A case of Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) related to rufinamide
Main Content
A case of Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) related to rufinamide
S Shahbaz1, RK Sivamani2, T Konia3,4, B Burrall2
Dermatology Online Journal 19 (4): 4
1. School of Medicine, University of California, Davis, Sacramento, California2. Department of Dermatology, University of California, Davis, Sacramento, California
3. Department of Dermatopathology, University of California, Davis, Sacramento, California
4. Department of Pathology, University of California, Davis, Sacramento, California
Abstract
Drug Rash (or Reaction) with Eosinophilia and Systemic Symptoms (DRESS) is a potentially life-threatening hypersensitivity reaction to drugs characterized by rash, fever, lymphadenopathy, hematologic abnormalities, and involvement of internal organs. Initially coined in 1996, the term is used to refer to an idiosyncratic reaction to several drugs, the most common of which are carbamazepine, allopurinol, sulfasalazine, and phenobarbital. We report the first case of DRESS related to rufinamide in a ten year old boy with a history of a complex seizure disorder.
Introduction
In an effort to decrease the ambiguity of the nomenclature used to describe cutaneous drug reactions, Drug Rash (or Reaction) with Eosinophilia and Systemic Symptoms was first coined in 1996 by Bocquet et al [1]. With an incidence ranging from 1 in 1000 to 1 in 10,000 drug exposures, the syndrome constitutes a severe drug reaction with anticonvulsants as the most common cause. Along with skin rash there is also fever, lymph node enlargement, hematologic abnormalities, and internal organ involvement. Hematologic abnormalities include eosinophilia, atypical lymphocytes, lymphocytopenia, thrombocytopenia, and lymphocytosis. Internal organ involvement has been noted to primarily affect the liver, but may also involve other organs such as the kidneys, lungs, heart, and pancreas. The reaction occurs approximately two to six weeks after initiating drug therapy and may persist or even worsen upon termination of the medication [1, 2].
We report a case of DRESS syndrome occurring in a patient using the novel anticonvulsant rufinamide. Marketed under the brand name Banzel, this triazole derivative was approved for use in the United States in 2008 as an anticonvulsant for complex seizure disorders. Although skin rash, multi-organ hypersensitivity, and Stevens-Johnson Syndrome (SJS) have previously been associated with rufinamide, it has never before, to our knowledge, been associated with DRESS syndrome [3, 4].
Case report
A ten-year-old fully immunized boy presented to the dermatology inpatient service with a one-day history of a generalized rash. His medical history was notable for a complex seizure disorder that required multiple medications including levetiracetam (since 2006), topiramate (since 2006), and rufinamde 300 mg twice daily, beginning one month prior to his hospitalization in 2011. He had tried and discontinued lamotrigine in the past because of the development of Stevens-Johnson Syndrome and phenobarbital because of the development of an unspecified rash.
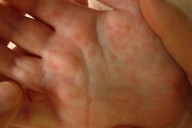 |
| Figure 1 |
|---|
| Figure 1. Multiple monomorphic, indurated, and erythematous papules with overlying vesicles on the palms. |
Physical exam revealed red, slightly indurated papules, coalescing into plaques and nodules on the cheeks and ears. Multiple monomorphic, indurated, and erythematous papules with overlying vesicles were noted on the arms, abdomen, dorsal hands, palms, legs, feet, soles, ventral foreskin, glans, and scrotum (Figure 1). An erythematous, indurated plaque was noted around the anus. There were no erosions and there was no involvement of the conjunctiva. Several purpuric macules were noted on the anterior hard palate. Left sided cervical lymphadenopathy was noted on initial presentation that generalized to bilateral cervical lymphadenopathy over the course of several days (Figures 2A and 2B). Direct immunofluorescence of the cutaneous vesicles revealed no Herpes Simplex or Varicella infection. His blood work was notable for lymphopenia at 800 cell/mm³.
 | 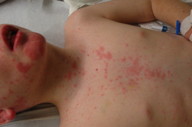 |
| Figure 2A | Figure 2B |
|---|---|
| Figure 2A. Prominent left sided cervical lymphadenopathy. Figure 2B. Note the erythematous papules on the face and chest as well. | |
A punch biopsy from one of the papules on his right flank revealed spongiosis with microvesicles overlying perivascular lymphohistiocytic infiltrates with rare eosinophils. The cornified layer was largely unaltered (Figure 3). The biopsy was consistent with spongiotic dermatitis with a component of interface dermatitis, consistent with a drug hypersensitivity reaction.
