity of cosmetic procedures for rejuvenating the face has undergone enormous growth over the past few years, such that at least one in four cosmetic procedures involves the use of soft-tissue augmentation. Of note is the trend away from surgical interventions and toward noninvasive cosmetic procedures, which now account for the majority of all cosmetic procedures performed in the United States. Adult patients of all ages are selecting soft-tissue augmentation, either as a precursor to or a substitute for surgery; there is a trend toward the use of injectable devices in younger patients (aged 35 – 50 years). Patients in different age groups have diverse treatment needs ranging from the correction of fine lines and wrinkles in younger patients to volume restoration in older patients. Thus, the treatment needs will dictate the treatment approach, particularly with injectable filler treatments. The aesthetic physician needs to help the patient understand and select the most appropriate rejuvenating treatment based on a variety of factors, specifically, patient age, motivating factors, timing, cosmetic area to be addressed, and desired outcome. This article suggests a series of steps to help determine the most appropriate approach for volume restoration with injectable devices for satisfying patient treatment expectations.
Introduction
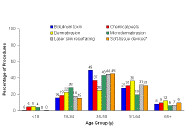 |
| Figure 1 |
|---|
| Figure 1. The cosmetic needs of patients change with age [87]. *Soft-tissue devices include autologous fat, calcium hydroxylapatite, bovine- and human-derived collagen, hyaluronic acid, and poly-L-lactic acid. |
Aesthetic (cosmetic) dermatology has become a fast-growing specialty as a result of an ever-increasing interest in soft-tissue augmentation among increasingly younger patients. According to statistics from the American Society for Aesthetic Plastic Surgery for 2008, 83 percent of all cosmetic procedures performed in the United States were noninvasive. Moreover, 47 percent of nonsurgical procedures in 2008 involved soft-tissue augmentation and botulinum toxin [1]. Injectable dermal fillers used for soft tissue augmentation included hyaluronic acid-based products, collagen-based products, autologous fat, calcium hydroxylapatite, or injectable poly-L-lactic acid (PLLA) [1]. Of particular interest is the trend toward the use of injectable dermal fillers for soft-tissue augmentation among adult patients 35 – 50 years of age [1].
The availability of a variety of nonsurgical treatment options, particularly injectable dermal fillers, enables the aesthetic physician to individualize treatment and maximize overall patient satisfaction. The many benefits of injectable dermal fillers also have facilitated their use in place of, as a precursor to, and in concert with other procedures. These devices, when used alone or in combination with one another, or with other rejuvenating approaches (such as botulinum toxin, radiofrequency tightening, or resurfacing techniques), often provide a longer-lasting correction of contour and volume deficits than does collagen alone [2, 3, 4, 5, 6]. With the introduction of newer approaches for soft-tissue augmentation, such as the injectable dermal fillers, it is possible to provide correction of a larger set of problems in a greater proportion of patients. As the number of patients seeking soft-tissue augmentation increases, it is important to understand what motivates patients of different ages in order to select the appropriate treatment approach to ensure satisfaction.
A primer on patient motivation: Why this? Why now?
The motivation for patients to undergo aesthetic procedures and the factors involved in their perceived satisfaction with these procedures are complex. In general terms, patients want to look more rested, younger, or less angry [7]. A 2006 Harris Interactive® survey involving nearly 800 American women aged 35 – 69 years, conducted on behalf of the American Society of Plastic Surgeons (ASPS), found that the primary reasons women consider cosmetic interventions are to (1) look better, (2) feel younger, and (3) increase their confidence [8]. The survey found that women would ideally like to look 13 years younger than their actual age and that 78 percent of the women surveyed expected the intervention to leave them with a natural looking, refreshed appearance [8]. Indeed, patients often state they "would like to look as young as [they] feel."
The ASPS survey supports previous research demonstrating that attractiveness is closely aligned with positive perceptions and rewards. Research has shown that attractiveness is intimately tied to sexual selection, mate choice, and reproductive strategies [9]. Social psychologists also have shown that attractive people are often thought to be nicer, better, healthier, and more intelligent than unattractive people; further, they are perceived as being more qualified for a job, are predicted to be more successful, and are often better compensated [10]. Several studies have suggested that physical attractiveness favorably influences income, the chances of being hired, the interviewer's perception that such candidates have a more appropriate personality for the job, and promotion decisions [11-17]. In summary, these studies support a fundamental scientific rationale for the perception that attractiveness is a means of obtaining greater self-confidence as well as conferring financial and social advantages.
