Severe atopic dermatitis and leflunomide: first clinical experience and highlights of pertinent experimental data
Published Web Location
https://doi.org/10.5070/D36cg6s9t4Main Content
Severe atopic dermatitis and leflunomide: First clinical experience and highlights of pertinent experimental data
Gottfried Wozel, Lilla Vitéz, Christiane Pfeiffer
Dermatology Online Journal 12 (2): 6
Department of Dermatology, Technical University Dresden, Germany. Christiane.Pfeiffer@mailbox.tu-dresden.deAbstract
Atopic dermatitis is a common chronically relapsing disease affecting about 10 percent of children and 3 percent of adults. Currently, no standard management exists for long-term treatment. Topical corticosteroids and recently calcineurin-inhibitors are effective in most patients. In severe cases, however, systemic agents have to be employed. Their use may be limited by unwanted effects or insufficient long-term efficiency. Leflunomide is an immunomodulating and disease-modifying antirheumatic drug with anti-inflammatory and immunosuppressive activity, exhibiting an extremely long in vivo half life. Because T cells and eosinophils play an important role in the pathophysiology of atopic dermatitis, and long-term treatment is often required, leflunomide seems to be ideally suited for treatment of severe atopic eczema. We present a case highlighting the application regimen of leflunomide, and discuss the pathophysiological mechanism of action in atopic dermatitis. Because treatment benefit differs between patients, we propose the design of a proof-of-principle study for leflunomide in atopic dermatitis encompassing an evaluation of predictive markers for successful application.
Introduction
Atopic dermatitis is a chronic inflammatory skin disease that may significantly impact patients' quality of life because of its severe pruritus and often unpredictable remitting-relapsing course [1]. The management of atopic dermatitis includes treatment with anti-inflammatory drugs, avoidance of trigger factors, and psychological interventions [2, 3, 4]. Topical and systemic treatment modalities, including corticosteroids and calcineurin inhibitors, induce remission in most patients. Numerous randomized clinical trials have served to optimize dosing and treatment schedules, but they minimize the historically high rebound rate after treatment discontinuation. Clinical trials underline that the intermittent use of topical corticosteroids twice weekly in conjunction with the use of emollients reduces the risk of severe flares [5, 6]. Similar observations have been made using topical calcineurin-inhibitors [7, 8]. In a recently published retrospective multicenter study, patients of different age groups were evaluated in terms of the steroid dosage used as well as the frequency and severity of adverse events. Most patients showed an improved or steady-state skin condition after 6 months of conventional topical corticosteroid therapy, however 19 percent of the patients did not respond despite a higher corticosteroid use [9]. Severely affected patients who do not respond to topical treatment are offered a range of systemic treatment options including phototherapy, systemic immunosuppressive agents, and immune response modifiers [2, 3]. Several controlled clinical studies demonstrate cyclosporine to be highly efficacious in the treatment of acute phases of eczema [10]. Unfortunately, a relapse within a few weeks of discontinuation of therapy is common. Unwanted effects include, for example, hypertension and renal toxicity. Azathioprine has been also studied extensively in treatment of atopic eczema [11]. Long-term administration in young patients is usually avoided because of potential side effects such as hepatitis and tumor induction. Methotrexate and mycophenolate mofetil are effective for some patients, however controlled studies are lacking. Thus, a need for additional systemic drugs exists.
Our group was able recently to demonstrate the benefit of leflunomide in two patients suffering from severe atopic dermatitis [13]. Leflunomide is an immunomodulating compound with immunosuppressive properties, targeting primarily the T-cell by inhibiting pyrimidine de novo synthesis [13]. Because abnormal T-cell activation and Th1/Th2 imbalance occur in atopic dermatitis, leflunomide could be useful to correct T-cell pathology.
Experimental evidence suggests that leflunomide can target eosinophils via a direct action on eotaxin, independent of leflunomide's antimetabolite activity [14, 15]. Clinical severity scores in atopic dermatitis have been observed to correlate with levels of eosinophil granule proteins (such as eosinophil-derived neurotoxin or EDN) and with eosinophil infiltration into lesional skin [16, 17]. Here, we will present an additional case, highlighting that not all patients with atopic dermatitis respond equally to leflunomide therapy. We also discuss factors that may help define the subgroup benefiting from leflunomide treatment based upon our in vitro data.
