Oral manifestations of pachyonychia congenita
Published Web Location
https://doi.org/10.5070/D36f5659xvMain Content
Oral manifestations of pachyonychia congenita
Paulo Sérgio da Silva Santos1, Francesco Mannarino2, Rute F Lellis3, Luis Henrique Osório2
Dermatology Online Journal 16 (10): 3
1. Bauru School of Dentistry, University of São Paulo, Brazil. paulosss@fob.usp.br2. Oral and Maxillofacial Surgery of Hospital Santa Casa of São Paulo
3. Department of Pathology of Hospital Santa Casa of São Paulo
Abstract
Pachyonychia congenita is a rare genetic disorder characterized mainly by hypertrophy of the nails and hyperkeratosis of the skin and mucosae. Fifty percent of all patients have oral leukokeratosis, which is often painful. The case reported here is of a 41-year-old patient who had white lesions in the form of irregular plaques; these affected multiple regions of the oral mucosa and were sensitive to touch. Histological examination revealed acanthosis, parakeratosis and ballooning of the epithelial cells, consistent with oral leukokeratosis. After therapy including topical steroids and prosthetic rehabilitation, the symptoms resolved.
Introduction
Pachyonychia congenita (PC) is a rare group of genodermatosis usually inherited as an autosomal dominant trait. The first description of the disease was in 1906, by Jadassohn-Lewandowsky [1] and since then approximately 250 cases have been reported [2]. There are several known types of PC manifestation and usually there is correlation between the mutated gene and the type of disease [3]. According to Schonfeld’s classification (1980), PC is divided into three types. Hyperkeratosis, severe curvature of the nail bed, distal onycholysis, and nail color changes to varying degrees are present in all types.
In type I (Jadassohn-Lewandowsky), hyperkeratosis is found on the palms and soles, the knees, and the elbows. There is also follicular keratosis and leukokeratosis of mucosae in the mouth, anus, and upper airways, as well as the larynx in some cases. In type II (Jackson-Lawler), the keratoderma and changes in the mouth are less marked or absent. However, neonatal teeth and multiple epidermoid cysts are common findings. In type III (Schafer-Brunauer), in addition to the described characteristics in type I, corneal leukokeratosis is also common. There is also a fourth type in which the clinical manifestations appear only during the second or third decade of life, which is also called late-onset PC [4]. Half of the patients affected by PC present with an oral manifestation and they are most commonly associated with type I PC [5]. This paper reports the case of a patient with type I PC with manifestation in the oral mucosa.
Case report
A 41-year-old woman complained of thickening of the fingernails and toenails associated with pain. When she was a 6-year-old she noticed these changes, and from the age of 13 had sought treatment from her dermatologist. Her family history revealed that her father and younger sister have similar changes in the toenails. She reported being a smoker for 25 years. She also reported undergoing seven nail correction plastic surgeries, with initial improvement but with later recurrence of the lesions. She also underwent excision of the epidermoid cyst in the nasolabial sulcus, without recurrence. She complained that the oral lesions had been painful for the previous three months.
 |  |
| Figure 1 | Figure 2 |
|---|---|
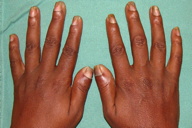
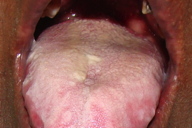
| Figure 1. The nails of the hands had severe curvature and thickness, subungual hyperkeratosis and a darkening in color. Figure 2. White hyperkeratotic plaques in the oral mucosa | |
 |  |
| Figure 3 | Figure 4 |
|---|---|
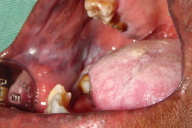
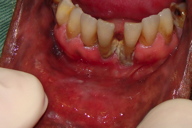
| Figure 3. White hyperkeratotic plaques in the retro molar region and right alveolar bone Figure 4. White hyperkeratotic plaques in the left lateral border of the tongue | |
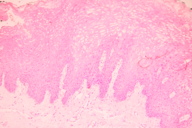 |
| Figure 5 |
|---|
| Figure 5. Acanthosis, parakeratosis and ballooning of epithelial cells (H&E) |
On physical examination, rounded palmoplantar hyperkeratosis, especially in the areas of greatest friction, was noted and the patient experienced difficulty in walking with the help of crutches. Hyperkeratotic plaques on the knees and elbows were also noted, as well as verrucous and vegetating plaques on both sides of the heels; perianal leukoplakia was also observed and this was pruritic. The nails of the hands and feet had associated deformities such as severe curvature and thickness of the nail bed, subungual hyperkeratosis, and a darkening in color (Figure 1). Oral examination revealed white hyperkeratotic plaques in the oral mucosa, retro molar region, right alveolar bone, left lateral border of the tongue, and lower lip (Figures 2, 3, and 4). It was not possible to scrape off the lesions and they were sensitive to touch.
