Pigmented spindle-cell nevus: A melanoma simulator
Published Web Location
https://doi.org/10.5070/D37wh9x504Main Content
Pigmented spindle-cell nevus: A melanoma simulator
Andrea Maurichi MD1, Marzia Baldi MD1, Cesare Bartoli MD2, Aldo Bono MD1, Tiziana Camerini PhD3, Daniele Moglia MD1, Roberto Patuzzo MD1, Elisabetta Pennacchioli MD1, Gabrina Tragni MD4, Mario Santinami MD1
Dermatology Online Journal 10 (2): 5
Melanoma Unit1, Day Surgery Unit2, Controlled Clinical Trials Unit3, Pathology and Cytopathology Unit4, Istituto Nazionale per lo Studio e la Cura dei Tumori, Milan, Italy. andrea.maurichi@istitutotumori.mi.it
The clinical differential diagnosis of melanoma presents a formidable challenge even to experienced physicians. The concern of the clinician of missing an early curable melanoma conflicts with his or her desire to avoid unnecessary minor or even major surgery in cases where benign lesions show doubtful features. These issues are particularly evident when considering melanoma simulators, those entities that may be clinically and sometimes histologically confused with melanoma [1, 2, 3].
Pigmented spindle-cell nevus (PSCN) is a benign melanocytic lesion first described by Reed et al. [4]. It is generally considered a separate clinicopathologic entity, even if some authors classify this nevus as a variant of Spitz nevus [5, 6, 7, 8, 9, 10, 11]. PSCN has clinical and histopathologic features that may create difficulty in differentiating it from melanoma. We consider this tumor to be a separate clinical entity from Spitz nevus, and so we chose to focus our interest on its clinical features.
Epiluminescence Microscopy (ELM), a noninvasive technique employed in the clinical diagnosis of pigmented skin lesions, permits a detailed examination of the pigmented structures of the epidermis and dermoepidermal junction. The result is the visualization of morphological features not visible to the naked eye, which enhances the clinical diagnosis of nearly all the pigmented lesions including PSCN [12, 13, 14, 15, 16, 17].
This study presents a clinical and dermoscopic description of 63 consecutive cases of PSCN observed and excised at our institute. It evaluates the improvement led by ELM in the diagnosis of this lesion in order to reduce the number of diagnostic operations in patients bearing PSCN.
Patients and methods
All patients with histologic diagnosis of PSCN observed at the Istituto Nazionale per lo Studio e la Cura dei Tumori of Milan from January 1994 to December 1998 were evaluated. Cases submitted for consultation, cases with diagnostic uncertainty, or cases recoded as Spitz nevi were excluded. In total, 63 lesions in 63 patients (43 females and 20 males) were selected. Those cases, all examined clinically and then excised under local anesthesia in our institute, met the features regarded as histological characteristics of PSCN according to widely accepted criteria [18].
All lesions had a recorded clinical diagnosis that has been coupled with ELM evaluation. ELM was performed with a hand-held monocular microscope equipped with an achromatic lens permitting a magnification of 10×.
The distribution of cases according to site and gender is shown in Table 1. The patients ranged from 5 to 62 years, with a median age of 25 years.
| Site | Male (%) | Female (%) | Total (%) |
|
|
|||
| Head and neck | 2 (3.2) | 1 (1.6) | 3 (4.8) |
| Trunk | 9 (14.3) | 8 (12.7) | 17 (27.0) |
| Upper extremity | 5 (7.9) | 6 (9.5) | 11 (17.4) |
| Lower extremity | 4 (6.4) | 28 (44.4) | 32 (50.8) |
|
|
|||
The following gross morphological attributes were abstracted from the clinical records:
- color
- uniformity of pigmentation
- presence of a raised component on the skin surface
- regular versus irregular shape
- well-defined versus ill-defined borders
Also recorded were the main dermatoscopic features and the diagnoses performed by ELM.
In the context of another study on pigmented lesions still ongoing at our institution, sixteen of our cases of PSCN were also analyzed by a telespectrophotometric system in vivo before surgery. This system, based on measurements of lesion reflectance (i.e., the fraction of light reflected or diffused by a pigmented lesion), provides an objective evaluation of color of skin lesion [19]. The patients underwent to a regular followup for 5 years after surgery.