The spectrum of soft-tissue augmentation and patient satisfaction: lessons learned
In my clinical experience, effective management of patient treatment expectations begins with a detailed assessment of the patient's age-related changes and their objectives. The cosmetic dermatology patient population includes an increasingly larger percentage of younger persons – 20 percent of patients receiving a nonsurgical procedure or 28.5 percent of patients receiving a surgical cosmetic procedure are within the ages of 19 and 34 years of age [1]. According to a survey conducted by the American Society for Aesthetic Plastic Surgery, younger patients tend to favor procedures such as lip augmentation, whereas older patients tend to favor facelifts and the use of injectable dermal fillers (ie., the hyaluronic acids, calcium hydroxylapatite, collagen, and injectable PLLA) [1]. However, there is an additional group of younger patients who wish to correct a physical defect, such as facial asymmetry or atrophy associated with a disease process (i.e., scleroderma, Parry-Romberg syndrome, acne scars).
In contrast, older patients often seek treatment for deeper wrinkles, folds or furrows, or for increased skin laxity due to facial volume loss that is typical of biologic aging. Over the past few decades, we have developed a far more comprehensive understanding of the structural changes of the aging face, particularly with regard to the importance of volume loss. Age-associated facial volume loss is a combination of fat atrophy, bone resorption, and tissue laxity – but not gravity [18, 19, 20, 21]. Recent studies also have shown that the distinct anatomic compartments of subcutaneous fat in the face age differently and that facial skeletal mass decreases with age, leading to a reduction in structural support for the overlying tissues [21, 22, 23, 24]. All of these changes may contribute to a state of "deflation" of the face [18] and resultant skin redundancy. A number of other factors also may be involved in facial aging [19], including smoking tobacco and extensive past exposure to the sun (photoaging). Thus, facial aging is a multifactoral process that may require more than one treatment approach in order to obtain optimal results. Consequently, patients need to understand what volume depletion is and why correction involves a specific treatment approach. Skin quality may be an issue for patients at any age.
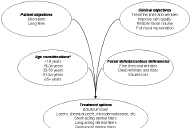 |
| Figure 2 |
|---|
| Figure 2. Choosing an appropriate treatment option requires effective assessment of several factors, beginning with a detailed assessment of the patient's treatment needs and knowledge of the patient's treatment objective. *Age groupings are based on those reported by the American Society for Aesthetic Plastic Surgery [87]. |
Determining patient objectives
It is important for cosmetic physicians to appreciate what is important to the patient. The patient's time frame for the desired correction also is an important determining factor in the treatment decision process [25]. Is the patient attending a class reunion or a child's wedding in the near future and seeking an intervention that provides an immediate result – even if it has a short duration of effect? Or is the patient interested in treatments that afford a more gradual yet longer-lasting effect? Is the patient interested in correcting one specific facial area (such as the forehead or nasolabial folds) or does the patient require full soft-tissue augmentation due to age-related volume loss? More extensive restoration necessitates the use of long-acting devices that also alleviate volume loss, but requires additional time. It may also entail the use of combination treatment, affording the patient both immediate and long-term rejuvenating effects [26].
Matching treatment objectives to available treatments
The aesthetic physician needs to help patients understand that treatment objectives and results are largely based on the characteristics of the specific treatment approach. For example, patients often do not understand volume depletion and volumetric replacement of the nasojugal fold. Without explanation to the patient that it will correct the nasolabial fold will lead the patient to think you did not address their concern. Soft-tissue augmentation approaches are not "one size fits all," rather, each technique, approach, or product has its own ideal use. It is the physician's responsibility to identify which treatment, or combination of treatments, will best attain the desired treatment goal and to understand the limitations of each product.
Chemical peels and lasers improve skin texture quality as well as superficial wrinkles, and can improve skin elasticity; however, these approaches do not replace lost volume [25, 27]. Whereas superficial peels can be used to treat superficial wrinkling and require minimal recovery time, medium-depth and deep peels, which are used to correct more severe photodamage, often require long and uncomfortable recovery periods, and may carry the risk of scarring [28, 29]. Similarly, intramuscular injections of botulinum toxin type A alleviate dynamic (motion-related) lines, wrinkles, and furrows by relaxing the muscles that purportedly cause them but do not replace lost volume. Consequently, botulinum toxin type A or medium-depth and deep peels are not appropriate monotherapy for the treatment of facial volume loss or static rhytides in the mid section of the face [30, 31].