A biopsy was performed to collect samples of the white hyperkeratotic plaque on the right buccal mucosa. Histopathology examination demonstrated acanthosis, parakeratosis, and ballooning of epithelial cells by sections stained with hematoxylin and eosin staining (Figure 5).
At the first consultation, the patient also revealed a malocclusion because of lack of teeth, which caused a deep bite that caused recurrent trauma to the alveolar mucosa. Prosthetic rehabilitation with removable partial dentures was then carried out. The cast models were mounted on a semi-adjustable articulator and the ideal vertical dimension was set in order to rehabilitate the patient’s function and eliminate the trauma caused by the malocclusion.
The patient is currently being treated on an outpatient basis with topical 10 percent urea and salicylated petroleum jelly on the skin lesions, which helps to reduce the pain. She has been advised to quit smoking. For the painful lesions of the oral mucosa, the patient was advised to use a three-times-a-day mouthwash with corticosteroid (0.01% dexamethasone elixir), which has significantly improved local sensitivity. Oral lesions are being clinically monitored twice per year and annual biopsies are performed. No malignancy was evident up until the time of publication of this paper.
Discussion
The oral manifestations of PC, including neonatal teeth and oral leukokeratosis, are often the first signs of the disease and may occur before the onset of typical changes in the nails [6].
Su et al. [2], through a retrospective analysis, assessed 12 patients in 10 families with PC, 10 of whom had oral leukokeratosis. In a recent study, Leachman et al. [5] reported that, of 57 patients in 41 families affected by PC, 75 percent had oral leukokeratosis. Although the authors describe a high incidence of oral manifestations, a literature review found that only eight cases described the oral changes (Table 1).
The pain in the leukokeratotic oral lesions in patients affected by PC often causes high morbidity; it manifests in relation to the chronic trauma caused by solid food [5, 12]. Usually, oral lesions have a striated hyperkeratotic aspect, involving only a portion of the cheek mucosa and/or the tongue or extending throughout the oral mucosa, including the lips, the retromolar region, and palate [2]. In our report, the oral mucosa lesions were painful and multiple, and involved the retromolar regions, the alveolar bone mucosa, the lateral border of the tongue, the cheek mucosa and the lower lip. However, the patient reported that the pain was not related to food intake.
Clinically, white lesions may resemble oral candidiasis, white sponge nevus, hairy tongue, and leukoplakia. Malignant transformation has not previously been found in leukokeratosis [2, 5]. The association between PC and candidiasis has been described by several authors [2, 10, 13]. Karen and Schaffer [14] showed an association between PC and median rhomboid glossitis. In our case, the possibility of associated fungal infection was ruled out because of the absence of clinical signs and laboratory findings.
Histopathological examination showing surface changes such as acanthosis, parakeratosis, and ballooning of epithelial cells is in agreement with other previous reports [2, 5, 6, 11].
Keratin mutations have been identified as the cause of a number of skin fragility and hyperkeratotic disorders. The CK6 and CK16 cytokeratins are considered hyperproliferative. They are normally found in the plant epidermis, hairy areas, nails, and non-keratinized stratified epithelium. When an injury occurs in the mucosa and skin, keratinocytes that express CK6 and CK16 migrate to the affected site; these cytokeratins may play a role in allowing keratinocytes to have sufficient pliability and plasticity to migrate and accomplish reepithelialization. At the molecular level it has been demonstrated that mutations in keratin K16 K6a causes PC-1. Although these cytokeratins are found in squamous cell carcinoma, there is no confirmation of this relationship between pachyonychia and malignant neoplasms [15, 16]. The clinical and histological features arising from these mutations were observed in this case report.
The oral rehabilitation of patients affected by PC should be directed to eliminate the possibility of chronic trauma to the oral mucosa. Despite the importance of oral rehabilitation, there are no reports of prosthetic devices for these patients. In this report, the prosthesis underwent a long cycle polymerization to reduce the amount of residual free monomer, which would be an irritant to the mucosa. The edges of the prosthesis and the cusps of the teeth were constructed to be of uniform thickness and rounded to avoid any further trauma to the tissues.