Results
The size of the lesions ranged from 2mm to 17 mm in diameter, with a median value of 5 mm. The clinical features of the 63 lesions are summarized in Table 2. Lesions were darkly pigmented in nearly all the cases. A uniform dark color was the hallmark of the majority of the lesions. The nevi were very often raised on skin surface, but not a significant number (22.2 %) of these tumors showed a flat conformation. A regular shape was present in 85.7 percent of cases, and a well-defined border was more common than an ill-defined border (87.3 % vs. 12.7 %).
| Feature | Number (%) | Feature | Number (%) |
|
|
|||
| Color | Elevation | ||
| Black | 29 (46.0) | Slightly elevated | 44 (69) |
| Brown | 20 (31.7) | Flat | 14 (22.2) |
| Black and brown | 9 (14.3) | Nodular | 5 (7.9) |
| Tan | 2 (3.2) | Total 63 (100) | |
| Black and blue | 1 (1.6) | Shape | |
| Black and grey | 1 (1.6) | Regular | 54 (85.7) |
| Brown and red | 1 (1.6) | Irregular | 9 (14.3) |
| Total 63 (100) | Total 63 (100) | ||
| Uniformity of pigmentation | Borders | ||
| Uniform | 52 (82.5) | Well-defined | 55 (87.3) |
| Not uniform | 11 (17.5) | Ill-defined | 8 (12.7) |
| Total 63 (100) | Total 63 (100) | ||
|
|
|||
All cases were also examined by ELM. Table 3 summarizes the clinical diagnoses of whole series of our cases. Table 4 shows clinical and dermatoscopic diagnoses of our 63 lesions.
|
| Figure 3 |
|---|
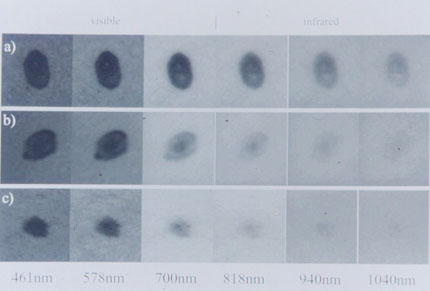
| Telespectrophotometric acquisition is represented, at selected wavelengths (nm) of (a) melanoma; (b) pigmented spindle-cell nevus; (c) compound nevus. |
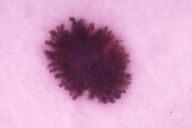
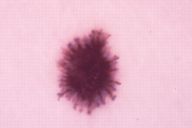
Our data put in evidence the two typical dermatoscopic presentations of PSCN: the first showing a brown to blue pigmentation and a peripheral rim of large brown globules (globular pattern) (Fig. 1), and the second revealing a dark diffused pigmentation and pseudopods regularly distributed at the periphery in a stellate or radiate pattern (starbust pattern) (Fig. 2). The first aspect was present in 34 cases (54 %), the second one in 27 cases (43 %). At ELM examination two lesions showed irregular pigment network and non-homogeneous pigmentationm and were regarded as suspected melanomas.
All sixteen lesions subjected to telespectrophotometric measurements showed a persistence of their images in the near-infrared band (between 940 and 1,040 nanometers). This datum represents an instrumental confirmation of the dark color of these lesions. This physical behavior of PSCN appears similar to that of melanoma [20]. Figure 3 shows typical reflectance of a melanoma, a PSCN and a common nevus, when imaged at different selected wavelengths. Numerical values of grey levels are proportional to the reflectance. Above 700 nm (near infrared) the common nevus is no longer visible and shows a reflectance close to that of the surrounding normal skin. In contrast, pigmentation of both melanoma and PSCN is detectable up to 1,040 nm.
