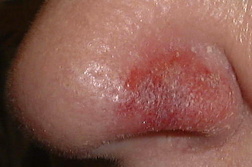
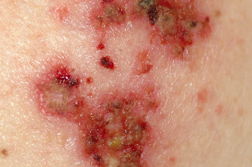
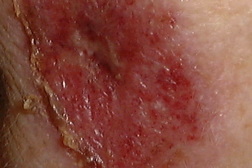
Local reactions to imiquimod in the treatment of basal cell carcinoma
Published Web Location
https://doi.org/10.5070/D36dm013fjMain Content
Local reactions to imiquimod in the treatment of basal cell carcinoma
Yasmeen Tandon1 MD, Robert Thomas Brodell2,3 MD
Dermatology Online Journal 18 (9): 1
1. Case Western Reserve University-Metro Health, Cleveland, Ohio2. University of Mississippi Medical Center, Jackson, Mississippi
3. University of Rochester School of Medicine and Dentistry, Rochester, New York
Abstract
Basal cell carcinoma (BCC) is the most common form of skin cancer. The most commonly utilized surgical therapies for BCC are curettage and electrodesiccation (E and C) and surgical excision. Whereas surgical modalities have acceptable levels of morbidity and a high cure rate, effective non-invasive topical medical treatments of BCC are of great interest. Imiquimod is FDA approved for the treatment of superficial BCC (sBCC), actinic keratoses, and genital warts. There are several situations in which imiquimod is commonly utilized. Patients with multiple or large sBCCs may wish to avoid surgical approaches more likely to be complicated by scarring. We have also found imiquimod to be useful in elderly patients with marginal involvement of nodular BCC when the patient wishes to avoid additional surgery. Imiquimod is also useful when sBCC recurs following surgical treatments such as electrodessication and currettage and when sBCC occurs in areas that heal poorly after surgery. It is essential for physicians to explain to patients that an exuberant skin reaction is a positive indication of treatment success. Using clinical images to demonstrate the range of possible local reactions may be more effective than verbal descriptions. This minimizes the potential for premature discontinuation of therapy by patients.
Introduction
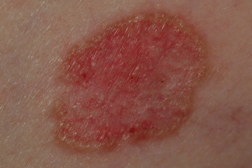 |
| Figure 1 |
|---|
| Figure 1. An example of classic basal cell carcinoma |
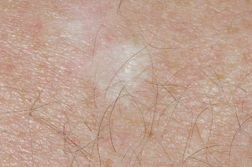
Basal cell carcinoma (BCC) is the most common form of skin cancer and accounts for approximately 80 percent of non-melanoma skin cancer [1]. According to the American Cancer Society, approximately 3.5 million basal and squamous cell skin cancers are diagnosed each year. Most of these are basal cell carcinomas. The exact number of basal and squamous cell skin cancers each year is unknown because these skin cancers are not reported to cancer registries [2]. BCC can be locally invasive but it has a low metastatic potential [1]. Red or blond hair, light-colored eyes, light skin, exposure to UV radiation, are all characteristics associated with an increased risk of BCC [3]. Basal cell carcinoma subtypes include nodular, morpheaform, superficial, infiltrative, cystic, and basosquamous forms. Nodular basal cell carcinomas appear as pearly nodules, which are often ulcerated or crusted and are often described as the “sore that will not heal.” Superficial basal cell carcinoma (sBCC) presents as an erythematous patch that is slightly scaling and well demarcated (Figure 1). Morpheaform basal cell carcinomas are ill defined and typically present with a scar-like appearance. Occasionally, basal cell carcinomas may be pigmented and can resemble malignant melanoma or seborrheic keratosis depending on the degree of pigmentation [4, 5].
Commonly utilized surgical therapies for BCC include electrodesiccation and currettage (E and C), and surgical excision [6, 7]. E and C is often selected by dermatologists for low-risk lesions. It is generally avoided in morpheaform or recurrent tumors, especially in those located at high-risk facial sites [6, 7]. Surgical excision is an excellent treatment modality that permits histopathologic margin assessment. Other treatment modalities include cryotherapy which may be utilized in the setting of multiple BCCs as an alternative to cold steel surgery. This modality, however, is less effective in large or recurrent lesions. For difficult-to-treat BCC, Mohs micrographic surgery results in high cure rates, but this modality is more time consuming and expensive [6, 7, 8].