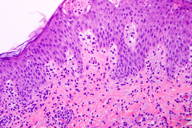 |
| Figure 3 |
|---|
| Figure 3. A punch biopsy from the right flank shows spongiosis with microvesicles overlying perivascular lymphohistiocytic infiltrates with rare eosinophils. |
The patient was diagnosed with DRESS and was initiated on a slow prednisone taper over three weeks with a starting dose at 1 mg/kg. The patient’s rash, lymphopenia, and lymphadenopathy resolved over the course of approximately three weeks. Although his seizures were initially well controlled, 1 year later he had a sporadic return of his seizures and is under consideration for a vagal nerve stimulator.
Discussion
Owing to the systemic involvement affecting a variety of internal organs, the diagnosis of this life-threatening drug reaction has been challenging. Although no gold standard for diagnosis exists at this time, a recent diagnostic approach using the RegiSCAR criteria was developed by Cacoub et al [2]. Using a registry of severe cutaneous adverse reactions (SCAR), cases were graded as “no” (score <2), “possible” (score 2-3), “probable” (score 4-5), or “definite” (score >5) cases of DRESS based on the following criteria: skin involvement, fever, lymphadenopathy, eosinophilia, atypical lymphocytes, internal organ involvement, resolution time, and the possibility of other causes. Skin involvement consists of maculopapular erythematous lesions often progressing to edema and exfoliative dermatitis [2].
Furthermore, a Japanese consensus group in 2006 developed a separate diagnostic approach for a more serious form of DRESS syndrome entitled Drug-Induced Hypersensitivity Syndrome (DIHS). A diagnosis for typical DIHS requires all of the following seven criteria to be met (atypical DIHS requires only the first five): maculopapular rash developing at least three weeks after initiating the drug, symptoms present two weeks post termination of the drug, fever, elevated liver enzymes, leukocyte abnormalities, lymph node enlargement, and Human Herpes Virus 6 reactivation [5]. Table 1 lists the diagnostic criteria for DRESS, DIHS, and SJS.
According to the RegiSCAR diagnostic criteria, our patient would be classified as a “probable” case because of the presence of fever (0 points), skin involvement (1 point), lymphadenopathy (1 point), lymphopenia (1 point), resolution past 15 days (0 points), internal organ involvement of the bone marrow and lymph nodes (1 point) and exclusion of other possible causes (1 point). Negative blood cultures and serology for Hepatitis A, B, C, Staphylococcus aureus, Streptococcus pyogenes, Chlamydia, and Mycoplasma, as well as absent antinuclear antibodies ruled out other potential causes such as viral hepatitis, toxic shock syndrome, staphylococcal scalded skin syndrome or connective tissue disorders. Furthermore, our patient’s distinct skin involvement of maculopapular erythematous lesions is consistent with the RegiSCAR description [2].
Other possible causes such as Stevens-Johnson Syndrome (SJS) and its more severe form Toxic Epidermal Necrolysis (TEN), Erythema Multiforme (EM), Acute Generalized Exanthematous Pustulosis (AGEP), and Kawasaki Disease were also entertained. Although both SJS and TEN involve the development of fever, mucocutaneous lesions, and hematological abnormalities, there is associated epidermal sloughing and in most cases conjunctival involvement, both of which are absent in our patient [6, 7]. Most cases of EM are thought to relate to HSV, which was not found in our patient. Furthermore, the patient lacked the hallmark targetoid lesions typically present in EM [8]. The absence of characteristic sterile pustules and the typical onset of eruption an average of five days after drug initiation decrease the possibility of AGEP. The oral mucosa is also typically spared in AGEP, which is inconsistent with the findings in our patient [9]. Given the age and clinical findings of cervical lymphadenopathy and erythematous rash seen in our patient, Kawasaki disease is certainly a possibility. However, the disease is not typically associated with medication usage but is rather an autoimmune condition with infectious and genetic etiologies. Additionally, the guidelines developed by Kawasaki would rule out this diagnosis owing to the lack of conjunctival injection and characteristic mucosal and peripheral extremity changes typically seen with the disease [10, 11].
