Signet ring-like cells: No longer a “signature” of glandular differentiation
Published Web Location
https://doi.org/10.5070/D36q36z4d7Main Content
Signet ring-like cells: No longer a “signature” of glandular differentiation
David N Lortscher1 MD, Elizabeth K Satter2 MD MPH, Laura S Romero1 MD
Dermatology Online Journal 18 (4): 3
1. Department of Medicine (Dermatology), University of California, San Diego, California2. Department of Dermatology, Naval Medical Center, San Diego, California
Abstract
The term “signet ring” is used to describe a cell with an eccentric nucleus that is compressed to the periphery of the cell by a large cytoplasmic vacuole or by a substance accumulated within the cytoplasm. In pathology, these cells have been traditionally associated with adenocarcinomas. We describe two cases of cutaneous tumors that contained signet ring-like cells. Because of the morphology of the cells and the fact that one patient had a past history of lung cancer, the possibility of metastatic adenocarcinoma was considered. Upon further investigation, the tumors proved to be a primary cutaneous squamous cell carcinoma and a basal cell carcinoma; surgical management was curative. Because signet ring changes may pose a diagnostic challenge, we review the various benign and malignant cutaneous lesions that have been reported to contain signet ring cells and we explore the possible causative mechanisms of this phenomenon.
Introduction
Historically, the term signet ring refers to a distinctive type of ring bearing an engraved seal that was used as a means to stamp official documents. In pathology, the term is used to describe cells with an eccentric nucleus that is compressed to the periphery of the cell by a large cytoplasmic vacuole or by a substance accumulated within the cytoplasm. Although once considered synonymous with adenocarcinoma, a variety of tumors have been reported to contain signet ring-like cells. Herein we present two cases of non-melanoma skin cancer with signet ring-like morphology.
Case 1
A 67-year-old man presented to the dermatology clinic for a centrally crusted, erythematous nodule of 1.5 cm diameter near the left lateral canthus. He had a history of lung cancer diagnosed 2 years previously, but no information on the stage or histologic type was available. The clinical differential diagnosis was basal cell carcinoma (BCC), squamous cell carcinoma (SCC), or cyst and a shave biopsy was performed.
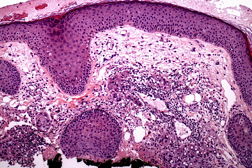
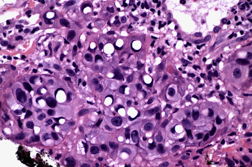
Histopathology revealed a poorly circumscribed, infiltrating tumor composed of irregular epithelioid cells with large nuclei and prominent nucleoli. Additionally noted were scattered cells that contained prominent empty perinuclear vacuoles that displaced the chromatin peripherally, imparting a signet ring-like appearance (Figures 1A and 1B). Because there was no apparent epidermal attachment and given that the patient had a history of lung cancer, a diagnosis of metastatic adenocarcinoma was considered. However, repeat biopsy disclosed areas more typical of SCC with only isolated signet ring-like cells. The tumor stained diffusely with cytokeratin 5/6, P63, and EMA, and was negative for CK7, CK20, and CEA. Therefore, a diagnosis of cutaneous SCC was given. Mohs micrographic surgery was performed and the patient has shown no evidence of recurrence for 13 months postoperatively.
Case 2
A 94-year-old female presented to dermatology clinic with a one-year history of a pearly telangiectatic papule on the left nasal sidewall. A BCC was clinically suspected and a shave biopsy was performed.
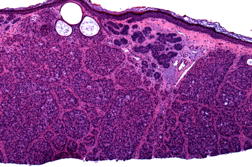
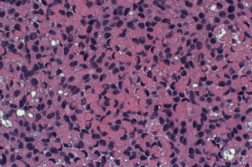
Histopathology revealed a proliferation of irregular nests and cords that emanated from the epidermis. The majority of cells contained a large eosinophilic cytoplasmic inclusion that resulted in eccentric displacement of the nucleus, imparting a signet ring-like appearance (Figures 2A and 2B). Deeper levels revealed a more classic basaloid proliferation, which was BER-EP4 positive and consistent with BCC. Mohs micrographic surgery was performed and the patient has shown no evidence of recurrence for 15 months postoperatively.
