Primary cutaneous plasmacytoma occurring after pacemaker implantation and recurring in scar tissue
Main Content
Primary cutaneous plasmacytoma occurring after pacemaker implantation and recurring in scar tissue
Anthony Li2 MBBS, Alexander Chen1 MBBS, Mark Gallagher2 MD, Pawel Kaczmarek1 MBBS, Brendan Tinwell2 MBBCh, James Sneddon1,2 MD, Sandeep Cliff1 MBBS
Dermatology Online Journal 19 (2): 3
1. East Surrey Hospital2. St. George’s Hospital
London, Great Britain
Abstract
Primary cutaneous plasmacytomas are rare and little is known about their treatment and progression. We describe for the first time the predilection of primary cutaneous plasmacytoma to occur in a scar or sites of trauma. We report an 89-year-old man who presented with a slowly expanding asymptomatic mass over his pacemaker implantation, 1 year after insertion. Further investigation ruled out multiple myeloma and histology confirmed it to be a cutaneous plasmacytoma. This was treated successfully by local radiotherapy after extraction of the pacemaker and implantation of a new pacemaker on the opposite side. The patient subsequently developed an additional cutaneous plasmacytoma over the new pacemaker site, followed by the development of progressive multiple myeloma. Primary cutaneous plasmacytoma can show predilection for sites of trauma or surgery. Surgical excision of the plasmocytoma and local radiotherapy may be a reasonable strategy in the first initially after pacemaker removal.
Introduction
Extramedullary plasmacytomas by definition occur outside the bone marrow and are part of a group of plasma cell dyscrasias that result from the proliferation of a single clone of plasma cells. Solitary (primary) cutaneous plasmacytomas occur in the absence of multiple myeloma and are rare; there are only a few case reports in the literature. Diagnosis requires biopsy proven clonal plasma cell proliferation. In addition, there should be less than 10% of clonal plasma cells in the bone marrow and absence of organ or tissue impairment related to the disorder [1].
Report
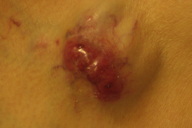 |
| Figure 1 |
|---|
| Figure 1. Cutaneous plasmacytoma overlying left pacemaker implant |
An 89-year-old man was admitted from cardiology clinic 1 year after implantation of a dual chamber pacemaker in a standard subcutaneous pocket in the left infraclavicular location. He had a past history of hypertension, polymyalgia rheumatic, and mild asthma. Over the previous 6 months he had noted a slowly growing lump overlying the pacemaker. Clinical examination showed a 5 cm x 6 cm firm, non-pulsatile, non-tender erythematous nodule. The overlying skin temperature was normal (Figure 1) and the patient was asymptomatic. Skin cultures were negative.
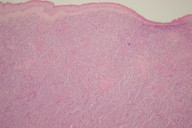
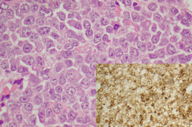
On exploration of the lesion, the center was found to be friable. Gentle blunt dissection resulted in the creation of a cavity extending down to the sleeve of the pacing leads. The tumor was debrided as fully as possible and samples of the tissue were sent for histology and for culture. Culture was negative but histological examination revealed soft tissue infiltration by sheets of atypical plasmacytoid cells displaying CD79a, CD138 (weak), CD45 and MUM-1 (Weak) (Figure 2). Blood results demonstrated a total protein of 74 g/L with an albumin of 36 g/L. Other test included a urea of 15.6 mmol/L, creatinine of 169 umol/L and normal adjusted calcium. He had a hemoglobin of 9.9 g/dl (normocytic), normal platelet count, and normal white cell count. Serum electrophoresis revealed a mid gamma globulin band with a paraprotein of 12.3 g/L confirmed on immunofixation to be IgG lambda. IgA and IgM levels were normal. Urine electrophoresis showed no Bence-Jones protein. Bone marrow biopsy showed normal hemopoiesis. CT of the neck/thorax/abdomen and full skeletal survey were unremarkable. A solitary plasmacytoma was diagnosed.
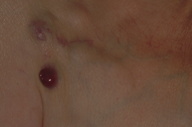 |
| Figure 3 |
|---|
| Figure 3. View of cutaneous plasmacytoma overlying the new right-sided pacemaker implant |
Before beginning radiotherapy, the pacemaker was removed and both leads were extracted. In a separate procedure, a new pacing system was implanted on the right side. The patient then underwent local radiotherapy with 20 Gy in 5 fractions resulting in complete remission. Within 2 months, 3 identical lesions developed over the site of the newly implanted pacemaker (Figure 3). The patient declined further local radiotherapy and further surgical revision of the pacing system, so systemic therapy with high dose dexamethasone was given. There was a poor response; 4 further nodules developed on the patient’s back over the following month. Progression to multiple myeloma occurred after 2 months and was treated with combination dexamethasone and thalidomide. Despite treatment, he died of progressive disease.