Although the pathogenesis of DRESS is not completely understood, several mechanisms have been proposed including arene oxide metabolite formation of anticonvulsant drugs and the subsequent immunological reactions to these metabolites. Reactivation of Human Herpes Virus (particularly HHV6 but also CMV and EBV) and paramyxovirus has also been implicated as a contributory factor to the development of DRESS [2]. The syndrome may have a genetic component because it occurs more frequently in those with certain HLA types [12]. Irrespective of the precise pathogenesis, several medications have been identified as the instigating event. Using the RegiSCAR system as a guide, over forty medications to date have been reported as at least “possible” causes of DRESS. The most common of these, in order of incidence, include carbamazepine, allopurinol, sulfazalazine, phenobarbital, nevirapine, lamotrigine, phenytoin, mexilletine, dapsone, and vancomycin. In general, anticonvulsants were responsible for most of the reported cases, though other drug classes such as antipsychotics, antihypertensives, antiarrhythmics, antibiotics, lipid-lowering agents, opioids, chemotherapeutics, H2 blockers, and antivirals were also documented [2].
Interestingly, anticonvulsants have been known to cause several forms of severe skin reactions, with the term Dilantin Hypersensitivity Syndrome first being proposed in 1950. Once the syndrome became associated with other arene oxide producing aromatic anticonvulsants (phenobarbital, carbamazepine and primidone), the term was changed to Anticonvulsant Hypersensitivity Syndrome (AHS) in 1988 [13]. More recently, lamotrigine has been shown to cross-react with the older aromatics. In fact, cross-reactivity between aromatic anticonvulsants has been documented to be as high as 75%, leading to recommendations to avoid similar aromatic anticonvulsants once a reaction has developed [14]. Clinical manifestations consist of a triad of fever, rash, and internal organ involvement occurring one to eight weeks after initiating the drug [13]. It is indeed possible that our patient experienced cross reactivity with rufinamide; he had experienced hypersensitivity reactions to both lamotrigine and phenobarbital in the past.
The culprit medication rufinamide is a triazole derivative approved in 2008 as an adjunctive medication for those over the age of four with complex seizure disorders, particularly Lennox-Gastaut Syndrome. This relatively novel anticonvulsant has an incompletely understood mechanism of action but has been shown to prolong the inactive state of sodium channels in vitro [4]. Interestingly, it has an aromatic ring structure but otherwise bears no chemical similarities to the other antiepileptics. The most common side effects include somnolence, vomiting, and headache, with a few cases of antiepileptic drug hypersensitivity syndrome that immediately resolved once the drug was stopped [15]. Of note is a recently documented case of Stevens-Johnson Syndrome after rufinamide use [3].
In considering further options for the treatment of new seizure activity in our patient, his documented adverse skin reactions to three different anticonvulsants must be kept in mind. Carbamazepine, phenytoin, lamotrigine, phenobarbital, and zonisamide have all been associated with DRESS syndrome [2] and should not be used. Primidone must also be avoided because one of its active metabolites is phenobarbital. Promising options, which have not been associated with DRESS or AHS and are helpful as adjunctive medications in complex seizure disorders, include valproic acid, the carbamic acid felbamate, and the newly available benzodiazepine clobazam. However, clobazam has been associated with SJS in combination treatment with lamotrigine and valproic acid, possibly by affecting the clearance of the other two drugs [15, 16].
The mainstay of DRESS treatment consists of immediate drug withdrawal and a slow taper of corticosteroid administration over a period of three to four weeks. Intravenous immunoglobulin has been given as an adjunct to corticosteroid treatment in a few cases, yet the mechanism and effectiveness of this treatment has yet to be elucidated. Resolution of symptoms may take up to two weeks after stopping the offending drug with a range of 0.5–90 weeks of treatment before full recovery [2]. Although no controlled trials have yet assessed the efficacy of corticosteroid treatment in DRESS, immediate action is necessary, especially with internal organ involvement, because the syndrome has a current mortality rate of 10 percent [17].
