Sweet syndrome associated with Hashimoto thyroiditis
Published Web Location
https://doi.org/10.5070/D36nm353kjMain Content
Sweet syndrome associated with Hashimoto thyroiditis
Sandra Medeiros1, Raquel Santos2, Victor Carneiro3, Francisca Estrela1
Dermatology Online Journal 14 (9): 10
1. Dermatology Department, Hospital do Divino Espírito Santo, Ponta Delgada. sandradmedeiros@clix.pt2. Dermatology Department, Hospital Curry Cabral, Lisbon
3. Pathology Department, Hospital do Divino Espírito Santo, Ponta Delgada
Abstract
Sweet syndrome was originally described in 1964 by the English dermatologist R. D. Sweet. Nakayama et al. first reported the simultaneous occurrence of this syndrome and Hashimoto thyroiditis in 1993. The role of cytokines in the development of both Hashimoto thyroiditis and Sweet syndrome may be the link between these two conditions.
Clinical Synopsis
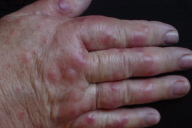
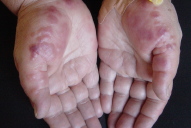
The authors report a case of a 65-year-old woman admitted to our hospital with a two day history of an abrupt onset of painful, purple-red papules asymmetrically distributed on her hands, forearms, neck, dorsal back, and feet that gradually coalesced to form small to medium, irregular, sharply bordered, edematous plaques with a vesicle-like appearance (Figs. 1 & 2).
 |  |
| Figure 1 | Figure 2 |
|---|---|
| Figures 1 and 2. Palmar and dorsal view of both hands with scattered tumid erythematous and edematous papules and plaques | |
The mucous membranes were spared. She complained of general malaise and ankle arthralgias. She denied fever, recent sore throat, rhinorrhea, cough, visual changes, or recent vaccination. Past medical history was irrelevant, except for the presence of sporadic epigastric symptoms for the last year and a previous hysterectomy with oophorectomy performed 20 years before. No gynecological malignancy was found. She denied the use of any medication and the remainder of the physical examination was normal.
A complete laboratory and imaging evaluation was performed in order to investigate possible associated systemic disorders, infectious, or inflammatory conditions. Routine laboratory tests were normal except for an elevated white blood cell count (12.2x10³/μL) with neutrophilia (74%); a C reactive protein of 3.4mg/dL; and an erythrocyte sedimentation rate of 50mm/h. In addition, renal function tests, serum protein electrophoresis, and immunoelectrophoresis were within normal values. Findings were also normal or negative for immunological tests (rheumatoid factor, anti-nuclear antibodies, anti-double stranded DNA antibodies, anti-extractable nuclear antigen antibodies, anti-neutrophil cytoplasmic antibodies, and complement), except for circulating antibodies to thyroid peroxidase that were present in high titers (1243 U/ml). Thyroid stimulating hormone (TSH) receptor antibodies and thyroglobulin antibody were normal, as well as the serological evaluation of thyroid function. Serological evaluations for hepatitis A, B, and C virus, coxsackie virus, Epstein-Barr virus, echovirus, parvovirus B-19, syncytial respiratory virus, CMV, HVH-6, HSV, VZV, influenza A and B virus, toxoplasma, yearsiniae and chlamydia were negative. No evidence of infection was found by serological tests for syphilis, blood cultures, or urine sediment and cultures. Neoplastic markers (CEA, CA-125, CA 15-3, B2 - microglobulin, α-FP, CA 19-9 and NSE) were normal. Findings were normal from an electrocardiogram, radiographs of the chest and affected joints, and from an ultrasound scan of the abdomen and pelvis. Moreover, an upper endoscopy was performed revealing no alterations. The patient had no symptoms that indicated a reason for colonoscopy.
