Fixed drug eruption caused by the Japanese herbal drug kakkonto
Published Web Location
https://doi.org/10.5070/D36dk9c35mMain Content
Letter: Fixed drug eruption caused by the Japanese herbal drug kakkonto
Megumi Furuichi MD, Hiroshi Hara MD, Yukie Asano MD, Teruhiko Makino MD PhD, Tadamichi Shimizu MD PhD
Dermatology Online Journal 16 (12): 13
Department of DermatologyGraduate School of Medicine and Pharmaceutical Sciences
University of Toyama, Sugitani, Toyama, Japan. shimizut@med.u-toyama.ac.jp
Abstract
Herbal drugs are now widely used throughout the world. The general public tends to believe these agents to be safe because of their natural origin; thus, they are used frequently. However, the side effects of many of these potent chemicals may be significant. Several cases of fixed drug eruption (FDE) caused by herbal drugs have been reported. We herein report a case of FDE caused by kakkonto.
Introduction
Kakkonto is comprised of 7 crude drugs and it is one of the most common Japanese herbal drugs used for colds, headaches, and myalgia. We herein report a case of fixed drug eruption (FDE) caused by kakkonto.
Case report
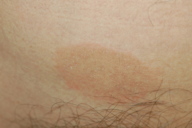
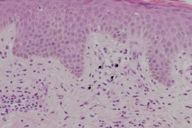
A 36-year-old Japanese male presented with a sudden-onset rash that had appeared on his abdomen 2 days previously. He had noticed pigmentation on the same area several months before the development of the rash. Physical examination revealed a slightly pigmented, red, oval patch that was 3 x 8 cm in diameter on the lower abdomen (Figure 1). Histopathological examination revealed vacuolar alteration of the basal cells, with necrotic keratinocytes and superficial perivascular infiltration of lymphocytes, eosinophils, and melanophages (Figure 2). A detailed history revealed that the patient had a 6-year history of taking 400 mg of an herbal drug, kakkonto, once or twice a month to prevent colds.
We suspected FDE related to kakkonto and performed patch tests after 1 month by using every active ingredient in kakkonto at concentrations of 0.1 and 10 percent in petrolatum: kakkon (Pueraria root), mao (Ephedra leaf), keihi (Cinnamomum bark), shakuyaku (Paeonia root), kanzo (Glycyrrhiza root), taiso (Zizyphus fruit), and shokyo (Zingiber rhizome). The test materials were placed in Haye’s Test Chambers (Haye’s Service B.V., Holland) and applied to the involved abdominal areas. The results were observed at 48 and 72 h according to the recommendations of the International Contact Dermatitis Research Group. Patch tests performed using kakkonto and shakuyaku, present in kakkonto showed positive reactions in the test area. Other active ingredients in kakkonto were negative in the patch test. Based on these clinical and histological findings, in addition to the patch test results, we diagnosed the patient’s eruption as FDE caused by kakkonto. Oral provocation was not carried out with kakkonto. The FDE soon became a pigmented patch and gradually disappeared after the patient stopped taking kakkonto. Since the patient stopped taking the herbal preparation eighteen months ago, he has not had a recurrence.
Discussion
Herbal drugs that are produced by combining multiple crude drugs, are mostly of plant origin but some are of animal or mineral origin. Many preparations contain a variety of active ingredients. Because herbal drugs generally contain many substances, the effect of these drugs is a combination of the various interactions of the constituent substances [1]. The administration of herbal drugs has been reported to be associated with diverse side effects such as interstitial pneumonia [2], renal failure [3], liver toxicity [4], and skin eruption. Fixed drug eruption is a characteristic cutaneous adverse reaction that recurs at fixed sites. Few cases of FDE caused by herbal drugs have been reported [5, 6]. Fujimoto and Tajima reported a case of FDE caused by kakkonto [6]. In their case, the clinical course and the provocation test strongly suggested FDE, whereas the patient showed negative results in the patch tests with kakkonto and the various substances contained in kakkonto [6]. In our case, the patient showed positive reactions in the patch tests with kakkonto and shakuyaku. The rate of positive patch tests on involved areas in FDE has been reported to be as high as 75 to 86 percent and the patch test is a reliable method for confirming the causative agent [6, 7].
In our patient, we showed that shakuyaku, one of the active ingredients in kakkonto, can also cause FDE. Shakuyaku also consists of many herbal drugs and is also a component of kakkonto. Only one case in which FDE was caused by shakuyaku has been specifically reported in the literature [8]. Tokishakuyakusan has been reported to cause an ashy dermatosis-like skin eruption [8]. A patient had a positive skin test for shakuyaku, one of the active ingredients of tokishakuyakusan. The clinical presentation of ashy dermatosis sometimes cannot be distinguished from that of multiple FDE [9].
Herbal drugs are now widely used throughout the world and they are generally believed to be safe agents because of their natural origin. Fixed drug eruptions usually comprise single or multiple patches or plaques that may be asymptomatic or have mild associated symptoms. In addition to supplements, herbal foot baths, herbal pillows, herbal lotions, and herbal shampoos are common, everyday items, which may be overlooked as non-orally administered herbal products that may cause FDE in some patients [10]. Therefore, dermatologists should be aware of the fact that both oral and non-oral herbal drugs may cause FDE.
References
1. Ikegami F, Fujii Y, Satoh T. Toxicological considerations of Kampo medicines in clinical use. Toxicology 2004; 198: 221-8. [PubMed]2. Kawasaki A, Mizushima Y, Kunitani H, Kitagawa M, Kobayashi M. A useful diagnostic method for drug-induced pneumonitis: a case report. Am J Chin Med 1994; 22: 329-36. [PubMed]
3. Vanherweghem JL, Depierreux M, Tielemans C, et al. Rapidly progressive interstitial renal fibrosis in young women: association with slimming regimen including Chinese herbs. Lancet 1993; 341: 387-91. [PubMed]
4. Kane JA, Kane SP, Jain S. Hepatitis induced by traditional Chinese herbs; possible toxic components. Gut 1995; 36: 146-7. [PubMed]
5. Matsumoto K, Mikoshiba H, Saida T. Nonpigmenting solitary fixed drug eruption caused by a Chinese traditional herbal medicinema huang (Ephedra Herba), mainly containing pseudoephedrine and ephedrine. J Am Acad Dermatol 2003; 48: 628-30. [PubMed]
6. Fujimoto N, Tajima S. Extensive fixed drug eruption due to the Japanese herbal drug kakkon-to. Br J Dermatol 2003; 149: 1303-4. [PubMed]
7. Alanko K, Stubb S, Reitamo S. Topical provocation of fixed drug eruption. Br J Dermatol. 1987; 116: 561-7. [PubMed]
8. Hanai H, Suzuki H. Ashy dermatosis caused by a traditional Chinese medicine extract (Toki-shakuyaku-san). Environ Dermatol 2000; 7: 169-72.
9. Hayashi H, Shimizu T, Shimizu H. Erythema dyschromicum perstance caused by acetaminophen. Clin Exp Dermatol 2003; 28: 455-6. [PubMed]
10. Tan C, Zhu WY. Chinese herbal medicine: a neglected offender for fixed drug eruptions. Eur J Dermatol 2010; 20: 1-2. [PubMed]
© 2010 Dermatology Online Journal