Dermal fillers, such as collagen, hyaluronic acid derivatives, and autologous fat can correct deeper wrinkles, as well as facial volume loss. Collagen-based products have a duration of effect of approximately 4 – 12 months, depending on the source (bovine-, human-, or porcine-derived) and extent of collagen fiber cross-linking provided in each product [32, 33, 34]. The collagens also may require frequent "touch-ups" [32]. Porcine collagen seems to have a lower incidence of adverse events and does not require skin testing whereas testing is necessary with bovine collagen. Hyaluronic acid, a natural component in the extracellular matrix of tissues – such as synovial fluid, connective tissue and skin – is found in all vertebrate animals [35]. In the skin, hyaluronic acid provides structure and volume; the amount of hyaluronic acid decreases with age leading to dehydration and wrinkle formation [36]. Hyaluronic acid is highly hydrophilic and cross-linking its polysaccharide chains slows its degradation [37, 38]. Depending on the degree of cross-linking, several hyaluronic acid-based dermal filler products with varying duration of effect (6 – 12 months) are available [34, 39]. It should be noted that adverse events such as swelling redness and bruising are associated with the use of these products. These events, however, typically abate within approximately 1 week [34, 35, 38, 40]. Autologous fat does not cause an allergic response, has no risk of rejection, and has an effect lasting from several months to several years, although the actual duration of effect can be unpredictable. However, autologous fat requires surgical harvest and may require frequent retreatment of dynamic areas, such as the nasolabial folds and marionette lines [34, 41, 42, 43]. In addition, adequate fat sources may be difficult to obtain in slender patients. The collagens, hyaluronic acids, and autologous fat provide immediate correction of volume and wrinkles.
Long-acting dermal fillers include calcium hydroxylapatite and injectable PLLA, and each is thought to produce their effects by inducing cellular responses that are hypothesized to result in the formation of collagen [44]. Both injectable devices are used for volume restoration. Calcium hydroxylapatite is a gel-based aqueous formulation of synthetic calcium hydroxylapatite microspheres consisting of calcium and phosphate ions, which are the mineral components of bone [44, 45, 46]. Calcium hydroxylapatite is indicated for subdermal implantation for the correction of moderate to severe facial wrinkles and folds, such as nasolabial folds and it is also intended for restoration and/or correction of the signs of facial fat loss (lipoatrophy) in people with human immunodeficiency virus (HIV) [47]. The use of calcium hydroxylapatite for volume restoration is thought to involve a biological response that results in collagen formation [44, 46, 48]. A histological analysis following calcium hydroxylapatite injections into the postauricular area in women revealed no inflammatory response 1 month after the injection. After 6 months, multinucleated giant cells were observed surrounding the calcium hydroxylapatite particles. In addition, the particles also were surrounded by thick collagen [46]. The absence of a cellular response to calcium hydroxylapatite injection also was noted in an earlier histologic study by Lemperle et al [48]. Calcium hydroxylapatite provides an immediate effect, which continues for approximately 12 months [47].
Injectable PLLA contains microparticles of poly-L-lactic acid, which is a biocompatible, biodegradable, synthetic polymer [49]. In the United States, injectable PLLA is intended for restoration and/or correction of the signs of facial fat loss (lipoatrophy) in people with HIV [49]. Studies in rats and mice have shown that implantation of solid PLLA disks and cylinders leads to a tissue response that results in a cascade of cellular events culminating in the formation of collagen [50, 51]. Although systematic histologic analyses of injectable PLLA use in humans have not been reported, recent reports have suggested that a similar tissue response and cellular cascade leading to collagen formation may occur following the administration of injectable PLLA in human subjects [29, 48]. Evidence for this cascade and subsequent collagen formation is suggested by studies in patients with human immunodeficiency virus-related facial lipoatrophy, showing that treatment with injectable PLLA leads to increased dermal thickness that lasts for up to 2 years [6].
The cellular cascade associated with injectable PLLA occurs over time, however, and requires patience on the part of both the treating physician and patient. Consequently, the application of injectable PLLA results in a gradual treatment effect. This also means that a limited correction must be made during each treatment session and that treatments must be spaced at least 4 to 6 weeks prior to the next session to avoid overcorrection. [18, 24, 29, 49, 52, 53, 54]. This interval between treatment sessions is longer than the recommended treatment intervals in the product insert [49]. This approach is best summarized as the "treat to repair, wait to restore, and assess to refine" method [54, 55]. Injection of the PLLA microparticles initiates the cellular cascade; the interval between treatments allows for the restorative biological processes to occur; and carefully assessing the facial contour changes brought on by the previous treatment allows for further refinements of the facial area undergoing treatment. Further, because the volumizing effect of injectable PLLA occurs gradually with a series of injections, the volumetric correction appears natural [29]. It is important to recognize that use of injectable PLLA requires carefully reconstituting the product in accordance with procedure outlined in the prescribing information. Reconstitution involves the addition of 5 mL sterile water for injection (SWFI) followed by allowing the material to stand at least 2 hours to allow for hydration [49]. In clinical practice, many physicians include lidocaine during the reconstitution step and use a higher reconstitution volume (i.e., adding >5 mL of SWFI) and longer hydration times, although these variations are off-label [18, 29, 54, 56, 57].
