Vasculopathy related to cocaine adulterated with levamisole: A review of the literature
Published Web Location
https://doi.org/10.5070/D36g18r9cvMain Content
Vasculopathy related to cocaine adulterated with levamisole: A review of the literature
Timothy Pearson BS, Matthew Bremmer MD, Jared Cohen BS, Marcia Driscoll MD Pharm D
Dermatology Online Journal 18 (7): 1
University of Maryland, Baltimore, MarylandAbstract
BACKGROUND: Recently, there have been numerous case reports and series describing patients presenting with cutaneous vasculopathy that has been linked to the levamisole frequently found in cocaine. OBJECTIVE: The purpose of this study was to review all published case reports and series of patients reported with cutaneous vasculopathic findings of lemavisole induced vasculopathy (LIV) associated with cocaine use. METHODS: A review of PubMed was performed searching the keywords: levamisole, cocaine, in combination with vasculitis, and vasculopathy. Twenty-two case reports and series were available with sufficient data on reported patients to be included. Four patients from the authors’ clinical experience are included as well. RESULTS: A number of common clinical and pathological findings are reviewed, including lower extremity (46/55 patients, 84%) and ear involvement (40/55 patients, 73%), and positive anti-neutrophil cytoplasmic antibodies (ANCA) findings (p-ANCA 42/48 patients, 88%; anti human neutrophil elastase 11/11 patients, 100%). Similar numbers of patients were treated with systemic corticosteroids as were treated conservatively; there was comparable improvement on follow up. CONCLUSIONS: There are a number of clinical and laboratory findings that are commonly found in patients with LIV. There is currently insufficient data to recommend treatment with systemic corticosteroids in patients with this condition.
Introduction
Levamisole is an antihelminthic and immunomodulatory drug that has been found in seized cocaine in increasing amounts. In 2009, the United States Drug Enforcement Agency estimated that 69 percent of cocaine was contaminated with levamisole [1]. It is believed that levamisole has been increasingly used as a cutting agent because it potentiates the effect of cocaine and remains relatively undetectable to crude practices that test the purity of cocaine [2, 3]. Since the first report of cocaine/levamisole-induced vasculopathy (LIV) in June 2010, multiple case reports and series with similar clinical presentations have been published [4]. Whereas some clinical features of cocaine/levamisole-induced vasculopathy, such as associated neutropenia, classic distribution of cutaneous lesions, and anti-neutrophil cytoplasmic antibody (ANCA) positivity have become readily apparent, other aspects of the clinical picture, such as other organ involvement, are less clear. Appropriate medical management is currently undetermined.
The authors reviewed all available case reports and series with LIV, including 4 of our own cases. The locations of lesions, involved organ systems, abnormal laboratory values, biopsy results including direct immunoflorescence (DIF), treatment, and clinical course were particularly noted. We reviewed 23 published case reports/series, which depicted 51 cases and included 4 cases observed by the authors.
Clinical presentation
It is often difficult to determine the time course relationship between use of cocaine and the onset of LIV. Most available reports did not specify a time delay, likely because this history is frequently difficult to elicit reliably from cocaine using patients. However, multiple reports of patients state that the time of last cocaine use was within 24 hours of hospital admission, indicating that the time to onset may be relatively rapid [5, 6, 7, 8].
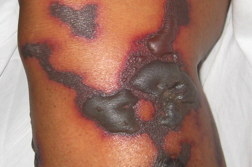
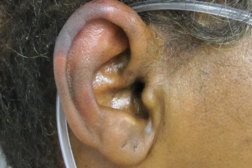
The most commonly involved site is the lower extremity with 46/55 (84%) patients having clinical involvement (Table 1). The most pathognomonic site of involvement is the ear, observed in 40/55 (73%) patients. This site is uncommonly involved in other vasculitis-like clinical presentations. In order, the most commonly involved sites are the lower extremities (46/55 patients), ears (40/55), upper extremities (34/55), face (26/55), trunk (22/55), nose (21/55), and oral region (4/55).