Clinical synopsis
A 33-year-old man presented with severe atopic dermatitis and allergic rhino-conjunctivitis since early childhood. Sensitizations to house dust mite and grass pollen were clinically relevant. Several topical and systemic therapies including antihistamines, phototherapy, and corticosteroids were tried, all with poor efficacy. Treatment with cyclosporine (4.5 mg/kg body weight) for 1 year was well tolerated, but not effective. An autoaggressive personality disorder with excessive scratching behavior was diagnosed by a psychotherapist. Stress attributed to unemployment and social isolation triggered severe exacerbations requiring multiple hospitalizations. Hospital treatment induced short-term remissions.
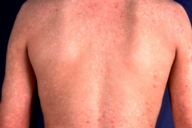 |
| Figure 1A |
|---|
| Clinical skin features of the patient before treatment with leflunomide |
In January 2002 the patient presented with nearly erythrodermic oozing erythema complicated by multiple excoriations, severe pruritus, and sleeplessness. The eczema and severity index (EASI) was 24.3 and the severity scoring of atopic dermatitis (SCORAD) was 57.9. Laboratory values were normal except for the level of total IgE (≥ 5000 kU/l). Treatment with leflunomide was initiated at a dosing schedule established in rheumatoid arthritis, starting with a loading dose of 100 mg daily for 3 days followed by a maintenance dose of 20 mg daily. For the treatment of pruritus, levocetirizine and hydroxyzine were prescribed. Topical therapy with prednicarbate for lesions on the body and fluocortine for the facial skin in addition to emollients was continued.
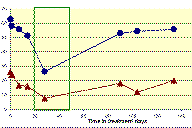 |
| Figure 2 |
|---|
EASI and SCORAD assessment before and during monotherapy with leflunomide and in combination with systemic prednisolone (green marked area). (blue dots: SCORAD, red triangles: EASI)(click for full-size version)
At the subsequent visits, physical examination, EASI, SCORAD and laboratory findings were assessed. Despite of the initiation of leflunomide therapy, eczema, pruritus, and sleeplessness became worse after 3 weeks; the EASI at this time being 16 and the SCORAD was 51.3. Additional systemic therapy with corticosteroids was started (prednisolone initial dose was 60 mg daily). On 20 mg prednisolone for 2 weeks, with ongoing alternating doses of 10 and 20 mg leflunomide daily, resumption of disease activity was noted (EASI 7.8, SCORAD 26.3). After reduction and finally cessation of prednisolone therapy, symptoms slowly worsened over the next 3 months. Therefore, the daily dose of leflunomide was increased to 20 mg. Staphylococcus aureus was cultured from skin. Flucloxacillin was administered in conjunction with topical treatment (prednicarbate and emollients) and within 10 days the eczema, pruritus and insomnia improved. The EASI dropped from initial 34.3 to 11.9. On long-term follow-up, however, the patient was not content with the skin improvement obtained on monotherapy with leflunomide. Thus, the drug was stopped after 32 weeks of treatment. EASI increased to 20 and the SCORAD to 55.7.
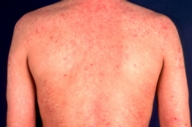 | 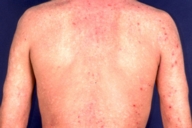 |
| Figure 1B | Figure 1C |
|---|---|
| Figure 1B. During combination treatment phase of leflunomide and systemic prednisolone Figure 1C. At the end of leflunomide treatment | |
The patient started psychotherapy with a special focus on interrupting itch-scratch cycles. His professional situation improved. He continued intensive topical therapy with steroids and emollients. During the 6 months following cessation of leflunomide therapy, his disease stabilized for the first time in more than five years of treatment.