To date there is no specific treatment for PC, but symptomatic support, such as the use of systemic retinoid therapy, has been very well accepted by patients and useful in controlling oral leukokeratosis [12]. However, conventional or high power laser surgical excision has been reported as suitable for these lesions [17]. In our study, topical corticosteroids were used with complete regression of pain in the oral lesions. Bearing in mind that the patient has risk factors for oral cancer by being a smoker, both our team and the patient are worried about malignant transformation of the leukokeratosis. Thus, regular consultations and biopsies are being carried out.
When there is a family history of the same condition, periodic oral examination is important both for early diagnosis and symptomatic treatment of oral lesions in PC. We recommend the use of topical steroids for symptomatic oral lesions in PC patients because successful reduction of pain was achieved in this case report. Because there is such a lack of studies that address the treatment of oral lesions in PC, more studies must be undertaken in order to improve the quality of life of these patients.
References
1. Jadassohn J, Lewandowsky F. Pachyonychia congenita. In: Neisser A, Jacoby E. Ikonographia dermatologica. Berlin: Urban and Schwarzenberg, 1906: 29-31.2. Su WP, Chun S, Hammond DE, Gordon H. Pachyonychia congenita: a clinical study of 12 cases and review of the literature. Pediatr Dermatol 1990; 7: 33-8. [PubMed]
3. McLean WH, Smith FJ, Cassidy AJ. Insights into genotype-phenotype correlation in pachyonychia congenita from the human intermediate filament mutation database. J Investig Dermatol Symp Proc 2005; 10: 31-6. [PubMed]
4. Paller A, Moore J, Scher R. Pachyonychia congenita tarda. Arch Dermatol 1991; 127: 701-3. [PubMed]
5. Leachman AS, Kaspar RL, Fleckman P, Florell SR, Smith FJ, McLean WH, et al. Clinical and pathological features of pachyonychia congenita. J Investig Dermatol Symp Proc 2005; 10: 3. [PubMed]
6. Anneroth G, Isacsson G, Lagerholm B, Lindvall AM, Thyresson N. Pachyonychia congenita. A clinical, histological and microradiographic study with special reference to oral manifestations. Acta Derm Venereol 1975; 55: 387-94. [PubMed]
7. Haber R, Rose T. Autosomal recessive pachyonychia congenita. Arch Dermatol 1986; 122: 919-23. [PubMed]
8. Wimmershoff MB, Stolz W, Schiffner R, Landthaler M. Type I pachyonychia congenita. Klin Pediatrc 1999; 211:179-83. [PubMed]
9. Lim TW, Paik JH, Kim NI. A case of pachyonychia congenita with oral leukoplakia and steatocystoma multiplex. J Dermatol 1999; 26: 677-81. [PubMed]
10. Hannaford RS, Stapleton K. Pachyonychia congenita tarda. Austr J Dermatol 2000; 41: 175-77. [PubMed]
11. Vucićević-Boras , Kotrulja L, Cekic A, Pirkic A, Vucic M. Pachyonychia congênita. Case report. Minerva Stomatol 2005; 54: 691-4. [PubMed]
12. Lugo AJR. Congenital pachyonychia treated by oral retinoid. Med Cutan Ibero Lat Am 1982; 10: 395-8.
13. Rohold A, Brandrup F. Pachyonychia congenita: Therapeutic and immunologic aspects. Pediatr Dermatol 1990; 7: 307-9. [PubMed]
14. Karen JK, Schaffer JV. Pachyonychia congenita associated with median rhomboid glossitis. Dermatol online J 2007;13:21. [PubMed]
15. Liao H, Sayers JM, Wilson NJ, et al. A spectrum of mutations in keratins K6a, K16 and K17 causing pachyonychia congenita.J Dermatol Sci 2007;48(3):199-205.
16. Moll R, Divo M, Langbein L. The human keratins: biology and pathology. Histochem Cell Biol 2008;129(6):705-33.
17. Vaccaro M, Guarneri F, Barbuzza O, Guarneri C. Pachyonychia congenita tarda affecting only the nails. Dermatol online J 2007; 14: 12. [PubMed]
© 2010 Dermatology Online Journal