Discussion
In the pathology literature, the term signet ring was originally used to describe cells that exhibited peripheral displacement of the nucleus caused by the accumulation of cytoplasmic mucin in some adenocarcinomas. However, over the years, a variety of benign and malignant primary cutaneous neoplasms have been reported to contain cells with a similar morphology [1]. These include BCC [2-13], SCC [14-16], melanocytic nevi [17], melanoma [18], apocrine hidradenoma [1], cylindroma [1], dermatofibroma [19], primary cutaneous signet ring carcinoma of the eyelid [20], liposarcoma [21], epithelioid sarcoma [22], and cutaneous lymphoma [23]. Additionally, signet ring cells have been reported in inflammatory conditions, including signet-ring mucicarminophilic histiocytosis [24], artifactual post-procedure inflammation [25], and signet-ring sinus histiocytosis [26]. These signet ring-like cells have also been referred to in the literature as plasmacytoid or hyaline cells [7].
The exact etiology of this phenomenon is unknown. However, there appear to be two distinct types of signet ring-like cells: those that arise because of cytoplasmic accumulation of various substances (such as sialomucin [5], immunoglobulin [23], glycogen [3, 4, 14], keratin [8, 10, 12], and other intermediate filaments [7, 9, 11]) and those that arise as a result of formation of cytoplasmic vacuoles related to hydropic degeneration [1], invaginations of the cytoplasmic membrane [27], dilatations of the endoplasmic reticulum [16], or as a result of a processing artifact [25].
In our first case, the optically clear spaces in the SCC did not stain with alcian blue, mucicarmine, or PAS, indicating that the clear spaces were not filled with mucin or glycogen. The spaces were in close approximation to the nuclear chromatin, suggesting that they may be intranuclear or possibly associated with the endoplasmic reticulum. Although electron microscopy was not performed in our case, McKinley et al reported a morphologically similar SCC; ultrastructural examination showed that the vacuoles represented large dilatations of cisternae of the rough endoplasmic reticulum [16].
The signet ring morphology in our second case occurred as a result of accumulation of eosinophilic material in the cytoplasm. Because only rare signet ring cells were seen on deeper levels, further characterization of the material was not possible. Multiple previous cases of signet ring BCC have shown positive staining with smooth muscle actin and high molecular weight cytokeratin, and have been referred to as BCC with myoepithelial differentiation [2, 6, 7, 9, 11]. Other signet ring BCCs have been reported to have inclusions composed of keratin [10, 12, 8], glycogen [3, 4], and sialomucin [5]; electron microscopy has demonstrated various types of intermediate filaments [7, 9, 10].
In conclusion, although the presence of signet ring cells in a cutaneous tumor should invoke consideration of metastatic adenocarcinoma, a number of other neoplasms have been reported to contain cells with a similar morphology. Therefore, signet ring presence is not necessarily indicative of glandular differentiation. Because the formation of these cells can occur via a variety of mechanisms, signet ring formation should be considered a cellular alteration similar to that seen in granular cells, oncocytic cells, balloon cells, and rhabdoid cells, rather than being lineage-specific [1].
References
1. Bastian BC, Kutzner H, Yen T, LeBoit PE. Signet-ring cell formation in cutaneous neoplasms. J Am Acad Dermatol 1999; 41(4):606-613. [PubMed]2. Aroni K, Lazaris AC, Nikolaou I, Saetta A, Kavantzas N, Davaris PS. Signet ring basal cell carcinoma. A case study emphasizing the differential diagnosis of neoplasms with signet ring cell formation. Pathol Res Pract 2001; 197(12):853-856. [PubMed]