All patients underwent to a regular followup for 5 years after surgery: no one had local relapse, regional or distant metastases.
|
|
| Figure 6 | Figure 7 |
|---|
|
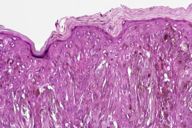
|
| Figure 8 | Figure 9 |
|---|
Conclusions
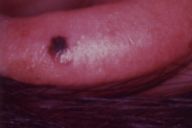
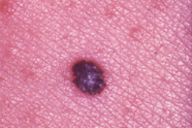
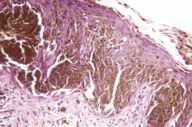
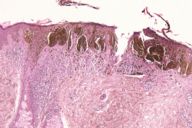
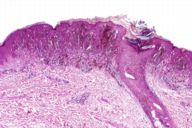
After the original description by Reed et al. [4], several series of PSCN have been published and the clinical and the histological features of these lesions have been defined [4, 5, 6, 7, 8, 9, 10, 11]. The data derived from our cases allow an integrated clinical and dermoscopic description of PSCN. The typical tumor is a small slightly raised lesion, generally showing a uniform black or brown color, with regular well-defined borders (Fig. 4). Its dark color makes PSCN a clinical simulator of melanoma. Notably the same tendency of PSCN of having a dark hue at telespectrophotometric analysis is showed by melanoma if examined by this technique [20]. These considerations, not to mention rare cases of lesions showing irregular borders or nonuniform pigmentation (Fig. 5), explain why PSCN is often misinterpreted as melanoma on clinical grounds. Several clinicopathologic studies have reported these difficulties in the clinical diagnosis of PSCN [5, 6, 8, 10]. When clinical diagnoses have been reported, 39-45 percent of PSCN cases had been diagnosed as melanoma [5, 6]. Also 25 percent of our cases had a clinical diagnosis of suspect melanoma (Tables 3 and 4).
| Diagnosis | Number (%) |
|
|
|
| PSCN | 38 (60.3) |
| Suspected melanoma | 16 (25.4) |
| Common nevus | 4 (6.3) |
| Spitz nevus | 2 (3.2) |
| Other | 3 (4.8) |
| Total | 63 (100) |
|
|
|
| Diagnosis | Clinical impression | Dermatoscopic diagnosis |
|
|
Number (%) | Number (%) |
|
|
||
| PSCN | 38 (60.3) | 61 (96.8) |
| Melanoma | 16 (25.4) | 2 (3.2) |
| Common nevus | 4 (6.3) | - (-) |
| Spitz nevus | 2 (3.2) | - (-) |
| Other | 3 (4.8) | - (-) |
| Total | 63 (100) | 63 (100) |
|
|
||
ELM has provoked a real improvement in the diagnosis of PSCN and Spitz nevus [16, 17]. By this technique we have performed a clearcut diagnosis in the overwhelming majority of PSCN cases. In our series the correct preoperative diagnosis was performed in 61 of 63 cases (97 %). A similar rate of correct diagnosis (93 %) was reached by Pehamberger et al. on a series of 54 cases of pigmented Spitz nevus [17]. However, dermatoscopic features suggesting melanoma are a reality even if rare, as two of our cases show. One of these cases, with a clinical and dermatoscopic diagnosis of melanoma was treated by a major excision with 1-cm margins.
Critical diagnosis and management decisions concerning pigmented lesions should always be based on a combination of clinical and dermatoscopic assessments. Indeed, in rare cases, melanoma can show the characteristics starbust of globular pattern usually seen in PSCN; in these cases, the correct diagnosis must be supported by anamnestic data concerning the history and the evolution of the lesion and by clinical features. This fact once more stresses the importance of not making a diagnosis of a pigmented lesion with ELM alone. In presence of a certain dermatoscopic diagnosis of PSCN on a lesion without a high clinical suspicion for melanoma, our current practice is not to proceed to a surgical excision for histological examination of the lesion; for these cases a careful clinic and dermatoscopic followup of the lesion can be sufficient.
By following this approach, we have reduced the number of diagnostic operations in patients bearing PSCN, sparing many patients unnecessary stress, avoiding complications and conserving medical resources.
Acknowledgments. This research was partially supported by the Italian Association for Cancer Research and the Italian Research Council. We are grateful to Francesca Falcetta and Luisa Morandi for data management. We are grateful to Giovanna Maimone and Claudia Vaccaro for photographic assistance.
References
1. Maize JC, Ackerman AB. Pigmented lesions of the skin. Philadelphia, Pa: Lea & Febiger 1987; 228-242.2. Ainsworth AM, Folberg R, Reed RJ, Clark WH Jr. Melanocytic nevi, melanocitomas, melanocytic dysplasia and uncommon forms of melanoma. In: Clark WH Jr, Goldman LI and Mastrangelo MJ, eds. Human Malignant Melanoma. New York, NY: Grune & Stratton, 1979: 8: 175-182.