After cutaneous involvement, the most common concomitant clinical complaint is the development of arthralgias (17/55). Involvement of other organ systems at this point is unclear. There is one case series that reported three of five patients with pulmonary findings ranging from hypersensitivity pneumonitis to pulmonary nodules [9]. Another case reported acute renal injury, which was thought likely to be due to levamisole [10].
Laboratory and histological findings
Neutropenia is a well-recognized adverse response to levamisole. Of our 52 patients, 31 (60%) had associated neutropenia (Table 2). Patients also tended to have ANCA abnormalities. The most frequent was a high level of perinuclear ANCA (p-ANCA), found to be elevated in 42 of 48 patients (88%). Cytoplasmic ANCA (c-ANCA) was elevated in 10 of 48 patients (21%). Anti-myeloperoxidase (anti-MPO) was elevated in 25 of 41 patients and anti-proteinase-3 (anti-PR3) in 26 of 42, usually concomitantly (see Table 2). ANA was elevated in 24 of 47 (51%) patients. Of note, human neutrophil elastase antibodies (HNE) were elevated in 11 of 11 patients.
Biopsy results were available in 50 patients. The pathologic diagnoses were categorized as a thrombotic vasculopathy, vasculitis, or a combination of thrombosis with vasculitis. Biopsy revealed thrombosis in 24 patients (48%), vasculitis with no noted evidence of thrombosis in 8 patients (16%), and a combination of thrombosis with vasculitis in 18 patients (36%).
Direct immunoflorescence was not routinely performed. Of available results, the most common appears to be vascular deposition of IgM, IgA, and C3, which were reported as positive in 3 of 4 patients [6, 11].
Treatment and outcome
Treatment of patients was specified in 43 cases (See Table 3). Of these, 25/43 (58%) were treated with systemic corticosteroids, whereas 15/43 (35%) were treated conservatively with wound management and antibiotics when deemed appropriate. Five of the patients required surgical management, either with skin grafting or with amputation in two of the patients [12, UMMC cases]. Two patients were treated with systemic corticosteroids in combination with cyclophosphamide [9]. One patient was treated with methotrexate in combination with corticosteroids [33]. One patient was treated with colchicine [8].
Some data concerning clinical course was available in 35 patients. Nine patients (26%) experienced rapid resolution (defined as less than 2 weeks or stated as rapid by the article); 22 patients (63%) experienced improvement; 4 patients (11%) failed to heal. Of the rapidly resolving patients, 5 of 9 (56%) were given systemic corticosteroids and 4 (44%) were treated conservatively. Of patients who experienced improvement, 13 of 22 (59%) were given systemic steroids, 8 (36%) were treated conservatively, and in one patient treatment was not reported. In patients who failed to heal, one patient was treated with systemic corticosteroids and the other three were treated conservatively. In one of the patients who failed to improve ongoing cocaine abuse was implicated [2].
Discussion
Levamisole-induced vasculopathy is an emerging condition. As more cases are reported, the clinical aspects of the condition are becoming clearer. Certainly the prevalence of levamisole in cocaine users is high; a study of 300 urine toxicology samples positive for cocaine by immunoassay showed that 203 of these samples (68%) were also positive for levamisole by gas chromatography [13]. The short half-life of levamisole (5.6 hours) limits the utility of detection of this substance in specifying the cause of cutaneous vasculopathy. In a patient known to have used cocaine, detection of levamisole should not be considered essential for diagnosis [4].
