Verruciform xanthoma: Report of two cases
Published Web Location
https://doi.org/10.5070/D37cw3906jMain Content
Verruciform xanthoma: Report of two cases
Vera Teixeira, José P Reis, Óscar Tellechea, Ricardo Vieira, Américo Figueiredo
Dermatology Online Journal 18 (5): 10
Department of Dermatology, Coimbra University Hospital, PortugalAbstract
Verruciform xanthoma (VX) is an uncommon benign condition of unknown etiology, which frequently affects the oral mucosa in adults. Other less common locations include the anogenital region and the skin. VX typically presents as an asymptomatic plaque showing a verrucous appearance. Histological examination is essential for the diagnosis and shows verrucous hyperplasia of the epidermis and xanthoma cells limited to the dermal papillae. We present herein two cases of VX and discuss the histopathological findings and possible correlation with a postulated etio-pathogenesis.
Introduction
Verruciform xanthoma (VX) is an uncommon benign lesion that was first described by Shafer in 1971 [1]. It generally affects the oral mucosa, although it may also involve the anogenital region [2, 3]. VX typically presents, as an asymptomatic papule or single plaque with a verrucous surface that is frequently misdiagnosed as verruca vulgaris, condyloma accuminatum, or verrucous carcinoma. Thus, the clinical diagnosis is challenging, requiring a high level of suspicion and confirmation by histological examination, which demonstrates verrucous hyperplasia of the epidermis and a variable number of xanthoma cells within the dermal papillae, the hallmark of VX. We report two cases. The first one presented on the penis, which is an uncommon location for VX, whereas the second case corresponding to a more a typical oral VX, in an immunosuppressed patient.
Case 1
An 85-year-old male with history of chronic eczema presented with a painless penile lesion that had been present for several months. The physical examination revealed a slightly infiltrated, well-circumscribed, verrucous plaque of 1 x 1.5 cm in diameter, on the distal part of the foreskin (Figure 1A). There was no regional lymphadenopathy and the remainder of the physical examination was unremarkable. The lesion was clinically diagnosed as a verrucous carcinoma. Syphilis serologic test was negative and routine laboratory parameters were all within normal limits, including a serum lipid profile.
Case 2
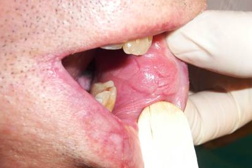 |
| Figure 1 |
|---|
| Figure 1. Clinical aspect of the verruciform xanthoma of penis (A) and oral mucosa (B). |
A 48-year-old man was referred to our clinic for an asymptomatic lesion on the left side of the oral mucosa. The lesion had appeared six years earlier, during immunosuppressive treatment after allogenic hematopoietic stem cell transplantation for multiple myeloma. At physical examination, a well-demarcated, slightly raised grayish pink plaque with a papillomatous surface on the left buccal mucosa was identified (Figure 1B). Oral florid papillomatosis was the presumptive clinical diagnosis. Routine chemical and lipid evaluation were unremarkable.
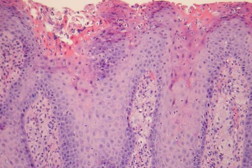
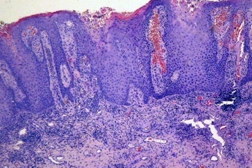
The histological examination of the lesions of the two patients disclosed identical findings (Figures 2 and 3): A well circumscribed regularly acanthotic and verrucous epithelial lesion, with a rectilinear inferior border, shows columns of parakeratosis invaginated within the thickened and enlarged rete ridges. The invaginated parakeratotic columns result from the superposition of eosinophilic keratinocytes with abundant hyaline cytoplasm and a relatively preserved nucleus. Spinous keratinocytes exhibiting minute cytoplasmic vacuolar change underly these changes. Additionally, exocytosis of neutrophils is demonstrated and although there was no cytological atypia, they created a pseudo atypical aspect in areas. The dermal papillae contain a variably rich infiltrate of mononucleated cells with a large pale cytoplasm that is foamy at higher magnification. These cells are immunoreactive for CD 68 confirming that they correspond to foamy macrophages (xanthoma cells). No giant cells or Touton cells were identified. Diagnosis of VX was made on these findings. Polymerase chain reaction amplification of HPV, in the lesion of patient 2, was negative.
In the first patient the destruction of the lesion with carbon dioxide laser was performed and no recurrence of the lesion was seen in the ensuing twelve months. In the second patient complete destruction of the lesions with carbon dioxide laser was carried out. However, 6 months later, multifocal recurrence was appreciated, which was efficiently treated by spray cryotherapy.