Discussion
To our knowledge, there have been only 2 previous case reports of plasmacytoma involving a pacemaker. The first involved a 48-year-old man who developed a secondary plasmacytoma related to myeloma 16 months after a pacemaker generator substitution; the second was in a 74-year-old female who developed a plasmacytoma overlying a pacemaker implantated 2 years after chemotherapy for myeloma. Both patients were treated unsuccessfully by local Cobalt therapy and chemotherapy, respectively. In both cases generator explant eventually proved necessary, but both patients subsequently died of rapidly progressive disease [2, 3].
The link between pacemaker implantation and development of plasmacytoma is unclear. The only other case report with an associated metallic implant involved am intramedullary nail insertion for a fractured humerus [4]. The material used in this case was not specified, but most modern nails are made of titanium, the same material from which virtually all pacemakers are constructed. A predilection of plasmacytomas for sites of trauma or scarring may also be a mechanism. There are reports of secondary cutaneous plasmacytomas occurring at sites of recent trauma in patients with established multiple myeloma [5]. Trauma in the form of central venous catheter placement, injection sites, and leg injury leading to the development of lesions has been implicated. One report describes occurrence of secondary plasmacytoma in both recent trauma and long established scar tissue [4]. In that case, a 76-year-old male, followed for 14 years with monoclonal gammopathy of undetermined significance, developed multiple myeloma after the occurrence of plasmacytomas in scar tissue 3 months after surgery for a traumatic fracture of the humerus. Local radiotherapy resulted in resolution of the lesions, but similar lesions then developed in a sternotomy scar from cardiac surgery performed 14 years previously. Systemic treatment with melphalan and prednisolone failed and the patient deteriorated.
The mechanism by which cutaneous plasmacytomas favor sites of injury or scar are not fully understood. One proposed theory involves the selection of a clonal plasma cell population expressing the CXCR4 cytokine receptor that responds to inflammatory mediators released locally in response to trauma. In all previous cases in which trauma has been followed by plasmacytoma, there was a history of multiple myeloma treated by systemic chemotherapy, part of the proposed mechanism of selecting the clonal cell population expressing the CXCR4 receptor [6]. This does not explain the varying temporal relationship between the initial trauma to development of lesions, which has varied from a few weeks to 14 years. The second theory involving chemotherapy would not be relevant in the case we describe in which the plasmocytoma was the first manifestation of disease.
To date, few cases of primary cutaneous plasmacytoma have been reported in the literature. There is therefore little evidence to guide management, but it appears that surgical excision with adjunctive radiotherapy is a reasonable strategy. The appearance of solitary vs. multiple primary lesions and lesion size may be important factors in determining prognosis. In the most recent review of case reports, 5 of 9 of patients presenting with multiple lesions died from systemic disease in contrast to 1 in 14 with solitary lesions. Patients with single lesions that did progress to systemic disease were also noted to have larger primary lesions [7].
Here we describe the first case of primary cutaneous plasmacytoma over a scar from pacemaker implantation, which was treated by radiotherapy aided by pacemaker extraction. However, development of another plasmacytoma over the site of the new implant on the contralateral chest occurred, followed by evidence of systemic disease.
References
1. International Myeloma Working Group. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 2003. Jun;121(5):749-57. [PubMed]2. Hamaker WR, Lindell ME, Gomez AC. Plasmacytoma arising in a pacemaker pocket. Ann Thorac Surg 1976 Apr;21(4):354-6. [PubMed]
3. Sasaki M, Katoh Y, Niwakawa M, Andoh A, Hodohara K, Fujiyama Y, Bamba T, Hosoda S, Kanoh T. Multiple myeloma with a mass formation in a pacemaker pocket. Rinsho Ketsueki. 1992 Nov;33(11):1747-52. [PubMed]
4. Cotter M, Enright H. Plasmacytoma occurring in scar tissue. Br J Haematol 2003 Jun;121(5):680. [PubMed]
5. Knoell KA. Rapidly enlarging mass on the leg develops after local trauma. Trauma-induced secondary cutaneous plasmacytoma (TISP). Arch Dermatol. 2010 Nov;146(11):1301-6. [PubMed]
6. Rosenblum MD, Bredeson CN, Chang CC, Rizzo JD. Subcutaneous plasmacytomas with tropism to sites of previous trauma in multiple myeloma patient treated with an autologous bone marrow transplant. Am J Haem. 2003 Apr;72(4):274-7. [PubMed]
7. Muscardin LM, Pulsoni A, Cerroni L. Primary cutaneous plasmacytoma: report of a case with review of the literature. J Am Acad Dermatol. 2000 Nov;43(5 Pt 2):962-5. [PubMed]
© 2013 Dermatology Online Journal