Imported paracoccidioidomycosis in Japan
Published Web Location
https://doi.org/10.5070/D363s4b4zqMain Content
Letter: Imported paracoccidioidomycosis in Japan
Hiroshi Onda, Mayumi Komine, Satoru Murata, Mamitaro Ohtsuki
Dermatology Online Journal 17 (12): 11
Department of Dermatology, Jichi Medical University, JapanAbstract
A 39-year-old man from Argentina, who had come to Japan 13 years previously, had been suffering from oral pain for several months. He was biopsied twice and treated with oral predonisolone without improvement. A number of white granular lesions with erosions were observed on the hard palate and buccal mucosa. Histopathological examination revealed a well-demarcated abscess with an accumulation of neutrophils, surrounded by epithelioid cell granulomas. Round eosinophilic bodies, considered to be fungal elements, positive for PAS and Grocott staining, were observed. Chest CT revealed cavities in the lung. A white yeast-like colony was cultured from bronchial lavage fluid and Paracoccidioides brasiliensis was identified. The patient was treated with liposomal amphotericin B followed by oral itraconazole 400 mg/day, with a favorable clinical course. Paracoccidioidomycosis, an imported mycosis, is rare in Japan and sometimes causes difficulty in diagnosis, resulting in inappropriate treatment.
Introduction
Imported mycoses in Japan, including paracoccidioidomycosis, are rare and sometimes cause difficulty in diagnosis. We encountered a case of paracoccidioidomycosis with typical symptoms, treated inappropriately for five months because of a lack of awareness of this rare disease.
Case report
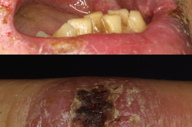
A 39-year-old man born in Argentina, who had come to Japan 13 years previously, visited our hospital in June, 2006. He had oral pain and had noticed skin eruptions on a left finger in January. He visited a dentist and underwent biopsies twice without a definite diagnosis. Prednisolone was started at 30 mg daily, without improvement. Clinical examination revealed erosion with crusts on a swollen lip. White granular lesions with erosions were observed on the oral mucosa (Figure 1a). A red-purple nodule lesion was observed on the left middle finger (Figure 1b). No laboratory abnormalities were noted, except for elevated white blood cell counts and mild elevation of C-reactive protein.
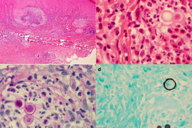
A biopsy specimen from the finger showed marked epidermal thickening and abscess in the upper dermis (Figure 2a). The accumulation of neutrophils within the abscess was observed, surrounded by epithelioid cell granulomata containing numerous multinucleated giant cells intermingled with lymphocytes and histiocytes. Round yeast-like cells of 12 to 13 μm diameter, fungal elements (Figure 2b), were noted and were positive for PAS (Figure 2c) and Grocott (Figure 2d) staining.
The yeast cells have thick walls, birefringent by H&E staining, and positive for PAS staining. Grocott staining showed multiple budding yeast cells; the daughter cells differed in size and were connected by small bridges. The differential diagnosis of yeast cells seen in H&E and PAS staining includes Blastomyces dermatitidis, but these multiple narrow-based budding yeast cells visualized by Grocott staining were characteristic of Paracoccidioides brasiliensis, referred to as Mickey mouse heads or marine pilot’s wheel. Similar findings were noted in a mucosal biopsy specimen.
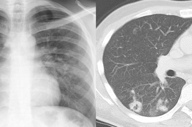
Chest X-ray showed diffuse infiltrative shadows in both lung fields, whereas chest computed tomography (CT) showed a cavity and linear, reticular shadows (Figure 3).
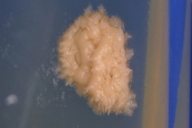
Bronchial lavage fluid culture for 2 months in Sabouraud dextrose agar at room temperature revealed white yeast-like creamy colonies, elevated and wrinkled, with a smooth surface. The isolated fungus showed yeast cells with multiple budding, like a marine pilot’s wheel, as seen in Grocott staining of skin and mucosa. Considering these findings and the clinical history, we diagnosed this case as infection with Paracoccidioides brasiliensis.
Liposomal amphotericin B, 200 mg/day, was intravenously administered for 18 days, followed by oral itraconazole, 400 mg/day for 8 weeks, 300 mg/day for 8 weeks, and 200 mg/day for 22 weeks. There was no sign of recurrence when he was seen at our outpatient clinic in December 2008.
Discussion
Paracoccidioidomycosis is caused by fungi, which are not native flora of Japan [1]. Because of its rarity in Japan, a proper diagnosis is often difficult and inappropriate treatment sometimes worsens the disease. Similar difficulties have been reported in Germany [2] and the Netherlands [3].
Only 20 cases (male 18, female 2) of imported paracoccidioidomycosis in Japan have been reported exclusively in Japanese citizens, except for one suspected case by Koya [4] (Table 1). Kamei K [1] and Fujio J [5] summarized the reported cases in the English literature as reviews. Sano et al [6] reported that estrogen inhibited the growth of P. brasiliensis, which may cause the male predominance. The causative agent, P. brasiliensis, has been isolated from soil and its endemic areas are limited to Central and South America. P. brasiliensis infects via an airborne route, forms lesions in the lungs, and disseminates to other tissues, such as the mucus membranes, skin, and lymph nodes. Our case was diagnosed by the presence of a skin lesion, which is relatively rare among the previously reported cases. P. brasiliensis is known to show two types of colonies depending on temperature and media. They usually show mycelial colonies with a white cotton-like surface when cultured in Sabouraud dextrose agar at room temperature, whereas they show yeast-like colonies when cultured in brain heart infusion (BHI) medium at 37°Celsius. It has been reported that there are yeast form variants, which show yeast-like colonies even when cultured in Sabouraud dextrose media at room temperature [7]; this may apply to our case (Table 1).
References
1. Kamei K, Sano A, Kikuchi K, et al. The trend in imported mycoses in Japan. J Infect Chemother. 2003 Mar; 9(1):16-20. [PubMed]2. Horre R, Schumacher G, Alpers K, Seitz HM, Adler S, Lemmer K, de Hoog GS, Schaal KP, Tintelnot K. a case of imported paracoccidioidomycosis in a German legionnaire. Med Mycol. 2002 Apr; 40(2): 213-216. [PubMed]
3. Van Damme PA, Bierenbroodspot F, Telgtt DS, Kwakman JM, De Wilde PC, Meis JF. A case of imported paracoccidioidomycosis: an awkward infection in the Netherlands. Med Mycol. 2006 Feb; 44(1): 13-18. [PubMed]
4. Koya G. An autopsy case of mixed infection of Leishmania dovonani and blastomyces brasiliensis with emphasis on the histopathogenesis of reticuloendotheliosis. Acta Pathol Jpn. 1964 May; 14: 223-229. [PubMed]
5. Fujio J, Nishimura K, Miyaji M. Epidemiological survey of the imported mycoses in Japan. Jpn J Med Mycol. 1999; 40(2):103-109. [PubMed]
6. Sano A, Miyaji M, Nishimura K. Studies on the relationship between the estrous cycle of BALB/c mice and their resistance to paracoccidioides brasiliensis infection. Mycopathologia. 1992; Sep119(3):141-145. [PubMed]
7. Villar LA, Salazar ME, Restrepo A. Morphological study of a variant of Paracoccidioides brasiliensis that exists in the yeast form at room temperature. J Med Vet Mycol. 1988; 26(5): 269-276. [PubMed]
© 2011 Dermatology Online Journal