Acrodermatitis enteropathica
Published Web Location
https://doi.org/10.5070/D366v664n2Main Content
Acrodermatitis enteropathica
Emanual Maverakis MD, Peter J Lynch MD, and Nasim Fazel MD, DDS
Dermatology Online Journal 13 (3): 11
University of California, Davis Department of Dermatology. nasim.fazel@ucdmc.ucdavis.edu Abstract
A 13-year-old girl presented with a history of red scaly plaques involving the chest, arms and legs beginning in infancy. Punch biopsy revealed psoriasiform hyperplasia and pallor of the epidermis. The patient's serum zinc level was 36 μg/dl [nl. 66-144 μg/dl]. A diagnosis of acrodermatitis enteropathica was established and the patient responded well to zinc replacement therapy. Acrodermatitis enteropathica is a rare autosomal recessive disorder caused by mutations in SLC39A4, which encodes the tissue-specific zinc transporter ZIP4.
Clinical synopsis
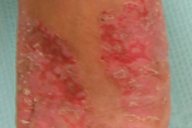
A 13-year-old girl was referred to the UC Davis dermatology clinic for evaluation of red scaly plaques covering 20 percent of her body surface area. The patient had initially presented at age 22 months at which time she was hospitalized for evaluation of a chronic diaper rash and erosions of her chest, arms, and legs. During this hospitalization she was treated with IV cefazolin and prednisone. Her serum zinc level was low and a skin biopsy was consistent with deficiency dermatitis. Zinc replacement therapy was added to her medical regimen at a dose of 21 mg daily. She was discharged after partial clinical improvement was noted. Unfortunately, she never completely cleared and experienced a waxing and waning course over the following decade. Her past medical history was otherwise unremarkable, except for poor school performance and mild obesity.
Upon presentation to the UC Davis department of dermatology at age 13, clinical examination revealed large well-defined and confluent erythematous plaques with grey to white scale over the extremities. Similar lesions were located periorally and perinasally. There was distal onycholysis of her toenails. With this clinical picture and her apparent history of unresponsiveness to zinc replacement therapy, a clinical diagnosis of psoriasis was made and the patient was started on fluocinonide (Lidex) ointment. At the time serum zinc level was checked and measured at 36 μg/dl (nl. 66-144 μg/dl). The mother was instructed to bring her daughter's zinc replacement regimen to a follow-up appointment in 1 month.
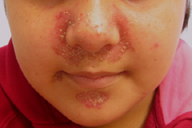
At the 1-month follow-up visit the patient's mother informed us that her daughter's dose of zinc was 147 mg of zinc sulfate per day. Interestingly, at this visit periorificial lesions, which had been absent at her last visit, were now marked (Fig. 1). Subsequent histopathologic examination revealed psoriasiform spongiotic dermatitis with focal epidermal pallor and keratinocyte necrosis (Fig. 2). Her physical exam, low serum zinc level, histopathological features, and lack of response to topical steroid lead to a conclusive diagnosis of AE. The patient's zinc replacement was increased from 147 mg zinc sulfate daily to 660 mg daily. At this dose her skin lesions almost entirely resolved within 1 month.
Comment
Acrodermatitis enteropathica is a rare autosomal recessive disorder of zinc deficiency. Although a deficiency dermatitis caused by low dietary zinc has the exact clinical and histologic features of AE, the term AE should be reserved only for genetic causes of zinc deficiency. In humans AE, first described by Danbolt and Closs in 1943 [1], is the only known hereditary defect of zinc metabolism. Individuals with AE suffer from severe zinc deficiency derived from a defective uptake of zinc in the duodenum and jejunum [2]. AE usually presents in infancy within days if an infant is bottle-fed and soon after weaning in infants breast-fed, such as our patient. Several studies have tried to explain this well characterized difference [3, 4, 5] and as of yet there is no consensus. Initially, the cause of AE was unknown and the disease was often fatal. The link between zinc deficiency and human pathology was not realized until 30 years after the initial clinical description of AE [6, 7, 8]. Homozygosity mapping of consanguineous Jordanian and Egyptian kindreds with AE localized the genetic defect to 8q24 [9]. Soon thereafter the defective gene was identified as SLC39A4 [10, 11], which encodes a ZIP protein, ZIP4. ZIP proteins are characterized by eight transmembrane domains organized into two blocks of three and five, which are separated by a histidine rich cytoplasmic metal binding site [12, 13]. The function of ZIP4 has been well characterized [14, 15, 16, 17, 18]. This tissue-specific zinc-regulated zinc transporter is abundantly expressed in enterocytes and functions to absorb dietary zinc. Zinc deficiency causes increased expression of ZIP4 whereas zinc supplementation causes decreased expression.