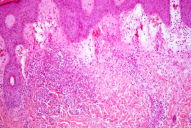
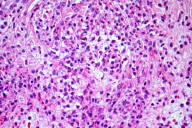
Histopathological evaluation of a skin biopsy showed a dense inflammatory infiltrate of neutrophilis and eosinophils with marked edema in the papillary dermis and leukocytoclasia without signs of vasculitis. These features confirmed the clinically suspected diagnosis of Sweet syndrome (Figs. 3 & 4).
 |  |
| Figure 3 | Figure 4 |
|---|---|
| Figures 3 and 4. Dense inflammatory infiltrate of neutrophils, eosinophils, and marked edema in the papillary dermis (H&E, original magnification x10 and x100) | |
The thyroid ultrasound-scan showed a multinodular thyroid with a normal size and a dominant nodule in the left lobe. A fine-needle aspiration of the nodule was performed and the cytologic examination was consistent with chronic lymphocytic thyroiditis: sheets of follicular cells and oncocytes with a flat arrangement.
The patient was treated with prednisolone at an initial dose of 30 mg daily; rapid improvement of the skin lesions occurred. She was referred to the endocrinology department and was medicated with levothyroxine After 24 months of follow-up, she had no recurrences of the dermatosis. She continues levothyroxine and has normal thyroid function tests.
Discussion
Since the original description, hundreds of patients with acute febrile neutrophilic dermatosis have been reported [1, 2, 3]. The manifestations of this dermatosis in these individuals have not only confirmed those originally described, but also introduced new features that have expanded the clinical and pathologic concepts of this disease [2]. Sweet syndrome may be idiopathic or associated with a variety of systemic disorders that may occur either before, concurrent with, or following the diagnosis [2, 3]. The occurrence of Sweet syndrome may be associated with the development of these conditions.
Hashimoto thyroiditis is an autoimmune thyroid disease that occurs as a result of an immune response directed against the thyroid gland. It is characterized clinically by hypothyroidism and a diffuse goiter. In this disease a wide range of laboratory findings include increased TSH, low T4, hypercholesterolemia, and the presence of thyroid auto-antibodies [4]. Our patient showed antibodies to thyroid peroxidase in high titers, but the concentrations of T3, free T4, and TSH were normal and the thyroid volume was normal. Therefore, we consider that this case corresponds to the early or subclinical phase of Hashimoto thyroiditis. Taking into account that a complete clinical and laboratory investigation did not demonstrate a common possible association, such as hematologic malignancies (most commonly acute myelogenous leukemia), solid tumors (most commonly carcinomas of the genitourinary organs, breast and gastrointestinal tract), inflammatory bowel disease, autoimmune disorders, or medications, we consider that the appearance of Sweet syndrome in this particular patient may be associated with Hashimoto thyroiditis.
Based on a literature search, the association of these two pathologies has been reported only once. In 1993 Nakayama and colleagues documented the occurrence of Sweet syndrome with Takayasu arteritis and Hashimoto thyroiditis in a 39-year-old woman [5]. The pathogenesis of these diseases remains to be definitively determined [3, 4]. Several features suggest that both these conditions result from a hypersensivity reaction to an eliciting antigen; the source of that antigen may be diverse, possibly bacterial or viral [3, 4] although in the present case laboratory investigaton failed to detect any rise in the antibody titer towards several viruses and bacteria. The authors of that report believe that the appearance of these two conditions in the same patient is likely to be mechanistically associated with Sweet syndrome and the immune mediated effects of helper T cell type 1 cytokines (IL1, IFN-γ, TNF-α) may be the link between these disorders [3, 4].
References
1. Sweet RD. Acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;75:349-356. PubMed2. Nakayama H, Shimao S, Hamamoto T, Munemura C, Nakai A. Neutrophylic dermatosis of the face associated with aortitis syndrome and Hashimoto's Thyroiditis. Acta Derm Venereol. 1993; 73: 380-381. PubMed
3. Cohen Philip R, Kurzrock Razelle. Sweet's syndrome revisited: a review of disease concepts. Int J Dermatol. 2003;42: 761-778. PubMed
4. Freedberg I M, Eisen A Z, Wolff K, Austen K F, Goldsmith L A, Katz S I. Acute febrile neutrophilic dermatosis Fitzpatrick's Dermatology in General Medicine. 2003; 1:949-959.
5. Ai J, Leonhart J M, Heymann W R. Autoimmune thyroid diseases: etiology, pathogenesis, and dermatologic manifestations. J Am Acad Dermatol. 2003; 48(5):641-657. PubMed
© 2008 Dermatology Online Journal