Considering the effectiveness, cost, final cosmetic appearance, and associated morbidity, no single treatment is clearly superior [8]. Whereas surgical modalities have acceptable levels of morbidity and a high cure rate, effective non-invasive topical medical treatments of BCC are of great interest to both physicians and their patients. 5-fluorouricil 5 percent cream can be used effectively for actinic keratoses and superficial basal cell carcinoma [9]. Imiquimod 5 percent cream (1-(2-methylpropyl)-1H-imidazol [4, 5-c] quinolin-4-amin) (trade name Aldara®), a low-molecular-weight synthetic compound, was more recently approved by the United States Food and Drug Administration for sBCC, actinic keratoses, and genital warts. It is a potent inducer of interferon alfa in vivo and has potent antitumor and antiviral activity [10, 11, 12]. Recently, 3.75 percent imiquimod cream (Zyclara®) was approved by the FDA in the United States for the treatment of actinic keratoses.
Mechanism of action
Imiquimod belongs to a novel class of topical immune response modifiers. Although the mechanism of action is not fully understood, it involves the stimulation of both innate and cell-mediated immune pathways. It causes the induction of several different cytokines. These cytokines include tumor necrosis factor-α, interferon-α, and interleukin-12 from macrophages and monocytes after it binds to Toll-Like receptor-7 [13, 14]. Additionally, imiquimod stimulates other aspects of the innate immune response, including natural killer cell activity [15]. It is believed that through the induction of interferon-α, imiquimod could augment antigen presentation. This is accomplished by increasing the expression of class 1 major histocompatibility complex (MHC) proteins. In addition, interleukin-12 enhances the development of a Th1-type immune response. Improved antigen processing and presentation results from the maturation and migration of Langerhans cells [16].
Dosing ranges
A variety of dosing schedules have been recommended. The most promising dosing regimens for sBCC have been the 5 times per week and the 7 times per week regimens. Studies have evaluated regimens that are 6 and 12 week in duration and one utilized occlusion [17, 18, 19]. A 6-week treatment period may be sufficient duration for treatment because it has been observed that clearance rates for sBCC following therapy with imiquimod 5 percent cream using a 7-day per week regimen were comparable for 6 and 12 week duration. Results from a phase 3 randomized, vehicle-controlled study demonstrated histological clearance of 82 percent in sBCC lesions treated with imiquimod 5 percent cream five times per week over a period of 6 weeks and 79 percent in seven day per week dosing. These authors recommended a 5 times per week regimen for 6 weeks, which we have adopted in our practice [19].
Indications
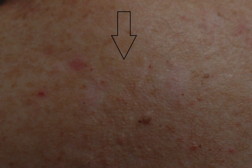 | 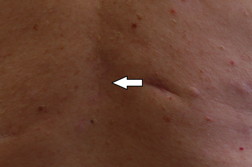 |
| Figure 2c | Figure 2d |
|---|---|
| Figure 2c. Upper mid back lesion (black arrow) 2 months after treatment completed Figure 2d. Lower mid back lesion (white arrow) 2 months after treatment | |
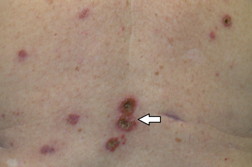
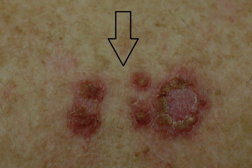
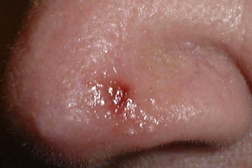
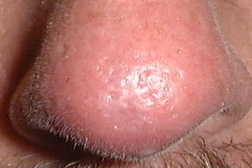
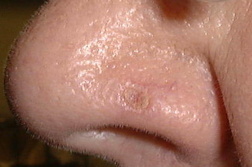
There are several clinical situations in which imiquimod is commonly utilized. Patients with multiple or large sBCCs may wish to avoid surgical approaches more likely to be complicated by scarring (Figures 2a, 2b, 2c, 2d, 3a, 3b, 4a, and 4b). It is also utilized in patients with recurrent superficial basal cell carcinoma following surgery in the distant past (Figures 5a, 5b, and 5c). Excellent cosmetic results can be achieved with this modality.