Thus, the two important distinguishing characteristics between the two long-acting dermal fillers, or collagen stimulators, calcium hydroxylapatite and injectable PLLA are their onset of effect and their duration of effect. Patients requesting long-term (about 12 months) and immediate effect would likely be more satisfied with calcium hydroxylapatite. Conversely, patients who are interested in attaining gradual and natural-looking corrections that may last for up to 2 years may be happier with injectable PLLA. It should be noted that injectable PLLA should not be considered for patients requiring treatment of superficial wrinkles or the periocular and perioral areas. Because of their higher viscosity, neither injectable PLLA nor calcium hydroxylapatite is recommended for lip volumization [58]. Cosmetic physicians today use more than one technique in a given patient. However, the safety of combining such techniques to achieve soft-tissue augmentation, including the use of the long-acting fillers, has not been studied in long-term clinical trials or approved by the US Food and Drug Administration.
Potential adverse effects and aligning patient expectations
Prior to beginning any therapeutic intervention, it is important to clearly communicate to the patient what he or she can expect with each available injectable treatment option. Whereas all injectable devices can cause some degree of postinjection transient bruising and swelling, there also are specific adverse effects associated with each product [59, 60], such as hypersensitivity to bovine-derived collagen [33]. The recently approved porcine collagen has a duration of effect of at least 6 months and most of the antigenic molecules in the product are reported to be removed by enzymatic digestion [32]. In contrast to the hyaluronic acids, collagen injections are less likely to produce bruising and inflammation.
Common adverse events associated with the use of collagens, hyaluronic acid derivatives, calcium hydroxylapatite, or injectable PLLA include swelling, bruising, and erythema, are related to the injection procedure, and resolve within a few days [40, 58, 59, 61, 62]. Because bruising is common with all the injectable devices, use of devices that provide immediate correction such as the hyaluronic acids, collagens, or calcium hydroxylapatite in close proximity to a social event should be planned well enough in advance to avoid such issues.
Nodules and papules, which have been reported for all the injectable devices, can appear immediately or in the weeks to months after the injection [6, 37, 40, 63-70]. For example, in studies in which the reconstitution volume for injectable PLLA totaled less than 5 mL, the incidence of nodules and papules ranged from 13 percent – 44 percent [6, 67, 68, 71, 72, 73]. However, studies using reconstitution volumes of injectable PLLA ≥5 mL have reported nodule/papule incidence rates of 8 percent or less [53, 74, 75, 76]. An incidence rate ranging from 1 percent – 12 percent has been reported for patients treated with calcium hydroxylapatite [5, 65, 77, 78]. Studies with hyaluronic acid-based products have reported a large nodule/papule incidence range of 16 percent – 83 percent [66, 79, 80].
Although the reasons for such a wide range in the incidence of nodules and papules are unclear, one investigator proposed that the formation of nodules or papules may be related to the viscosity of the hyaluronic acid product and/or the volume injected [79]. These complications can usually be avoided by following the proper injection technique associated with the particular injectable device [18, 70]. Although most nodules/papules resolve spontaneously over several months, subcision or excision may be needed to remove visible nodules [58, 70]. Hyaluronic acid nodules can be dissolved with injectable hyaluronidase [38, 58].
The importance of knowing where and how to inject each agent to optimize results and minimize the appearance of adverse effects cannot be overstated. Physicians should undergo training in the specific injection (and postinjection) techniques for each product. In the case of using injectable PLLA or calcium hydroxylapatite, for example, immediate postinjection massage by the injector is recommended [18]. Further, over the following week after treatment with injectable PLLA, it is important for the patient to thoroughly massage the treatment area to evenly distribute the product [49]. In contrast, patient self-massage is not recommended after injection of hyaluronic acid fillers [29, 35, 54]. Massage can be performed immediately following treatment with collagen, to ensure smooth integration into the tissue [33]. In addition, calcium hydroxylapatite is intended for the restoration of specific facial areas, such as the nasolabial folds; injections into other facial areas, such as the lips, or at the incorrect depth may result in an increase in adverse events and should be avoided [40, 58]. For example, injections of calcium hydroxylapatite into the lips have resulted in the development of nodules [34, 81]. Injectable PLLA is also not recommended for use in the lips and special care must be taken when using the device in areas of thin skin, such as the periorbital area [49, 82, 83]. In addition, injectable PLLA should be administered into the deep dermis and superficial injections should be avoided to minimize the occurrence of nodules and papules [49, 82]. Superficial injections and injections into hyperkinetic muscles, such as the perioral area, can lead to complications including the development of nodules and papules with the hyaluronic acids, calcium hydroxylapatite, and injectable PLLA [40, 58, 82, 84]. With hyaluronic acids and calcium hydroxylapatite, superficial injections also may impart a bluish or whitish color to the skin [70]. All injectable fillers should be avoided or used with caution in the glabellar region and the lips as arterial occlusion (from compression or embolization) can result in tissue necrosis and scarring [85, 86].