Fortunately, the clinical presentation of LIV has a relatively unique clinical picture. Whereas the most common site of presentation is the lower extremity with 84 percent of patients having involvement of this site, 73 percent of patients had involvement of the ears, which is less commonly involved in vascular conditions (see images 1 and 2). It is of importance to consider other diagnoses in patients who present with a florid vasculopathic clinical picture. In particular, the possibility of septic emboli and cryoglobulinemia, which can both lead to similar purpuric lesions in extremities, should be considered. Whereas the combination of ear involvement with the frequently large areas of involvement on the lower extremities, along with typical clinical history indicates a high likelihood of LIV, appropriate culture and laboratory workup are indicated to rule out other conditions.
The utility of laboratory evaluation at this point remains unclear. Whereas the majority of patients with LIV are positive for p-ANCA (88%) and approximately two thirds are positive for MPO and PR3, these values are frequently positive in other vasculitic conditions. Our results are similar to results found in a review of 30 patients with ANCA positivity and cocaine exposure from Massachusets General Hospital. All of these patients were positive for MPO antibodies, and 50 percent had PR3 antibodies [14]. This study also notes that MPO titers were up to 15 times higher than in their patients with idiopathic ANCA-associated vasculitis, indicating high MPO titers may be seen in LIV. C- and p-ANCAs were not available in their review. Although not specific for the condition, positivity of p-ANCA, MPO, or PR3 antibodies is consistent with LIV. Additionally, the finding of multiple ANCA positivity is a strong indication of a drug-induced vasculopathy as opposed to other primary vasculitides [14].
Human neutrophil elastase antibodies may have a significant role in the identification of LIV. In our review, 11 of 11 patients evaluated for this antibody were positive. Whether or not positivity for HNE is related to levamisole exposure or cocaine exposure is unclear. In 2004 a study of HNE in correlation with cocaine-induced midline destructive lesions (CIMDL) found that 84 percent of 25 patients with CIMDL were HNE positive, as compared with 1.3 percent of 604 controls [15]. Importantly, among controls, 64 of the patients had Wegener granulomatosis and 14 had microscopic polyangiitis; none of these patients had HNE positivity. It is of note that these samples were collected between 1991 and 2001, before the first report of LIV associated with cocaine use. Additionally, CIMDL has not been associated with LIV. It is therefore more likely that positivity for HNE is an indication of the cocaine use associated with LIV. Importantly, the aforementioned study strongly indicates that detection of HNE antibodies would make other systemic vasculitides less likely.
Neutropenia has been recognized as a complication of levamisole therapy because it was used as a primary immunomodulatory agent, with rates of agranulocytosis ranging from 2.5 to 13 percent when used for long periods [16]. In our review, 60 percent of patients had documented neutropenia associated with their cutaneous findings. Although neutropenia is an expected and well-recognized association with LIV, it is not necessary to make the diagnosis, nor is it an inevitable consequence of levamisole exposure. In a recent review of 95 patients who were cocaine positive and underwent a comprehensive toxicology test with an available CBC, the incidence of neutropenia was 4.2 percent in 47 levamisole positive patients and 2.1 percent in levamisole negative patients [17].
During the time that levamisole was used in a medical context, the vasculopathy that occurred had variable pathologic features. In a case series of levamisole vasculopathy in children from 1999, biopsy findings ranged from leukocytoclastic and thrombotic vasculitis to vascular occlusion without true vasculitis [18]. In our review, 48 percent of biopsies did not observe vasculitis. Thrombosis with vasculitis was observed in 36 percent and vasculitis without thrombosis was seen in only 16 percent. This suggests that the majority of cases are a primarily occlusive/thrombotic process leading to secondary inflammatory changes. This may be a key finding because it has implications regarding the currently observed lack of striking benefit seen with the administration of systemic corticosteroids in the management of this condition. However, it also has implications regarding the potential use of anticoagulants in the management of this condition. To date, this is not a therapeutic modality that has been reported, but may be worth consideration if the condition is found early.