3. Orchard DC, Dowling JP, Kelly JW. Spitz naevi misdiagnosed histologically as melanoma: prevalence and clinical profile. Australasian J Dermatol 1997; 38(1): 12-14.
4. Reed RJ, Ichinose H, Clark WH Jr, Mihm MC Jr. Common and uncommon melanocytic nevi and borderline melanomas. Semin Oncol 1975; 2: 119-147.
5. Smith NP. The pigmented spindle cell tumour of Reed; an underdiagnosed lesion. Semin Diagn Pathol 1987; 4: 75-87.
6. Sagebiel RW, Chinn EW, Egbert BM. Pigmented spindle-cell nevus: clinical and histologic review of 90 cases. Am J Surg Pathol 1984; 8: 645-653.
7. Barnhill RL, Mihm MC Jr. The pigmented spindle-cell nevus and its variants: distinction from melanoma. Br J Dermatol 1989; 121: 717-726.
8. Barnhill RL, Barnhill MA, Berwick M, Mihm MC Jr. The histologic spectrum of pigmented spindle-cell nevus. A review of 120 cases with emphasis on atypical variants. Hum Pathol 1991; 22(1): 52-58.
9. Sau P, Graham JH, Helwig EB. Pigmented spindle-cell nevus (Abstract). Arch Dermatol 1984; 120: 1615.
10. Sau P, Graham JH, Helwig EB. Pigmented spindle-cell nevus: A clinicopathologic analysis of ninety-five cases. J Am Acad Dermatol 1993; 28: 565-571.
11. Paniago-Pereira C, Maize JC, Ackerman AB. nevus of large spindle and/or epithelioid cells (SpitzÕs nevus). Arch Dermatol 1987; 114: 1811-1823.
12. Soyer HP, Smolle J, Kerl H, Stettner H. Early diagnosis of malignant melanoma by surface microscopy. Lancet 1987; II: 803.
13. Pehamberger H, Steiner A, Wolff K. In vivo Epiluminescence Microscopy of pigmented skin lesions. I. Pattern analysis of pigmented skin lesions. J Am Acad Dermatol 1987; 17: 584-591.
14. Stolz W, Bilek P, Landthaler M, Merkle T, Braun Falco O. Skin surface microscopy. Lancet 1989; II: 864-865.
15. Bahmer FA, Fritsch P, Kreusch J, Pehamberger H, Rohrer C, Schindera I, Smolle J, Soyer HP, Stolz W. Terminology in Surface Microscopy: Consensus Meeting of the Committee on Analytic Morphology of the Arbeitsgemeinschaft Dermatologische Forschung, Hamburg, Federal Republic of Germany, Nov 17, 1989. J Am Acad Dermatol 1990; 23: 1159-1162.
16. Steiner A, Pehamberger H, Binder M, Wolff K. Pigmented Spitz nevi: Improvement of the diagnostic accuracy by Epiluminescence Microscopy. J Am Acad Dermatol 1992; 27: 697-701.
17. Pehamberger H, Binder M, Steiner A, Wolff K. In vivo Epiluminescence Microscopy: Improvement of Early Diagnosis of Melanoma. J Inv Dermatol 1993; 100(3) Suppl: 356-362.
18. Elder DE, Murphy GF. Melanocytic tumors of the skin. In: Atlas of tumor pathology. Washington, DC: Armed Forces Institute of Pathology, 1991: 110-119.
19. Marchesini R, Tomatis S, Bartoli C, Bono A, Clemente C, Cupeta C, Del Prato I, Pignoli E, Sichirollo AE, Cascinelli N. In vivo spectrophotometric evaluation of neoplastic and non-neoplastic skin pigmented lesions. III. CCD camera-based reflectance imaging. Photochem Photobiol 1995; 62: 151-154.
20. Bono A, Tomatis S, Bartoli C, Tragni G, Radaelli G, Maurichi A, Marchesini R. The ABCD System of Melanoma Detection. A Spectrophotometric Analysis of the Asymmetry, Border, Color and Dimension. Cancer 1999; 85: 72-77.
© 2004 Dermatology Online Journal