Treatment approach considerations
Patients of all ages are pursuing cosmetic interventions; it is the physician's responsibility to select the most appropriate intervention(s) based on the patient's age, physical needs and concerns, extent and location of volume loss, and cosmetic goals. In order to optimize patient satisfaction, it is important for the treating physician to understand this variability between patients and to select a treatment approach accordingly.
In general, the selection of the appropriate treatment can easily be matched with the patients' treatment objectives and expectations. Figure 2 provides a graphic representation of the factors to consider when selecting an appropriate treatment approach. What is the patient's treatment objective? What is the nature of the complaint (fine lines or deeper wrinkles or volume loss)? What can be achieved clinically? All dermal fillers except injectable PLLA provide immediate results. Thus, patients of all ages who require immediate wrinkle correction because of a social engagement occurring in the very near future are likely to be best served by dermal fillers that provide an immediate effect. Wrinkles or folds that are the result of facial volume loss, however, require treatment with a dermal filler capable of restoring the lost volume, such as calcium hydroxylapatite and injectable PLLA. Short-acting dermal fillers include the collagens (bovine, human, and porcine) and hyaluronic acid derivatives. When bruising and inflammation are not a concern but immediate results with a short effect are desired, collagens or hyaluronic acid derivatives may be preferred. Long-acting injectable devices, including calcium hydroxylapatite and injectable PLLA, are more suitable for patients who exhibit more pronounced facial volume loss and those who desire long-lasting results.
Conclusion
The use of noninvasive cosmetic procedures has greatly increased in recent years, and injectable devices in particular are now more popular than invasive surgical procedures for soft-tissue augmentation. In all, the availability of several injectable devices for soft-tissue augmentation has led to a wide variety of procedures that the aesthetic physician can utilize to correct the effects of aging. Knowledge of product attributes (i.e., best for superficial line filling, not for long-term correction of volume loss) and limitations (i.e., not for use in the lips) will aid the physician in the careful and appropriate selection of injectable dermal fillers for setting and satisfying realistic treatment expectations for their patients.
Acknowledgment: Editorial support for this article was provided by the editorial staff at Embryon. This article was funded by Dermik Laboratories, a business of sanofi-aventis U.S. The author gratefully acknowledges Lynne Schneider, PhD, who assisted in the preparation of this article based on author-provided comments. The opinions expressed in the current article are those of the author. The author received no honoraria or other form of financial support related to the development of this manuscript.
References
1. 2008 Cosmetic Surgery National Data Bank Statistics. The American Society for Aesthetic Plastic Surgery; 2009.2. Baumann L, Replacing dermal constituents lost through aging with dermal fillers. Semin Cutan Med Surg. 2004; 23:160-166. [PubMed]
3. Lindqvist C, Tveten S, Bondevik BE et al., A randomized, evaluator-blind, multicenter comparison of the efficacy and tolerability of Perlane versus Zyplast in the correction of nasolabial folds. Plast Reconstr Surg. 2005; 115:282-289. [PubMed]
4. Lupo MP, Smith SR, Thomas JA et al., Effectiveness of Juvederm Ultra Plus dermal filler in the treatment of severe nasolabial folds. Plast Reconstr Surg. 2008; 121:289-297. [PubMed]
5. Smith S, Busso M, McClaren M et al., A Randomized, Bilateral, Prospective Comparison of Calcium Hydroxylapatite Microspheres versus Human-Based Collagen for the Correction of Nasolabial Folds. Dermatol Surg. 2007; 33:S112-S121.
6. Valantin MA, Aubron-Olivier C, Ghosn J et al., Polylactic acid implants (New-Fill) to correct facial lipoatrophy in HIV-infected patients: results of the open-label study VEGA. AIDS. 2003; 17:2471-2477. [PubMed]
7. Fagien S, Cox SE, Finn JC et al., Patient-reported outcomes with botulinum toxin type A treatment of glabellar rhytids: a double-blind, randomized, placebo-controlled study. Dermatol Surg. 2007; 33:S2-S9. [PubMed]
8. American Society of Plastic Surgeons. Perception of the injection - American women's perception of cosmetic facial injectables. A Harris Interactive Survey. 2006 Dec.
9. Grammer K, Fink B, Moller AP et al., Darwinian aesthetics: sexual selection and the biology of beauty. Biol Rev Camb Philos Soc. 2003; 78:385-407. [PubMed]
10. Bashour M, History and current concepts in the analysis of facial attractiveness. Plast Reconstr Surg. 2006; 118:741-756. [PubMed]
11. Cam A, Siegfried WD, Pearce L, Forced attention to specific applicant qualifications: Impact on physical attractiveness and sex of applicant biases. Pers Psychol. 1981; 34:65.
12. Dipboye RL, Fromkin HL, Relative importance of applicant sex, attractiveness, and scholastic standing in evaluation of job applicant resumes. J Appl Psychol. 1975; 60:39-43.