There are, however, indications that this is an inflammatory condition. C-reactive protein (CRP) was elevated in 66 percent of reviewed patients. It has been noted that ANCAs are frequently elevated. Clinically, arthralgias are seen to be present in 31 percent of patients. One case report includes a patient who had joint, cutaneous, and pulmonary symptoms,who flared upon the attempt to taper prednisone [9]. Another patient’s ANCA titers tended to increase when the patient subjected herself to further cocaine use [9].
The pathogenesis of this condition is not clear. We know from prior medical reports [18] of patients using unadulterated levamisole that it is capable of producing these lesions on its own. It would be logical that the vasoconstrictive properties of cocaine exacerbate the condition. Regarding a possible inflammatory component to the condition, levamisole has been shown to have number of immunomodulating effects, including stimulation of neutrophil chemotaxis [9]. The exact influence of these effects on the in vivo manifestations of the condition are yet to be elucidated.
Beyond the obvious discontinuation of levamisole-contaminated cocaine, appropriate management of levamisole-induced vasculopathy has yet to be defined. Despite evidence that there is frequently an inflammatory component to this condition, current evidence suggests that patients tend to improve regardless of whether or not systemic corticosteroids are used. Of the 35 reviewed patients with whom follow up was available, 31 out of 35 of these patients experienced improvement or resolution. Of these improved or resolved patients, 60 percent were treated with systemic corticosteroids and 40 percent were treated with wound care and antibiotics as necessary. Whereas these numbers are subject to biases including under reporting and non-randomization, it indicates that currently there is a lack of evidence that systemic corticosteroids modify the clinical course of LIV.
Although numbers do not support the routine use of corticosteroids, in individual patients with striking signs of inflammation, corticosteroids might be considered. If a patient presents with a debilitating arthropathy, a strikingly elevated CRP, or a biopsy showing vasculitis without evidence of thrombosis, it is theoretically possible that systemic corticosteroids may improve their outcome.
Rare clinical findings in LIV include renal and pulmonary involvement. In the case of a patient with suspected renal involvement, the patient had “noted a 2-day history of darkened urine and diminished urine output” and was subsequently found to have a creatinine of 2.8 on admission, which subsequently returned to normal values by discharge [10]. In the case series of ANCA positive patients, two patients were reported to have acute renal injury, which may have been related to levamisole [14].
A few cases of pulmonary involvement have been reported, including three patients in a case series by Ullrich et al, who exhibited pulmonary findings that ranged from “bronchitis and bronchiolitis” to “desquamative interstitial pneumonia and hypersensitivity pneumonitis” and “pulmonary nodules” [9]. In the case series of ANCA positive patients, 4 of 18 patients with clinical and laboratory data available had pulmonary disease, including two of the four with pulmonary hemorrhage [14].
An additional, theoretical concern with levamisole that has yet to be seen clinically is that of the conversion of levamisole in vivo to aminorex and the subsequent development of idiopathic pulmonary hypertension (IPH). Aminorex is a drug that has in the past been marketed in Europe to promote weight loss. In countries in which it was available, it has been responsible for a small epidemic of pulmonary hypertension [19]. Additionally, it has been shown that levamisole is converted to aminorex in humans [20]. It is yet to be shown whether or not the degree of conversion in cocaine users is sufficient to produce cases of IPH associated with LIV or cocaine use.
In conclusion, there are a number of clinical and laboratory findings that support a diagnosis of LIV, including ear involvement, ANCA positivity, and anti-HNE antibodies that help to distinguish this condition from autoimmune-type vasculitis. There is currently no firm evidence that systemic corticosteroids or any treatment beyond supportive care and cessation of cocaine use is indicated in treatment. However, in patients with indications of systemic inflammation, corticosteroids may be appropriate.
