Generalized essential telangiectasia
Published Web Location
https://doi.org/10.5070/D36dd8m6rkMain Content
Generalized essential telangiectasia
Julie K Karen MD, Stephanie J Mengden MD, Hideko Kamino MD, Jerome L Shupack MD
Dermatology Online Journal 14 (5): 9
Department of Dermatology, New York UniversityAbstract
A 56-year-old woman presented with multiple, erythematous cutaneous vessels that blanched with diascopy and were symmetrically distributed over both lower and upper extremities. The lesions had been present for approximately 6 years and were asymptomatic. Generalized essential telangiectasia (GET) is an idiopathic syndrome of widespread, asymptomatic telangiectases of unknown cause. In our patient, an extensive laboratory evaluation showed low levels of circulating vitamin C as well as the presence of urinary matrix metalloproteinases. The relevance of these abnormal findings is unclear. No uniformly effective treatment exists for GET. Our patient was started on oral doxycycline and was referred for laser therapy.
Clinical synopsis
A 56-year-old woman presented with a 6-year history of asymptomatic, progressive reddening of her extremities. The eruption began with discrete, symmetrically-distributed telangiectases that involved the dorsal aspects of the feet and ankles. The lesions ascended to involve her thighs, and, more recently, both upper extremities. She reported no associated pruritus or pain. There was no history of epistaxis or other hemorrhage. Medical history included a meningioma, ulcerative colitis, and childhood allergic rhinitis. Because of her colitis, she restricted her diet and consumed no citrus fruits. Surgical history included resection of the meningioma in 2005 and a bilateral oophorectomy in 2006. Medications included mesalamine, spironolactone, docusate sodium, vitamin D, calcium, and biotin. No family members had similar lesions. Treatment with oral doxycycline was initiated. The patient was referred for laser treatment.
Physical examination
Innumerable, fine, erythematous telangiectases that blanched with diascopy were distributed symmetrically over the dorsal aspects of the feet, toes, lower legs, thighs, and upper extremities. There was no evidence of purpura, pigmentary alteration, or atrophy. Mucosal lesions were absent.
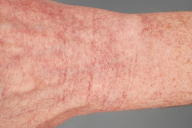 | 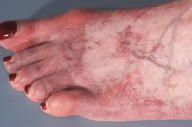 |
| Figure 1 | Figure 2 |
|---|
Laboratory data
Complete blood count, comprehensive metabolic panel, thyroid function tests, and immunoglobulin electrophoresis were normal. A thrombophilia panel, which included PT, INR, PTT, thrombin time, fibrinogen, antithrombin III, protein S, factor VIII, activated protein C, functional protein C, and DRVVT, was normal except for an elevated fibrinogen of 456 mg/dl (reference internal:150-350). Antinuclear, anti-Ro, anti-La, aniti-cardiolipin, anti-thyroid peroxidase, and anti-thyroglobulin antibodies were negative. Serum vitamin C was low at 0.3 mg/dL (reference interval: 0.4 to 2.0). The patient's urine was analyzed by Dr. Judah Folkman's Vascular Biology Laboratory at Boston Children's Hospital for the presence of various angiogenic factors. The following intact matrix metalloproteinases (MMPs) were detected: <150kDa (MMP dimer), 140 kDa (MMP-9/NGAL complex), 92 kDa (MMP-9), and 65 kDa (MMP-2). Tests for basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VEGF) were negative.
Histopathology
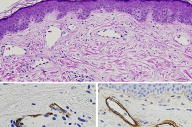 |
| Figure 3 |
|---|
There are telangiectases in the papillary dermis and upper reticular dermis and a sparse, perivascular infiltrate of lymphocytes. Immunostains for CD31, CD34, and Ulex demonstrate a continuous layer of positive endothelial cells in the pre-existing vessels and in the telangiectatic vessels. Muscle specific actin (A) and smooth-muscle actin show partial-to-complete loss of pericytes. Collagen IV (B) shows some telangiectases with a multilayered basement membrane. In the reticular dermis, there are thick and thinner collagen bundles in a haphazard array.