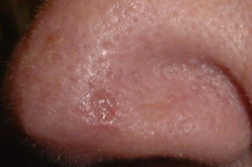
We have also found imiquimod to be useful in elderly patients in which biopsy of nodular BCC demonstrates marginal involvement following punch biopsy (Figures 6a and 6b) or shave biopsy (Figures 7a, 7b, 8a, 8b, 9a, 9b, and 9c) or when patients wish to avoid additional surgery.
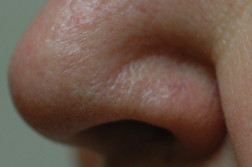 |
| Figure 9c |
|---|
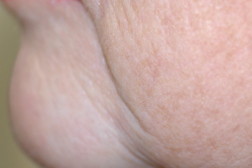
| Figure 9c. Seven years after treatment with imiquimod, the lesion has not reoccurred and has healed with good cosmetic appearance. |
This non-FDA approved indication has not been studied in a large series, but when combined with careful post-operative follow-up may help some patients avoid additional surgery.
Imiquimod is also an excellent modality when sBCC occurs in areas that traditionally heal poorly after surgery, such as the lower leg. Finally, some patients prefer a less-scarring, patient-administered treatment even while accepting that cure rates may be lower. In all of these cases, careful clinical follow-up for signs of recurrent or persistent tumor is recommended for years to identify early signs of recurrence and initiate prompt surgical treatment of these tumors.
Side effects
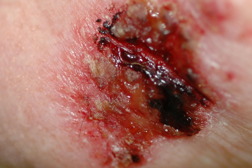
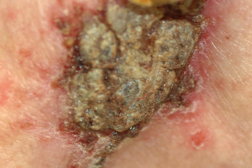
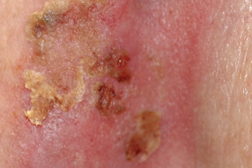
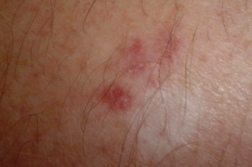
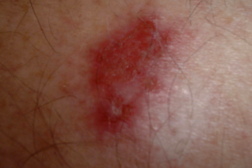
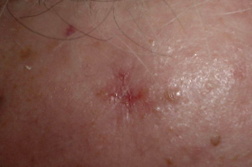
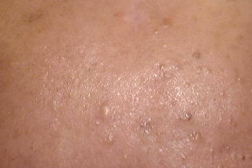
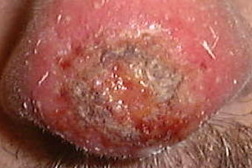
The most common side effects of imiquimod are application site reactions. These include burning, itching, and pain at the target site. Other local skin reactions have been reported including erythema, scabbing, erosions, induration, edema, ulceration, excoriation/flaking, and vesiculation [18, 19, 20]. (Figures 10 and 11) Most patients find these side effects to be tolerable and decreasing the frequency of applications eases side effects when they are intolerable [19, 20]. Interestingly, the potential for clearing of sBCC is associated with increasing severity of erosion, erythema, and scabbing/crusting [19]. In the absence of a visible application site reaction, clearance of sBCC is unlikely. At this point, surgical intervention is recommended.
Summary
Topically applied imiquimod 5 percent cream is an excellent patient-applied medical therapy that is FDA-indicated for sBCC and is especially useful in patients with large or multiple lesions. We have also found it useful in some patients with marginal involvement after excisional surgery of nodular BCC and as a monotherapy for nodular basal cell carcinoma in elderly patients who desire to avoid surgical intervention. In these cases, it is important that patients understand and accept that imiquimod has not been definitively proven to be effective in these clinical settings. Clinical images demonstrate the range of local reactions that occur. Both patients and physicians need to be aware that these exuberant skin reactions are a positive indication of treatment success. This minimizes the potential for premature discontinuation of therapy by unprepared physicians and patients.