References
1. Centers for Disease Control and Prevention (CDC). Agranulocytosis associated with cocaine use - four States, March 2008-November 2009. MMWR Morb. Mortal Wkly. Rep. 58, 1381-1385 (2009). [PubMed]2. Walsh NM. Green PJ, Burlingame RW, Pasternak S, Hanly JG. Cocaine-related retiform purpura: evidence to incriminate the adulterant. J Cutan Pathol. 2010 Dec;37(12):1212-9. [PubMed]
3. A Chang, J Osterloh, J Thomas. Levamisole, a dangerous new cocaine adulterant. Clin Pharmacol Ther. 2010; 88: 408-1. [PubMed]
4. Bradford M, Rosenberg B, Moreno J, Dumyati G. Bilateral Necrosis of Earlobes and Cheeks: Another Complication of Cocaine Contaminated With Levamisole. Ann Intern Med. 2010 Jun 1;152(11):758-9. [PubMed]
5. Reutemann P, Grenier N, Telang GH. Occlusive vasculopathy with vascular and skin necrosis secondary to smoking crack cocaine. J Am Acad Dermatol. 2011 May;64(5):1004-6. [PubMed]
6. Jenkins J, Babu K, Hsu-Hung E, Robinson-Bostom L, Kroumpouzos G. ANCA-positive necrotizing vasculitis and thrombotic vasculopathy induced by levamisole-adulterated cocaine: A distinctive clinicopathologic presentation. J Am Acad Dermatol 2011; 65: e14-6. [PubMed]
7. Dy I, Pokuri V, Olichney J, Wiernik P. Levamisole-adulterated in cocaine causing agranulocytosis, vasculopathy, and acquired protein S deficiency. Ann Hematol 2012; 91(3): 477-8. [PubMed]
8. Poon SH, Baliog CR Jr, Sams RN, Robinson-Bostom L, Telang GH, Reginato AM. Syndrome of Cocaine-Levamisole-Induced Cutaneous Vasculitis and Immune-Mediated Leukopenia. Semin Arthritis Rheum. 2011 Aug 23. [PubMed]
9. Ullrich K, Koval R, Koval E, Bapoje S, Hirsh JM. Five consecutive cases of a cutaneous vasculopathy in users of levamisole-adulterated cocaine. J Clin Rheumatol 2011; 17: 193-6. [PubMed]
10. Zwang NA, Van Wagner LB, Rose S. A case of levamisole-induced systemic vasculitis and cocaine-induced midline destructive lesion: a case report. J Clin Rheumatol. 2011 Jun;17(4):197-200. [PubMed]
11. Chung C, Tumeh PC, Birnbaum R et al. Characteristic purpura of the ears, vasculitis, and neutropenia-a potential public health epidemic associated with levamisole-adulterated cocaine. J Am Acad Dermatol 2011 Oct; 65(4): 722-5. [PubMed]
12. Ching JA, Smith DJ Jr. Levamisole-Induced Necrosis of Skin, Soft Tissue, and Bone: Case Report and Review of Literature. J Burn Care Res. 2011 Nov 10. [PubMed]
13. Buchanan JA, Heard K, Burbach C, Wilson ML, Dart R. Prevalence of levamisole in urine toxicology screens positive for cocaine in an inner-city hospital. JAMA 2011; 305: 1657-8. [PubMed]
14. McGrath MM, Isakova T, Rennke HG, Mottola AM, Laliberte KA, Niles JL. Contaminated cocaine and antineutrophil cytoplasmic antibody-associated disease. Clin J Am Soc Nephrol 2011; 6: 2799-805. [PubMed]
15. Wiesner O, Russell KA, Lee AS, et al. Antineutrophil cytoplasmic antibodies reacting with human neutrophil elastase as a diagnostic marker for cocaine-induced midline destructive lesions but not autoimmune vasculitis. Arthritis Rheum 2004; 50: 2954-65. [PubMed]