3. Barr RJ, Williamson C. Clear-cell basal cell carcinoma. Arch Dermatol 1984; 120(8):1086.
4. Cohen RE, Zaim MT. Signet-ring clear-cell basal cell carcinoma. J Cutan Pathol 1988; 15(3):183-187. [PubMed]
5. Kim do Y, Cho SB, Chung KY, Kim YC. Clear cell basal cell carcinoma with sialomucin deposition. Yonsei Med J 2006; 47(6):870-872. [PubMed]
6. Kim YC, Vandersteen DP, Chung YJ, Myong NH. Signet ring cell basal cell carcinoma: a basal cell carcinoma with myoepithelial differentiation. Am J Dermatopathol 2001; 23(6):525-529. [PubMed]
7. Na SY, Choi YD, Choi C, Kim SJ, Won YH, Yun SJ. Basal cell carcinoma with myoepithelial differentiation: a distinct plasmacytoid cell variant with hyaline inclusions. J Cutan Pathol 2011; 38(5):448-452. [PubMed]
8. Roh MR, Kwon JE, Lee KG, Lee JH. Signet-ring cell basal cell carcinoma. J Cutan Pathol 2010; 37(11):1188-1189. [PubMed]
9. Sahin AA, Ro JY, Grignon DJ, Ordonez NG. Basal cell carcinoma with hyaline inclusions. Arch Pathol Lab Med 1989; 113(9):1015-1018. [PubMed]
10. Seo IS, Warner TF, Priest JB. Basal cell carcinoma - signet ring type. Ultrastructural study. J Cutan Pathol 1979; 6(2):101-107. [PubMed]
11. Suster S, Ramon y Cajal S. Myoepithelial differentiation in basal cell carcinoma. Am J Dermatopathol 1991; 13(4):350-357. [PubMed]
12. White GM, Barr RJ, Liao SY. Signet ring cell basal cell carcinoma. Am J Dermatopathol 1991; 13(3):288-292. [PubMed]
13. Zheng S, Guo Y, Mones JM. Basal cell carcinoma with myoepithelial differentiation. Am J Dermatopathol 2011; 33(8):863-866. [PubMed]
14. Cramer SF, Heggeness LM. Signet-ring squamous cell carcinoma. Am J Clin Pathol 1989; 91(4):488-491. [PubMed]
15. El Demellawy D, Onuma K, Alowami S. Signet ring squamous cell carcinoma--the forgotten variant: case report and review of the literature. J Cutan Pathol 2011; 38(3):306-8. [PubMed]
16. McKinley E, Valles R, Bang R, Bocklage T. Signet-ring squamous cell carcinoma: a case report. J Cutan Pathol 1998; 25(3):176-181. [PubMed]
17. LiVolsi VA, Brooks JJ, Soslow R, Johnson BL, Elder DE. Signet cell melanocytic lesions. Mod Pathol 1992; 5(5):515-520. [PubMed]
18. al-Talib RK, Theaker JM. Signet-ring cell melanoma. light microscopic, immunohistochemical and ultrastructural features. Histopathology 1991; 18(6):572-575. [PubMed]
19. Garrido-Ruiz MC, Carrillo R, Enguita AB, Peralto JL. Signet-ring cell dermatofibroma. Am J Dermatopathol 2009; 31(1):84-87. [PubMed]
20. Rosen Y, Kim B, Yermakov VA. Eccrine sweat gland tumor of clear cell origin involving the eyelids. Cancer 1975; 36(3):1034-1041. [PubMed]
21. Flenker H. Myxoid liposarcoma. Light and electron microscopic investigation. Virchows Arch A Pathol Anat Histol 1976; 371(2):171-176. [PubMed]
22. Mills SE, Fechner RE, Bruns DE, Bruns ME, O'Hara MF. Intermediate filaments in eosinophilic cells of epithelioid sarcoma: a light-microscopic, ultrastructural, and electrophoretic study. Am J Surg Pathol 1981; 5(2):195-202. [PubMed]
23. Weiss LM, Wood GS, Dorfman RF. T-cell signet-ring cell lymphoma. A histologic, ultrastructural, and immunohistochemical study of two cases. Am J Surg Pathol 1985; 9(4):273-280. [PubMed]
24. Kuo TT, Hsueh S: Mucicarminophilic histiocytosis. A polyvinylpyrrolidone (PVP) storage disease simulating signet-ring cell carcinoma. Am J Surg Pathol 1984; 8(6):419-428. [PubMed]
25. Alguacil-Garcia A: Artifactual changes mimicking signet ring cell carcinoma in transurethral prostatectomy specimens. Am J Surg Pathol 1986; 10(11):795-800. [PubMed]
26. Guerrero-Medrano J, Delgado R, Albores-Saavedra J: Signet-ring sinus histiocytosis: a reactive disorder that mimics metastatic adenocarcinoma. Cancer 1997; 80(2):277-285. [PubMed]
27. Grogan TM, Richter LC, Payne CM, Rangel CS. Signet-ring cell lymphoma of T-cell origin. An immunocytochemical and ultrastructural study relating giant vacuole formation to cytoplasmic sequestration of surface membrane. Am J Surg Pathol 1985; 9(9):684-692. [PubMed]
© 2012 Dermatology Online Journal