Discussion
Leflunomide's mode of action
Leflunomide is characterized by its immunosuppressive, antiproliferative, anti-inflammatory, and antirheumatic properties [18]. In animal models leflunomide suppresses the rejection of transplants [13]. The compound is considered a prodrug since it is rapidly metabolized following oral administration to its active metabolite (A 771726). The metabolite reversibly inhibits the enzyme dihydroorotate dehydrogenase blocking pyrimidine de novo synthesis and thus interfering with T-cell proliferation and production of autoantibodies [19, 20]. Additionally, leflunomide exhibits other anti-inflammatory effects such as inhibition of COX-2 or histamine release in basophils [13, 21]. Based on its immunomodulating properties the drug is approved [22] for the management of rheumatoid arthritis [23], psoriatic arthritis [24, 25], and it is being evaluated for use in organ transplant recipients to prevent rejection reaction [13]. There is also evidence that leflunomide is active in the treatment of autoimmune bullous diseases [26], SAPHO syndrome [27], and contact dermatitis (in experimental models) [28].
Leflunomide may interact with eosinophil influx in atopic dermatitis
Eosinophils play an important role in skin infiltrates in atopic eczema. Recent reports highlight the relationship between eotaxin-3 levels and severity of disease [29]. We were able to demonstrate that the active metabolite of leflunomide inhibits eotaxin release by IL-4- and TNFα-stimulated fibroblasts [14]. More importantly, this inhibition is also observed on mRNA-expression level and is not reversed by uridine addition, therefore suggesting a mechanism not mediated by inhibition of pyrimidine de novo synthesis [15]. Manna et al. show that leflunomide can also inhibit TNFα action by preventing IκB-degradation and thereby preventing release of NFκB [30]. IL-4 and TNFα also allow endothelial cells to express VCAM-1, a homing molecule that favors homing of eosinophils, as these in contrast to neutrophils express VCAM-1's counterreceptor VLA-4 [31]. VLA-4 expression on eosinophils is upregulated upon eotaxin-exposure [32]. After transmigration, eosinophils follow gradients of chemotactic factors, with eotaxin's chemotactic strength surpassing other chemokine receptor 3 (CCR3) binding chemokines such as RANTES and MCP-4 up to a hundredfold [16]. A subset of activated T lymphocytes also express VLA-4. Th2 lymphocytes, known to be important in the early stages of atopic eczema lesions, also carry the CCR3 receptor for eotaxin [33]. Leflunomide's blocking of eotaxin expression may thus be an explanation for its observed activity in patients with an erythrodermic pattern.
Leflunomide may be employed in severe atopic dermatitis
We recently published two cases of severe, recalcitrant atopic dermatitis in whom leflunomide treatment induced a long-lasting remission [12]. In contrast to the case reported in the present paper, in those patients leflunomide monotherapy induced long-term remission. In the current patient, however, an acceptable skin response could be obtained only by a combination of leflunomide with medium doses of prednisolone. Because leflunomide has a very long terminal elimination half-life of 1-2 years, induction of remission may outlast the actual duration of therapy by many weeks [34]. The remission in the present case, which occurred concomitantly with the addition of psychotherapy, even if obtained shortly after the cessation of leflunomide, may still have started while on significant serum levels of active leflunomide metabolite.
In all our patients treated with leflunomide the drug was well tolerated; we observed no adverse events. In contrast, 15 percent of rheumatoid arthritis patients receiving leflunomide in clinical trials discontinued the drug because of adverse effects [13]. Moreover, leflunomide's active metabolite demonstrates antiviral activity using CMV-infected human fibroblasts and endothelial cells. Although the clinical importance of this observation is unclear, it could be useful for treatment of AD, which is often complicated by herpes infections [35].