In conclusion, we present a unique case of DRESS syndrome related to the relatively new anticonvulsant rufinamide. Using the RegiSCAR criteria, we believe it appropriate to add rufinamide to the list of drugs associated with this life-threatening hypersensitivity reaction. Patients on this medication should be monitored for any signs and symptoms of both skin and internal organ involvement. Diagnosis and treatment should follow current guidelines, although further research is required to more adequately understand this complex syndrome.
References
1. Bocquet H, Bagot M, Roujeau JC (1996) Drug-induced pseudolymphoma and drug hypersensitivity syndrome (Drug Rash with Eosinophilia and Systemic Symptoms: DRESS). Semin Cutan Med Surg 15: 250-257. [PubMed]2. Cacoub P, Musette P, Descamps V, Meyer O, Speirs C, et al. (2011) The DRESS syndrome: a literature review. Am J Med 124: 588-597. [PubMed]
3. Chambel M, Mascarenhas MI, Regala J, Gouveia C, Prates S (2012) Clinical Stevens-Johnson syndrome and rufinamide: A clinical case. Allergol Immunopathol (Madr): [PubMed]
4. Deeks ED, Scott LJ (2006) Rufinamide. CNS Drugs 20: 751-760; discussion 761. [PubMed]
5. Shiohara T, Iijima M, Ikezawa Z, Hashimoto K (2007) The diagnosis of a DRESS syndrome has been sufficiently established on the basis of typical clinical features and viral reactivations. Br J Dermatol 156: 1083-1084. [PubMed]
6. Bastuji-Garin S, Rzany B, Stern RS, Shear NH, Naldi L, et al. (1993) Clinical classification of cases of toxic epidermal necrolysis, Stevens-Johnson syndrome, and erythema multiforme. Arch Dermatol 129: 92-96. [PubMed]
7. Morales ME, Purdue GF, Verity SM, Arnoldo BD, Blomquist PH (2010) Ophthalmic Manifestations of Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis and Relation to SCORTEN. Am J Ophthalmol 150: 505-510 e501. [PubMed]
8. Huff JC, Weston WL (1989) Recurrent erythema multiforme. Medicine (Baltimore) 68: 133-140. [PubMed]
9. Sidoroff A (2012) Acute generalized exanthematous pustulosis. Chem Immunol Allergy 97: 139-148. [PubMed]
10. Burns JC, Glode MP (2004) Kawasaki syndrome. Lancet 364: 533-544. [PubMed]
11. Fujita Y, Nakamura Y, Sakata K, Hara N, Kobayashi M, et al. (1989) Kawasaki disease in families. Pediatrics 84: 666-669. [PubMed]
12. McCormack M, Alfirevic A, Bourgeois S, Farrell JJ, Kasperaviciute D, et al. (2011) HLA-A*3101 and carbamazepine-induced hypersensitivity reactions in Europeans. N Engl J Med 364: 1134-1143. [PubMed]
13. Schlienger RG, Shear NH (1998) Antiepileptic drug hypersensitivity syndrome. Epilepsia 39 Suppl 7: S3-7. [PubMed]
14. Knowles SR, Shapiro LE, Shear NH (1999) Anticonvulsant hypersensitivity syndrome: incidence, prevention and management. Drug Saf 21: 489-501. [PubMed]
15. Wheless JW, Constantinou JE (1997) Lennox-Gastaut syndrome. Pediatr Neurol 17: 203-211. [PubMed]
16. Ertam I, Sezgin AO, Unal I (2009) A case with Stevens Johnson syndrome triggered by combination of clobazam, lamotrigine, and valproic acid treatment. Int J Dermatol 48: 98-99. [PubMed]
17. Walsh SA, Creamer D (2011) Drug reaction with eosinophilia and systemic symptoms (DRESS): a clinical update and review of current thinking. Clin Exp Dermatol 36: 6-11. [PubMed]
18. Mockenhaupt M, Viboud C, Dunant A, Naldi L, Halevy S, et al. (2008) Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. J Invest Dermatol 128: 35-44. [PubMed]
© 2013 Dermatology Online Journal