16. Thompson JS, Herbick JM, Klassen LW et al. Studies on levamisole-induced agranulocystsis. Blood 1980; 56: 388-96. [PubMed]
17. Chai PR, Bastan W, Machan J, Hack JB, Babu KM. Levamisole exposure and hematologic indices in cocaine users. Acad Emerg Med 2011; 18: 1141-7. [PubMed]
18. Rongioletti F, Ghio L, Ginevri F, Bleidl D, et al. Purpura of the ears: a distinctive vasculopathy with circulating autoantibodies complicating long-term treatment with levamisole in children. Br J Dermatol 1999;140: 948-51. [PubMed]
19. Karch SB, Mari F, Bartolini V, Bertol E. Aminorex poisioning in cocaine abusers. Int J Cardiol 2011 Jul 15. [PubMed]
20. Bertol E, Mari F, Milia MG, Politi L, Furlanetto S, Karch SB. Determination of aminorex in human urine samples by GC-MS after use of levamisole. J Pharm Biomed Anal 2011; 55: 1186-9. [PubMed]
21. Jacob RS, Silva CY, Powers JG, et al. Levamisole-Induced Vasculopathy: A Report of 2 Cases and a Novel Histopathologic Finding. Am J Dermatopathol 2011 Nov 15. [PubMed]
22. Fthenakis A, Klein PA. Retiform purpura in a patient with a history of cocaine use. Dermatol Online J 2011; 17: 12. [PubMed]
23. Mouzakis J, Somboonwit C, Lakshmi S et al. Levamisole induced necrosis of the skin and neutropenia following intranasal cocaine use: a newly recognized syndrome. J Drugs Dermatol 2011; 10: 1204-7. [PubMed]
24. Gross RL, Brucker J, Bahce-Altuntas A, et al. A novel cutaneous vasculitis syndrome induced by levamisole-contaminated cocaine. Clin Rheumatol 2011 Oct ; 30(10): 1385-92. [PubMed]
25. Click J. Levamisole-induced Retiform Purpura. J Drugs Dermatol 2011; 10: 217. [PubMed]
26. Farhat EK, Muirhead TT, Chaffins ML, Douglass MC.Levamisole-induced cutaneous necrosis mimicking coagulopathy. Arch Dermatol. 2010; 146: 1320-1. [PubMed]
27. Geller L, Whang TB, Mercer SE, Phelps R. Retiform purpura: A new stigmata of illicit drug use? Dermatol Online J 2011; 17: 7. [PubMed]
28. John S, Manda S, Hamrock D. Cocaine-induced thrombotic vasculopathy. Am J Med Sci 2011; 342: 524-6. [PubMed]
29. Waller Jm, Feramisco JD, Alberta-Wszolek L, McCalmont TH, Fox LP. Cocaine-associated retiform purpura and neutropenia: Is levamisole the culprit? J Am Acad Dermatol 2010 ; 63:530-5. Epub 2010 Mar 20. [PubMed]
30. Graf J, Lynch K, Yeh CL, Tarter L, Richman N, Nguyen T, Kral A, Dominy S, Imboden J. Purpura, cutaneous necrosis, and antineutrophil cytoplasmic antibodies associated with levamisole-adulterated cocaine. Arthritis Rheum 2011; 63: 3998-4001. [PubMed]
31. Han C, Sreenivasan G, Dutz JP. Reversible retiform purpura: a sign of cocaine use. CMAJ 2011 14; 183: E597-600. Epub 2011 Mar 14. [PubMed]
32. de la Hera I, Sanz V, Cullen D, Chico R, Petiti G, Villar M, Burgués M, Sanz J, Castellanos M, Zarco O, Ortiz PL, Rodríguez-Peralto JL, Vanaclocha F. Necrosis of ears after use of cocaine probably adulterated with levamisole. Dermatology 2011; 223: 25-8. Epub 2011. [PubMed]
33. Khan TA, Cuchacovich R, Espinoza LR et al. Vasculopathy, hematological, and immune abnormalities associated with levamisole-contaminated cocaine use. Semin Arthritis Rheum 2011; 41: 445-54. [PubMed]
© 2012 Dermatology Online Journal