Comment
Generalized essential telangiectasia (GET) refers to an uncommon, acquired syndrome that is characterized by widespread, progressive telangiectases of unknown cause [1]. As the name implies, telangiectases develop without preceding or coexisting cutaneous or systemic disease. Lesions of GET are symmetrically distributed and classically involve the lower extremities first, with subsequent spread to the upper extremities and trunk. The dilated blood vessels represent capillary telangiectases. Lesions are pink-to-erythematous in color and are macular. Externally applied pressure results in blanching. Lesions can be punctate, linear, or reticulate and, when numerous, may confer a speckled appearance to involved skin. The clinical appearance of the telangiectases is worsened by dependent positions. Although uncommon, involvement of the mucosa and conjunctive has been reported in some cases of GET [2, 3]. Lesions are generally asymptomatic although patients sometimes report tingling, burning, or numbness [1]. The incidence and prevalence of GET are not known. The majority of cases of GET are sporadic although there are rare reports of familial occurrence [4]. Adult women are most commonly affected, with a typical age of onset during the late fourth or fifth decade. Generalized essential telangiectasia appears to be more common in Caucasian individuals [1]. The pathogenesis of GET remains elusive. It is possible that low vitamin C, the consequence of a diet devoid of citrus fruits, contributed to our patient's phenotype. Vitamin C functions as an essential co-factor for various enzymes that are involved in collagen biosynthesis. In guinea pigs, vitamin C deficiency has been shown to lead to down-regulation of type IV collagen gene expression [5]. Endothelial-cell production of type IV collagen is critical to the maintenance of vascular integrity. One of the most characteristic features of vitamin C deficiency (or scurvy) is cutaneous hemorrhage. Surprisingly, purpura was not a feature of our patient's clinical or microscopic examination. The majority of cases of GET are isolated and benign findings; however, gastrointestinal tract bleeding has been reported [6]. Additionally, there are reports of GET occurring in association with underlying autoimmune thyroiditis [7, 8]. Routine laboratory work is consistently normal. Clinically, GET must be distinguished from hereditary hemorrhagic telangiectasia, which shares in common with GET the finding of widespread telangiectases. Adult onset, lack of an inheritance pattern, predilection for the lower extremities, and lack of associated hemorrhage are features that help to distinguish GET from this entity. In the majority of cases, telangiectases persist indefinitely without a tendency for spontaneous regression. Although generally asymptomatic, lesions of GET are usually widespread and can be cosmetically disfiguring. Uniformly effective therapy does not exist. Capillary telangiectases, unlike their venous counterparts, are typically refractory to sclerotherapy. Destruction with the flashlamp-pumped pulsed dye laser [9] and long-pulse, frequency-doubled Nd:YAG laser [10] have been reported to be successful in certain cases. Clearance of telangiectases has been achieved with acyclovir [8] and ketoconazole [11]; however, the vast majority of patients with GET do not improve with these medications. The literature also contains reports of successful treatment with oral tetracyclines [12]. The precise mechanism by which the tetracyclines effect resolution remains obscure. Tetracyclines are known to inhibit matrix metalloproteinases (MMPs) [13], which are a family of zinc-dependent endopeptidases that collectively degrade various molecular components of the basement membrane. Enzymatic degradation of the basement membrane by MMPs facilitates angiogenesis and represents an important hallmark of cancer progression. The detection of intact MMPs in the urine of cancer patients has been reported to be an independent predictor of disease status [14, 15]. One might posit an etiopathogenic role for pro-angiogenic MMPs in the development of telangiectases in patients with GET. The importance of the detection of MMPs in our patient's urine is not known. It is unclear whether she improved during 7 weeks of therapy with doxycycline, as might have been expected. She experienced dyspepsia requiring her to discontinue the medication.
References
1. McGrae JD Jr, Winkelmann RK. Generalized essential telangiectasia: report of a clinical and histochemical study of 13 patients with acquired cutaneous lesions. JAMA 1963; 185:9092. Ali MM, et al. Generalized essential telangiectasia with conjunctival involvement. Clin Exp Dermatol 2006; 31:781
3. Mannis MJ, et al. Ocular manifestations of generalized essential telangiectasia. Cornea 1999; 18:731
4. Bourlond A, et al. Micronodular generalized familial angiomatosis (generalized essential telangiectasia): clinical cases; ultrastructural study [Abstract]. Ann Dermatol Venereol 1988; 115:909
5. Moahmoodian F, Peterkofsky B. Vitamin C deficiency in guinea pigs differentially affects the expression of type IV collagen, laminin, and elastin in blood vessels. J Nutr 1999; 129:83
6. Checketts SR, et al. Generalized essential telangiectasia in the presence of gastrointestinal bleeding. J Am Acad Dermatol 1997; 37:321
7. Buckley R, et al. Generalized essential telangiectasia in a patient with Graves' disease: should the spectrum of autoimmune diseases associated with generalized telangiectasia be expanded? Cutis 2000; 65:175
8. Shelley WB, Shelley ED. Essential progressive telangiectasia in an autoimmune setting: successful treatment with acyclovir. J Am Acad Dermatol 1989; 21:1094
9. Buscaglia DA, Conte ET. Successful treatment of generalized essential telangiectasia with the 585-nm flashlamp-pumped pulsed dye laser. Cutis 2001; 67:107
10. Gambichler T, et al. Generalized essential telangiectasia successfully treated with high-energy, long-pulse, frequency-doubled Nd:YAG laser. Derm Surg 2001; 27:355
11. Shelley WB, Fierer JA. Focal intravascular coagulation in progressive ascending telangiectasia: ultrastructural studies of ketoconazole-induced involution of vessels. J Am Acad Dermatol 1984; 10:876
12. Shelley WB. Essential progressive telangiectasia: successful treatment with tetracycline. JAMA 1971; 216:1343
13. Schneider BS, et al. Tetracyclines inhibit intracellular muscle proteolysis in vitro. Biochem Biophys Res Commun 1992; 188:767
14. Moses MA, et al. Increased incidence of matrix metalloproteinases in urine of cancer patients. Cancer Res 1998; 58:1395
15. Fernandez CA. The matrix metalloproteinase-9/neutrophil gelatinase-associated lipocalin complex plays a role in breast tumor growth and is present in the urine of breast cancer patients. Clin Cancer Res 2005; 11:5390
© 2008 Dermatology Online Journal