Markers that predict successful action in subsets of patients with severe atopic dermatitis would be useful
Our data indicate that the next step should be the initiation of a proof-of-principle study including a larger number of atopic eczema patients in order to confirm our clinical experience. Patients suffering from atopic eczema present a wide spectrum of disease including intrinsic and extrinsic forms, a plethora of trigger factors with variable importance in the individual patient, and a multitude of clinically different patterns [4]. The response of these patients to different treatment modalities mirrors this wide spectrum. Thus, it would be useful to reveal predictive markers for treatment success by studying larger patient populations. To assess the importance of involvement of eosinophils in an individual patient, serum eosinophil cationic protein and urine eosinophil-derived neurotoxin may be feasible surrogate markers [34]. To determine the importance of T cells, soluble IL2-receptor can be measured. Determination of total IgE allows an indirect estimation of Th2 activity, because total IgE correlates with IL-4 levels. Finally, serum protein electrophoresis is a useful tool for estimation of acute phase proteins reflecting IL1- and IL6-release, i.e., monocyte lineage participation. Our in vitro data suggest that leflunomide activity is directed predominantly on eosinophil homing and activation, indicating that surrogate markers for eosinophil involvement in atopic eczema should be studied in leflunomide's clinical application.
In conclusion we have provided evidence that leflunomide exerts measurable antiinflammatory effects in patients with severe atopic dermatitis.
Acknowledgment. We thank Ms. Katharina Blümlein for her expert technical assistance.
References
1. Williams HC, Epidemiology of atopic dermatitis, Clin Exp Dermatol. 2000; 25:522-529. PubMed2. Sidbury R, Hanifin JM, Systemic therapy of atopic dermatitis, Clinical Exp Dermatol. 2000; 25:559-566. PubMed
3. Thestrup-Pedersen K, Treatment principles of atopic dermatitis, J Eur Acad Dermatol Venereol. 2002;16-1-9. PubMed
4. Leung DYM, Bieber T, Atopic dermatitis, Lancet. 2003; 361:151-160. PubMed
5. Hanifin J, Gupta AK, Rajagopalan R, Intermittent dosing of fluticasone propionate cream for reducing the risk of relapse in atopic dermatitis patients, Br J Dermatol. 2002; 147:528-537. PubMed
6. Berth-Jones J, Damstra RJ, Golsch S et al., Twice weekly fluticasone propionate added to emollient maintenance treatment to reduce risk of relapse in atopic dermatitis: randomised, double blind, parallel group study, BMJ. 2003; 326:1367. PubMed
7. Meurer M, Fölster-Holst R, Wozel G et al., Pimecrolimus cream in the long-term management of atopic dermatitis in adults: a six-month-study, Dermatology. 2002; 205:271-277. PubMed
8. Thaci D, Long term management of childhood atopic dermatitis with calcineurin inhibitors, Hautarzt. 2003; 54:418-423. PubMed
9. Furue M, Terao H, Rikihisa W et al., Clinical dose and adverse effect of topical steroids in daily management of atopic dermatitis, Br J Dermatol. 2003; 148:128-133. PubMed
10. Harper JI, Ahmed I, Barclay G et al., Cyclosporin for severe childhood atopic dermatitis: short course versus continuous therapy, Br J Dermatol. 2000; 142: 52-8. PubMed
11. Berth-Jones J, Takwale A, Tan E et al., Azathioprine in severe adult atopic dermatitis: a double-blind, placebo-controlled, crossover trial, Br J Dermatol. 2002; 147: 324-30. PubMed
12. Schmitt J, Wozel G, Pfeiffer C, Leflunomide as a novel treatment option in severe atopic dermatitis, Br J Dermatol. 2004; 150:1182-1185. PubMed
13. Leflunomide. In: AHFS Drug Information 2003. American Society of Health-System Pharmacists®, Bethesda 2003; 3639-3644
14. Kehrer T, Blümlein K, Wozel G, Eotaxin release is suppressed by the metabolite A 77 1726 of the novel immunomodulating agent leflunomide, Eur J Allergy Clin Immunol. 2001; 56 (S68):144.