Zinc is the second most abundant trace metal in humans. Given that there are multiple genes encoding zinc transporters, it is surprising that there are only a few inherited diseases known to involve zinc metabolism. The only known mutation in mouse zinc metabolism is found in the lethal milk (lm) mouse. These mice have an autosomal recessive defect resulting in low breast milk levels of zinc [19, 20]. Pups of any genotype who suckle a lm/lm dam die before weaning. The defective gene in lm mice is Znt4 [21].
Interestingly, there can be a great deal of overlap between the clinical presentation of deficiency dermatitis caused by low zinc and psoriasis. For example, chronic lesions of AE may appear psoriasiform with accompanying nail dystrophy. Characteristic lesions of AE and acquired deficiency dermatitis caused by low zinc, include dry scaly eczematous plaques of the face, scalp and anogenital area, as well as generalized alopecia; none of which were present at our patient's initial clinic visit. Establishing a diagnosis of zinc deficiency may be further complicated by the fact that, zinc levels may fall during states of inflammation. The mechanism for this has not been entirely elucidated but recently IL-6 has been shown to upregulate the Zinc transporter Zip14 [22]. This upregulation, which occurs in the liver, is thought to contribute to the hypozincemia of the acute-phase response. Thus, checking a serum zinc level may not be helpful in differentiating between psoriasis and a deficiency dermatitis caused by low zinc. In addition, the zinc level may be within normal limits in deficiency dermatitis that nevertheless responds to zinc replacement therapy. A low serum alkaline phosphatase (a zinc dependent enzyme) may aid in the diagnosis of zinc deficiency and is therefore worth checking. In general the clinical presentation should raise the question of a deficiency dermatitis at which time further work-up is warranted.
Histopathologic and laboratory studies are helpful but cannot definitively make a diagnosis of zinc deficiency. The histopathologic features of psoriasis and AE (or acquired deficiency dermatitis caused by low dietary zinc) can also be very similar. For example, psoriasiform hyperplasia is a common feature of zinc deficiency dermatitis. Other characteristic histopathologic features of zinc deficiency dermatitis include pallor of the upper part of the epidermis due to intracellular edema, diminution of the granular layer and focal dyskeratosis.
For AE, zinc replacement therapy should be started at 3 mg/kg/day of elemental zinc (there is 50 mg of elemental zinc per 220 mg zinc sulfate). Serum zinc levels should be monitored and the dose of zinc sulfate should be adjusted appropriately. Patients may require a higher dose than 3mg/kg/day of zinc sulfate to normalize their genetic defect of zinc metabolism. In deficiency dermatitis caused by low dietary zinc, replacement therapy should be initiated at 1mg/kg/day of elemental zinc.