13. Gilmore DC, Beehr TA, Love KG, Effects of applicant sex: Applicant physical attractiveness, type of rater and type of job on interview decisions. J Occup Psychol. 1986; 59:103-109.
14. Quereshi MY, Kay JP, Physical attractiveness, age, and sex as determinants of reactions to resumes. Soc Behav Pers. 1986; 14:103.
15. Morrow PC, McElroy JC, Stamper BG, The effects of physical attractiveness and other demographic characteristics an promotion decisions. J Manag. 1990; 16:723-736.
16. Frieze IH, Olson JE, Russell J, Attractiveness and income for men and women in management. J Appl Soc Psychol. 1991; 21:1039-1057.
17. Roszell P, Kennedy DEG, Physical attractiveness and income attainment among Canadians. J Psychol. 2008; 123:547-559.
18. Mandy SH, Fillers that work by fibroplasia: Poly-L-Lactic Acid. In: Carruthers J, Carruthers A, editors. Soft Tissue Augmentation. 2nd ed. Saunders; 2007. p. 101-4
19. Donath AS, Glasgold RA, Glasgold MJ, Volume loss versus gravity: new concepts in facial aging. Curr Opin Otolaryngol Head Neck Surg. 2007; 15:238-243. [PubMed]
20. Donofrio LM, Fat distribution: a morphologic study of the aging face. Dermatol Surg. 2000; 26:1107-1112. [PubMed]
21. Shaw RB, Jr., Kahn DM, Aging of the midface bony elements: a three-dimensional computed tomographic study. Plast Reconstr Surg. 2007; 119:675-681. [PubMed]
22. Rohrich RJ, Pessa JE, The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007; 119:2219-2227. [PubMed]
23. Pessa JE, Zadoo VP, Yuan C et al., Concertina effect and facial aging: nonlinear aspects of youthfulness and skeletal remodeling, and why, perhaps, infants have jowls. Plast Reconstr Surg. 1999; 103:635-644. [PubMed]
24. Vleggaar D, Fitzgerald R, Dermatological implications of skeletal aging: a focus on supraperiosteal volumization for perioral rejuvenation. J Drugs Dermatol. 2008; 7:209-220. [PubMed]
25. Rendon MI, Effron C, Edison BL, The use of fillers and botulinum toxin type A in combination with superficial glycolic acid (alpha-hydroxy acid) peels: optimizing injection therapy with the skin-smoothing properties of peels. Cutis. 2007; 79:9-12. [PubMed]
26. Broder KW, Cohen SR, An overview of permanent and semipermanent fillers. Plast Reconstr Surg. 2006; 118:7S-14S. [PubMed]
27. Goldman MP, Alster TS, Weiss R, A randomized trial to determine the influence of laser therapy, monopolar radiofrequency treatment, and intense pulsed light therapy administered immediately after hyaluronic acid gel implantation. Dermatol Surg. 2007; 33:535-542. [PubMed]
28. Bosniak S, Cantisano-Zilkha M, Purewal BK et al., Combination therapies in oculofacial rejuvenation. Orbit. 2006; 25:319-326. [PubMed]
29. Vleggaar D, Facial volumetric correction with injectable poly-L-lactic acid. Dermatol Surg. 2005; 31:1511-1517. [PubMed]
30. De Boulle KL, Botulinum neurotoxin type A in facial aesthetics. Expert Opin Pharmacother. 2007; 8:1059-1072. [PubMed]
31. Said S, Meshkinpour A, Carruthers A et al., Botulinum toxin A: its expanding role in dermatology and esthetics. Am J Clin Dermatol. 2003; 4:609-616. [PubMed]
32. Narins RS, Brandt FS, Lorenc ZP et al., Twelve-month persistency of a novel ribose-cross-linked collagen dermal filler. Dermatol Surg. 2008; 34 Suppl 1:S31-S39. [PubMed]
33. Baumann L, Kaufman J, Saghari S, Collagen fillers. Dermatol Ther. 2006; 19:134-140. [PubMed]
34. Narins RS, Bowman PH, Injectable skin fillers. Clin Plast Surg. 2005; 32:151-162. [PubMed]
35. Monheit GD, Coleman KM, Hyaluronic acid fillers. Dermatol Ther. 2006; 19:141-150. [PubMed]
36. Carruthers J, Carruthers A, Hyaluronic acid gel in skin rejuvenation. J Drugs Dermatol. 2006; 5:959-964. [PubMed]
37. Narins RS, Brandt F, Leyden J et al., A randomized, double-blind, multicenter comparison of the efficacy and tolerability of Restylane versus Zyplast for the correction of nasolabial folds. Dermatol Surg. 2003; 29:588-595. [PubMed]