15. Pfeiffer C, Kehrer T, Wozel G, Suppression of eotaxin expression and release by leflunomide's active metabolite A77 1726 is not reversed by uridine, XXIII EAACI congress, 2004; 12-16.06. Amsterdam
16. Amerio P, Frezzolini A, Feliciani C et al., Eotaxins and CCR3 receptor in inflammatory and allergic skin diseases: therapeutical implications, Current drug Targets-Inflammation and Allergy. 2003; 2:81-94. PubMed
17. Breuer K, Kapp A, Werfel T, Urine eosinophil protein X (EPX) is an in vitro parameter of inflammation in atopic dermatitis of the adult age, Allergy. 2001; 56: 780-784. PubMed
18. Breedveld FC, Dayer JM, Leflunomide: mode of action in the treatment of rheumatoid arthritis, Ann Rheum Dis. 2000; 59:841-849. PubMed
19. Rückemann K, Fairbanks L, Carrey EA et al., Leflunomide inhibits pyrimidine de novo synthesis in mitogen-stimulated lymphocytes from healthy humans, J Biol Chem. 1998; 273:21682-21691. PubMed
20. Siemasko KF, Chong AS, Williams JW et al., Regulation of B- cell function by the immunosuppressive agent leflunomide, Transplantation. 1996; 61:635-42. PubMed
21. Hamilton LC, Vojnovic I, Warner TD, A771726, the active metabolite of leflunomide, directly inhibits the activity of cyclo-oxygenase-2 in vitro and in vivo in a substrate-specific manner, Br J Pharmacol. 1999; 127:1589-1596. PubMed
22. Kaltwasser JP, Behrens F, Leflunomide :long-term clinical experience and new uses, Expert Opin Pharmacother. 2005; 6: 787-801. PubMed
23. Fox RI, Mechanism of action of leflunomide in rheumatoid arthritis, J Rheumatol. 1998; 25:S(53):20-26. PubMed
24. Reich K, Hummel KM, Beckmann I et al., Treatment of severe psoriasis and psoriatic arthritis with leflunomide, Br J Dermatol. 2002; 146: 335-6. PubMed
25. Schmitt J, Wozel G, Psoriasisarthritis, J Deutsch Dermatol Ges. 2004; 2:763-6. PubMed
26. Nousari HC, Anhalt GJ, Bullous pemphigoid treated with leflunomide, Arch Dermatol. 2000; 136:1204-1205. PubMed
27. Scarpato S, Tirri E, Successful treatment of SAPHO syndrome with leflunomide, Clin Exp Rheumatol .2005; 23:731. PubMed
28. Feng H, Li XY, Zheng JR, Gao JW, Xu LF, Tang MY, Inhibition of nuclear Factor-kB signaling pathway by leflunomide or triptolide also inhibits the anthralin- induced inflammatory response but does not affect keratinocyte growth inhibition, Biol Pharm Bull. 2005; 28(9):1597-1602. PubMed
29. Kagami S, Kakinuma T, Saeki H et al., Significant elevation of serum levels of eotaxin-3/CCL26, but not of eotaxin-2/CCL24, in patients with atopic dermatitis: serum eotaxin-3/CCL26 levels reflect the disease activity of atopic dermatitis, Clin Exp Immunol. 2003; 134:309-313. PubMed
30. Manna SK, Mukhopadhyay A, Aggarwal BB, Leflunomide inhibits TNF-induced cellular responses: effects on NFκB, activator protein-1, c-Jun N-terminal protein kinase, and apoptosis, J Immunol. 2000; 165:5962-5969. PubMed
31. Sriramarao P, DiScipio RG, Cobb RR et al., VCAM-1 is more effective than MAdCAM-1 in supporting eosinophil rolling under conditions of shear flow, Blood. 2000; 95(2):592-601. PubMed
32. Hohki G, Terada N, Hamano N et al., The effects of eotaxin on the surface adhesion molecules of endothelial cells and on eosinophil adhesion to microvascular endothelial cells, Biochemical and Biophysical Research Communications. 1997; 241:136-141. PubMed
33. Sallusto F, Mackay CR, Lanzavecchia A, Selective expression of the eotaxin receptor CCR3 by human T helper 2 cells, Science. 1997; 277: 2005-2007. PubMed
34. Wozel G, Pfeiffer C, Leflunomid - ein neues Medikament zur pharmakologischen Immunmodulation, Hautarzt. 2002; 53:309-315. PubMed
35. Wollenberg A, Zoch C, Wetzel S et al., Predisposing factors and clinical features of eczema herpeticum: a retrospective analyzing 100 cases, J Am Acad Dermatol. 2003; Aug; 49 (2):198-205. PubMed
© 2006 Dermatology Online Journal