References
1. Rubin A, Chen E, Ratner D. Basal Cell Carcinoma. N Engl J Med 2005; 353:2262-9. [PubMed]2. American Cancer Society. Skin Cancer: Basal and Squamous Cell. Available at: http://www.cancer.org/Cancer/SkinCancer-BasalandSquamousCell/DetailedGuide/skin-cancer-basal-and-squamous-cell-key-statistics. Accessed April 15, 2012.
3. Lear J, Tan B, Smith A, et al. Risk factors for basal cell carcinoma in the UK: case-control study in 806 patients. J R Soc Med 1997;90:371-374. [PubMed]
4. Scrivener Y, Grosshans E, Cribier B. Variations of basal cell carcinoma according to gender, age, location and histopathologic subtype. Br J Dermatol. 2002; 147: 41-7. [PubMed]
5. Kuijpers D, Thissen MR, Neumann MHA. Basal Cell Carcinoma: Treatment Options and Prognosis, a Scientific Approach to a Common Malignancy. American Journal of Clinical Dermatology. 2002; 3: 247-259. [PubMed]
6. Drake LA, Ceilley RI, Cornelison RL, et al. Guidelines of care for basal cell carcinoma. J Am Acad Dermatol. 1992: 117-20. [PubMed]
7. Telfer NR, Colver GB, Bowers PW. Guidelines for the management of basal cell carcinoma: British Association of Dermatologists. Br J Dermatol. 1999; 141: 415-23. [PubMed]
8. Preston DS, Stern RS. Nonmelanoma cancers of the skin. N Engl J Med. 1992; 327: 1649-62. [PubMed]
9. Moore, A. Clinical applications for topical 5-fluorouracil in the treatment of dermatological disorders. Journal of Dermatological Treatment. 2009; 20:328-335. [PubMed]
10. Stanley M. Imiquimod and the imidazoquinolones: mechanism of action and therapeutic potential. Clin Exp Dermatol. 2002;27:571-7. [PubMed]
11. Edwards L. Imiquimod in clinical practice. J Am Acad Dermatol. 2000; 43 (suppl): S12-7. [PubMed]
12. Spencer J. Pilot Study of Imiquimod 5% Cream as Adjunctive Therapy to Curettage and Electrodesiccation for Nodular Basal Cell Carcinoma. Dermatol Surg. 2006; 32: 63-69. [PubMed]
13. Hemmi H, Kaisho T, Takeuchi O, et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3:196-200. [PubMed]
14. Ito T, Amakawa R, Kaisho T, et al. Interferon-alpha and interleukin-12 are induced differentially by Toll-like receptor 7 ligands in human blood dendritic cell subsets. J Exp Med. 2002;195:1507-12. [PubMed]
15. Miller RL, Gerster JF, Owens ML, et al. Imiquimod applied topically: a novel immune response modifier and new class of drug. Int J Immunopharmacol. 1999;21:1-14. [PubMed]
16. Suzuki H, Wang B, Shivji GM, et al. Imiquimod, a topical immune response modifier, induces migration of Langerhans cells. J Invest Dermatol. 2000; 114: 135-41. [PubMed]
17. Sterry W, Ruzicka T, Herrera E, Takwale A, Bichel J, Andres K, et al. Imiquimod 5% cream for the treatment of superficial and nodular basal cell carcinoma: randomized studies comparing low frequency dosing with and without occlusion. Br J Dermatol. 2002;147:1227-1236. [PubMed]
18.Geisse J, Rich P, Pandya A, et al. Imiquimod 5% cream for the treatment of superficial basal cell carcinoma: A double blind, randomized, vehicle-controlled study. J Am Acad Dermatol. 2002; 47: 390-398. [PubMed]
19. Geisse J, Caro I, Lindholm J, et al. Imiquimod 5% cream for the treatment of superficial basal cell carcinoma: Results from two phase III, randomized, vehicle-controlled studies. J Am Acad Dermatol. 2004; 50: 722-733. [PubMed]
20. Marks R, Genauer K, Shumack S, et al. Imiquimod 5% cream in the treatment of superficial basal cell carcinoma: Results of a multicenter 6-week dose-response trial. J Am Acad Dermatol. 2001; 44: 807-813. [PubMed]
© 2012 Dermatology Online Journal