38. Lupo MP, Hyaluronic acid fillers in facial rejuvenation. Semin Cutan Med Surg. 2006; 25:122-126. [PubMed]
39. Juvéderm Ultra Plus [package insert], Santa Barbara, CA: Allergan, Inc. 2008.
40. Lowe NJ, Maxwell CA, Patnaik R, Adverse reactions to dermal fillers: review. Dermatol Surg. 2005; 31:1616-1625. [PubMed]
41. Burgess CM, Principles of soft tissue augmentation for the aging face. Clin Interv Aging. 2006; 1:349-355. [PubMed]
42. Kaufman MR, Miller TA, Huang C et al., Autologous fat transfer for facial recontouring: is there science behind the art? Plast Reconstr Surg. 2007; 119:2287-2296. [PubMed]
43. Bucky LP, Kanchwala SK, The role of autologous fat and alternative fillers in the aging face. Plast Reconstr Surg. 2007; 120:89S-97S. [PubMed]
44. Schweiger ES, Riddle CC, Wernli BJ et al., Comparison of poly-l-lactic acid and calcium hydroxylapatite for treating human immunodeficiency virus-associated facial lipoatrophy. Cosmet Dermatol. 2007; 20:304-312.
45. Johl SS, Burgett RA, Dermal filler agents: a practical review. Curr Opin Ophthalmol. 2006; 17:471-479. [PubMed]
46. Marmur ES, Phelps R, Goldberg DJ, Clinical, histologic and electron microscopic findings after injection of a calcium hydroxylapatite filler. J Cosmet Laser Ther. 2004; 6:223-226. [PubMed]
47. Radiesse Injectable implant [package insert], Franksville, WI: BioForm Medical Inc. 2006.
48. Lemperle G, Morhenn V, Charrier U, Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthetic Plast Surg. 2003; 27:354-366. [PubMed]
49. Sculptra [prescribing information], Bridgewater, NJ: Dermik Laboratories; a business of sanofi-aventis U.S. LLC. 2006.
50. Bos RRM, Rozema FR, Boering G et al., Degradation of and tissue reaction to biodegradable poly(L-lactide) for use as internal fixation of fractures: a study in rats. Biomaterials. 1991; 12:32-36. [PubMed]
51. Gogolewski S, Jovanovic M, Perren SM et al., Tissue response and in vivo degradation of selected polyhydroxyacids: polylactides (PLA), poly(3-hydroxybutyrate) (PHB), and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHB/VA). J Biomed Mater Res. 1993; 27:1135-1148. [PubMed]
52. Sadick NS, Poly-l-lactic acid: a perspective from my practice. J Cosmet Dermatol. 2008; 7:55-60. [PubMed]
53. Woerle B, Hanke CW, Sattler G, Poly-L-lactic acid: a temporary filler for soft tissue augmentation. J Drugs Dermatol. 2004; 3:385-389. [PubMed]
54. Sherman RN, Sculptra: the new three-dimensional filler. Clin Plast Surg. 2006; 33:539-550. [PubMed]
55. Lowe NJ, Optimizing poly-l-lactic acid use. J Cosmet Laser Ther. 2008; 10:43-46. [PubMed]
56. Vleggaar D, Bauer U, Facial enhancement and the European experience with Sculptra (poly-l-lactic acid). J Drugs Dermatol. 2004; 3:542-547. [PubMed]
57. Lam SM, Azizzadeh B, Graivier M, Injectable poly-L-lactic acid (Sculptra): technical considerations in soft-tissue contouring. Plast Reconstr Surg. 2006; 118:55S-63S. [PubMed]
58. Gladstone HB, Cohen JL, Adverse effects when injecting facial fillers. Semin Cutan Med Surg. 2007; 26:34-39. [PubMed]
59. Andre P, Lowe NJ, Parc A et al., Adverse reactions to dermal fillers: a review of European experiences. J Cosmet Laser Ther. 2005; 7:171-176. [PubMed]
60. Carruthers JD, Carruthers A, Facial sculpting and tissue augmentation. Dermatol Surg. 2005; 31:1604-1612. [PubMed]
61. Christensen L, Breiting V, Janssen M et al., Adverse reactions to injectable soft tissue permanent fillers. Aesthetic Plast Surg. 2005; 29:34-48. [PubMed]
62. Narins RS, Jewell M, Rubin M et al., Clinical conference: management of rare events following dermal fillers--focal necrosis and angry red bumps. Dermatol Surg. 2006; 32:426-434. [PubMed]
63. Tzikas TL, Evaluation of the Radiance FN soft tissue filler for facial soft tissue augmentation. Arch Facial Plast Surg. 2004; 6:234-239. [PubMed]
64. Alam M, Yoo SS, Technique for calcium hydroxylapatite injection for correction of nasolabial fold depressions. Dermatol Surg. 2007; 56:285-289. [PubMed]
65. Jansen DA, Graivier MH, Evaluation of a calcium hydroxylapatite-based implant (Radiesse) for facial soft-tissue augmentation. Plast Reconstr Surg. 2006; 118:22S-30S, discussion. [PubMed]
66. Baumann LS, Shamban AT, Lupo MP et al., Comparison of Smooth-Gel Hyaluronic Acid Dermal Fillers with Cross-linked Bovine Collagen: A Multicenter, Double-Masked, Randomized, Within-Subject Study. Dermatol Surg. 2007; 33:S128-S135.