Discussion
Clinically, VX is usually described as an asymptomatic solitary verrucous, papillomatous, papule or plaque with a color varying from reddish pink to gray and a diameter ranging from 0.2 cm to 2 cm. It may mimic a viral wart, condyloma accuminatum, verrucous carcinoma or, less frequently, squamous cell carcinoma. It is crucial to rule out squamous cell carcinoma because of its therapeutic implications. VX usually occurs in the oral mucosa, with few reports in the anogenital region and skin. To our knowledge, only 18 cases of VX of the penis have been reported so far.
Histopathologically, VX is characterized by hyperkeratosis, columnar invaginated parakeratosis, verrucous acanthosis, elongation of rete rigdes, and dermal macrophage foam cells. These xanthoma-like cells, a hallmark of VX, are restricted to the dermal papillae and do not extend below rete pegs tips. Although no nuclear atypia is found, the focal epithelial disorganization and the cytological pleomorphism can evoke the diagnosis of dysplasia. Distinction from verrucous carcinoma with dermal xanthomatous changes can be difficult and should stand on architectural criteria and the absence of nuclear atypia.
The etio-pathogenesis of VX remains elusive and many hypotheses have been suggested, including trauma, viruses, and impaired immune response. In 1974, Zegarelli et al [4] proposed that VX may result from epithelial degeneration by local irritation or trauma with the releasing of lipids from keratinocytes, which are engulfed by macrophages in the dermal papillae (histiocytic response). The vacuolar change in keratinocytes beneath the parakeratotic invaginated columns, as seen in both our cases, would support this hypothesis. Mohsin et al [5] claimed that chemotactic cytokines produced by degenerative keratinocytes attract neutrophils and stimulate rapid epidermal growth. Therefore, it has been speculated that VX may be a unique reaction pattern to an initiating event that leads to selective recruitment and persistent accumulation of foamy macrophages in the dermal papillae.
HPV has been suggested as a causative agent of VX [6]. However, numerous studies (including our own) have failed to detect HPV in VX lesions [7, 8].
However, the methods used to identify HPV DNA in the sample do not cover the wide spectrum of HPV types and also vary widely from study to study. Thus, a negative result does not exclude the presence of HPV DNA in VX lesions because it might represent a false negative. On the other hand, because HPV is a ubiquitous organism that can be isolated from apparently normal tissues, detection of HPV DNA in VX may represent a coincidental finding.
Besides the fact that VX mainly affects healthy adults, there has been an increase in the number of reports of VX in the context of immunosuppression (bone-marrow transplantation, chronic graft-versus- host disease, HIV infection) in recent years [9, 10]. Thus, the impaired immune response may also have a role in the pathogenesis of these lesions. Further studies are needed to definitively establish the causes of this interesting clinical entity.
References
1. Shafer WG. Verruciform xanthoma. Oral Surgery 1971; 31:784-9. [PubMed]2. De Rosa G, Barra E, Gentile R, et al. Verruciform xanthoma of the vulva: case report. Genitourin Med 1989; 65:252-254. [PubMed]
3. Kraemer BB, Schmidt WA, Foucar E, et al. Verruciform xanthoma of the penis. Arch Dermatology 1981; 117:516-518. [PubMed]
4. Zegarelli DJ, Aegarelli-Schmidt EC, Zegarelli EV. Verruciform xanthoma. A clinical, light microscopic, and electron microscopic study of two cases. Oral Surg Oral Med Oral Pathol 1974; 38:725. [PubMed]
5. Mohsin SK, Lee MW, Amin MB, et al. Cutaneous verruciform xanthoma: a report of five cases investigating the etiology and nature of xanthomatous cells. Am J Surg Pathol 1998; 22:479. [PubMed]
6. Khaskhely NM, Uezato H, Kamiyama T, et al. Association of human papillomavirus type 6 with a verruciform xanthoma. Am J Dermatopathol 2000; 22:447. [PubMed]
7. Orchard GE, Jones EW, Jones RR. Verruciform xanthoma. An immunocytochemical study. Brit J Biomed Sci 1994; 1:28-34. [PubMed]
8. Ersahin C, Szpaderska AM, Foreman K, et al. Verruciform xanthoma of the penis not associated with human papillomavirus infection. Arch Pathol Lab Med 2005; 129:62-64. [PubMed]
9. J.Kanitakis, S.Euvrard, A.C.Butnaru, A.Claudy. Verruciform xanthoma of the scrotum in a renal transplant patient. British Journal of Dermatology 2004; 150(1):155-177. [PubMed]
10. Sibaud V, Marit G, Deminiere C, et al. Multiple verruciform xanthomas of the oral mucosa associated with graft versus host disease. Ann Dermatol Venereol. 2006; 133:995-9. [PubMed]
© 2012 Dermatology Online Journal