References
1. Danbolt, N., and K. Closs. 1943. Acrodermatitis enteropathica. Acta Derm Venereol 23:172.2. Lombeck T, Schnippering HG, Ritzl F, Feinendegen LE, Bremer HJ. Letter: Absorption of zinc in acrodermatitis enteropathica. Lancet. 1975 Apr 12;1(7911):855. PubMed
3. Eckhert CD, Sloan MV, Duncan JR, Hurley LS. Zinc binding: a difference between human and bovine milk. Science. 1977 Feb 25;195(4280):789-90. PubMed
4. Hurley LS, Duncan JR, Sloan MV, Eckhert CD. Zinc-binding ligands in milk and intestine: a role in neonatal nutrition? Proc Natl Acad Sci U S A. 1977 Aug;74(8):3547-9. PubMed
5. Cousins RJ, Smith KT. Zinc-binding properties of bovine and human milk in vitro: influence of changes in zinc content. Am J Clin Nutr. 1980 May;33(5):1083-7. PubMed
6. Barnes PM, Moynahan EJ. Zinc deficiency in acrodermatitis enteropathica: multiple dietary intolerance treated with synthetic diet. Proc R Soc Med. 1973 Apr;66(4):327-9. PubMed
7. Moynahan EJ, Barnes PM. Zinc deficiency and a synthetic diet for lactose intolerance. Lancet. 1973 Mar 24;1(7804):676-7. PubMed
8. Neldner KH, Hambidge KM. Zinc therapy of acrodermatitis enteropathica. N Engl J Med. 1975 Apr 24;292(17):879-82. PubMed
9. Wang K, Pugh EW, Griffen S, Doheny KF, Mostafa WZ, al-Aboosi MM, el-Shanti H, Gitschier J. Homozygosity mapping places the acrodermatitis enteropathica gene on chromosomal region 8q24.3. Am J Hum Genet. 2001 Apr;68(4):1055-60. Epub 2001 Mar 12. PubMed
10. Kury S, Dreno B, Bezieau S, Giraudet S, Kharfi M, Kamoun R, Moisan JP. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat Genet. 2002 Jul;31(3):239-40. Epub 2002 Jun 17. PubMed
11. Wang K, Zhou B, Kuo YM, Zemansky J, Gitschier J. A novel member of a zinc transporter family is defective in acrodermatitis enteropathica. Am J Hum Genet. 2002 Jul;71(1):66-73. Epub 2002 May 24. PubMed
12. Grotz N, Fox T, Connolly E, Park W, Guerinot ML, Eide D. Identification of a family of zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc Natl Acad Sci U S A. 1998 Jun 9;95(12):7220-4. PubMed
13. Zhao H, Eide D. The yeast ZRT1 gene encodes the zinc transporter protein of a high-affinity uptake system induced by zinc limitation. Proc Natl Acad Sci U S A. 1996 Mar 19;93(6):2454-8. PubMed
14. Dufner-Beattie J, Wang F, Kuo YM, Gitschier J, Eide D, Andrews GK. The acrodermatitis enteropathica gene ZIP4 encodes a tissue-specific, zinc-regulated zinc transporter in mice. J Biol Chem. 2003 Aug 29;278(35):33474-81. Epub 2003 Jun 11. PubMed
15. Kim BE, Wang F, Dufner-Beattie J, Andrews GK, Eide DJ, Petris MJ. Zn2+-stimulated endocytosis of the mZIP4 zinc transporter regulates its location at the plasma membrane. J Biol Chem. 2004 Feb 6;279(6):4523-30. Epub 2003 Nov 11. PubMed
16. Wang F, Kim BE, Dufner-Beattie J, Petris MJ, Andrews G, Eide DJ. Acrodermatitis enteropathica mutations affect transport activity, localization and zinc-responsive trafficking of the mouse ZIP4 zinc transporter. Hum Mol Genet. 2004 Mar 1;13(5):563-71. Epub 2004 Jan 6. PubMed
17. Dufner-Beattie J, Kuo YM, Gitschier J, Andrews GK. The adaptive response to dietary zinc in mice involves the differential cellular localization and zinc regulation of the zinc transporters ZIP4 and ZIP5. J Biol Chem. 2004 Nov 19;279(47):49082-90. Epub 2004 Sep 9. PubMed
18. Liuzzi JP, Bobo JA, Lichten LA, Samuelson DA, Cousins RJ. Responsive transporter genes within the murine intestinal-pancreatic axis form a basis of zinc homeostasis. Proc Natl Acad Sci U S A. 2004 Oct 5;101(40):14355-60. Epub 2004 Sep 20. PubMed
19. Piletz JA, Ganschow RE. Lethal milk mutation results in dietary zinc deficiency in nursing mice. Am J Clin Nutr. 1978 Apr;31(4):560-2. No abstract available. PubMed
20. Piletz JE, Ganschow RE. Zinc deficiency in murine milk underlies expression of the lethal milk (lm) mutation. Science. 1978 Jan 13;199(4325):181-3. PubMed
21. Huang L, Gitschier J. A novel gene involved in zinc transport is deficient in the lethal milk mouse. Nat Genet. 1997 Nov;17(3):292-7. PubMed
22. Liuzzi JP, Lichten LA, Rivera S, Blanchard RK, Aydemir TB, Knutson MD, Ganz T, Cousins RJ. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute-phase response. Proc Natl Acad Sci U S A. 2005 May 10;102(19):6843-8. Epub 2005 Apr 29. PubMed
© 2007 Dermatology Online Journal