67. Moyle GJ, Brown S, Lysakova L et al., Long-term safety and efficacy of poly-L-lactic acid in the treatment of HIV-related facial lipoatrophy. HIV Med. 2006; 7:181-185. [PubMed]
68. Mest DR, Humble G, Safety and efficacy of poly-L-lactic acid injections in persons with HIV-associated lipoatrophy: the US experience. Dermatol Surg. 2006; 32:1336-1345. [PubMed]
69. Lemperle G, Rullan PP, Gauthier-Hazan N, Avoiding and treating dermal filler complications. Plast Reconstr Surg. 2006; 118:92S-107S. [PubMed]
70. Duffy DM, Complications of fillers: overview. Dermatol Surg. 2005; 31:1626-1633. [PubMed]
71. Guaraldi G, Orlando G, De FD et al., Comparison of three different interventions for the correction of HIV-associated facial lipoatrophy: a prospective study. Antivir Ther. 2005; 10:753-759. [PubMed]
72. Onesti MG, Renzi LF, Paoletti F et al., Use of polylactic acid in face lipodystrophy in HIV positive patients undergoing treatment with antiretroviral drugs (HAART). Acta Chir Plast. 2004; 46:12-15. [PubMed]
73. Moyle GJ, Lysakova L, Brown S et al., A randomized open-label study of immediate versus delayed polylactic acid injections for the cosmetic management of facial lipoatrophy in persons with HIV infection. HIV Med. 2004; 5:82-87. [PubMed]
74. Burgess CM, Quiroga RM, Assessment of the safety and efficacy of poly-L-lactic acid for the treatment of HIV-associated facial lipoatrophy. J Am Acad Dermatol. 2005; 52:233-239. [PubMed]
75. Hanke CW, Redbord KP, Safety and efficacy of poly-L-lactic acid in HIV lipoatrophy and lipoatrophy of aging. J Drugs Dermatol. 2007; 6:123-128. [PubMed]
76. Mest DR, Humble GM, Retreatment with Injectable Poly-l-Lactic Acid for HIV-Associated Facial Lipoatrophy: 24-Month Extension of the Blue Pacific Study. Dermatol Surg. 2009; 35:350-359.
77. Roy D, Sadick N, Mangat D, Clinical trial of a novel filler material for soft tissue augmentation of the face containing synthetic calcium hydroxylapatite microspheres. Dermatol Surg. 2006; 32:1134-1139. [PubMed]
78. Sadick NS, Katz BE, Roy D, A Multicenter, 47-Month Study of Safety and Efficacy of Calcium Hydroxylapatite for Soft Tissue Augmentation of Nasolabial Folds and Other Areas of the Face. Dermatol Surg. 2007; 33:S122-S127.
79. Bugge H, Negaard A, Skeie L et al., Hyaluronic acid treatment of facial fat atrophy in HIV-positive patients. HIV Med. 2007; 8:475-482. [PubMed]
80. Carruthers A, Carey W, De Lorenzi C et al., Randomized, double-blind comparison of the efficacy of two hyaluronic acid derivatives, restylane perlane and hylaform, in the treatment of nasolabial folds. Dermatol Surg. 2005; 31:1591-1598. [PubMed]
81. Jacovella PF, Calcium hydroxylapatite facial filler (Radiesse): indications, technique, and results. Clin Plast Surg. 2006; 33:511-523. [PubMed]
82. Narins RS, Minimizing adverse events associated with poly-L-lactic acid injection. Dermatol Surg. 2008; 34 Suppl 1:S100-S104. [PubMed]
83. Lowe NJ, Dispelling the myth: appropriate use of poly-L-lactic acid and clinical considerations. J Eur Acad Dermatol Venereol. 2006; 20:2-6. [PubMed]
84. Vleggaar D, Poly-L-lactic acid: consultation on the injection techniques. J Eur Acad Dermatol Venereol. 2006; 20:17-21. [PubMed]
85. Alam M, Gladstone H, Kramer EM et al., ASDS guidelines of care: injectable fillers. Dermatol Surg. 2008; 34 Suppl 1:S115-S148. [PubMed]
86. Coleman SR, Avoidance of arterial occlusion from injection of soft tissue fillers. Aesthetic Surg J. 2002; 22:555-557.
87. American Society for Aesthetic Plastic Surgery. Cosmetic surgery national data bank statistics. 2008.
© 2009 Dermatology Online Journal

