Cutaneous infection in a renal transplant patient: A case report
Published Web Location
https://doi.org/10.5070/D35tc8p38jMain Content
Cutaneous pseudallescheria boydii infection in a renal transplant patient: A case report
José Carlos Cardoso, David Serra, Raquel Cardoso, José Pedro Reis, Óscar Tellechea, Américo Figueiredo
Dermatology Online Journal 15 (10): 8
Serviço de Dermatologia, Hospitais da Universidade de Coimbra, Coimbra, Portugal. ze_carlos_cardoso@yahoo.com.brAbstract
We describe the case of a 59-year-old male renal transplant recipient who presented with a 1 month history of nodular, erythematous, and crusted lesions on the anterior surface of the left leg. There was no history of trauma. The patient had no systemic signs or symptoms. A skin biopsy revealed evidence of a deep fungal infection, with septate and branching hyphae amongst the dermal inflammatory infiltrate, but the culture was negative. Initial treatment with itraconazole proved ineffective and new lesions appeared. New tissue samples were obtained for culture, allowing the isolation of Pseudallescheria boydii. The patient underwent surgical excision of the lesions combined with voriconazole 400 mg/day for 2 months. After this period no new lesions appeared and the patient has remained without recurrence after 2 years of follow-up. Pseudallescheria boydii is an opportunistic fungus that can cause systemic infection mainly in immunocompromised patients that manifests as pulmonary, osteoarticular, ocular, vascular, cutaneous or central nervous system disease. Resistance to therapy can occur with infection progression and high mortality. Treatment with the combination of an antifungal agent, mainly voriconazole, and surgery, when feasible, probably provide the best results in cutaneous infections.
Introduction
In the last few decades infections caused by opportunistic agents, particularly certain species of fungi, have gained importance because of the increasing number of immunocompromised patients, namely transplant recipients [1].
Pseudallescheria boydii (the teleomorphic form of Scedosporium apiospermum) is a ubiquitous fungus known to be an agent of mycetoma in immunocompetent individuals. The incidence of this infection has become elevated in parallel with the increased numbers of immunocompromised patients, particularly solid organ transplant recipients [2, 3, 4, 5]. Along with the potential seriousness of the disease caused by this agent, with potential disseminated disease and fatal outcome, its importance relates also to the facts that it morphologically resembles other fungi (Aspergillus sp.), and it shows resistance to multiple antifungal agents, including amphotericin B [2, 3, 5, 6].
We report the case of a renal transplant recipient with cutaneous infection caused by Pseudallescheria boydii.
Case report
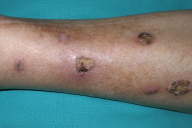
A 59-year-old male patient, renal transplant recipient for about 4 years, presented with a 1-month history of three lesions on the anterior aspect of his left leg. The lesions were asymptomatic and the patient had no systemic signs or symptoms. There was no history of previous trauma. His past medical history was remarkable for hypertension, ischemic heart disease and chronic renal failure. Beginning in 2001 he required dialysis and underwent renal transplantation in 2004. His usual medication included tacrolimus, mycophenolate mofetil, prednisolone, valsartan, furosemide, and omeprazole. By the time of our first observation he had already been treated with oral cefaclor and topical fusidic acid, with no response. Examination disclosed four erythematous, violaceous exudative nodules on the anterior aspect of the left leg, in a roughly linear, sporotrichoid-like, distribution (Fig. 1).
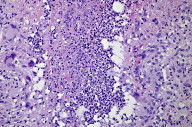
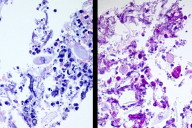
Histological examination of one lesion revealed a granulomatous dermal infiltrate composed of lymphomononucleate and polymorphonuclear cells (Fig. 2). PAS staining disclosed a small number of septate and branching hyphae in the deep dermis (Fig. 3). Fite-Faraco staining was negative. Mycological and mycobacterial cultures were negative. Empiric treatment with itraconazole was started at 100 mg twice a day.
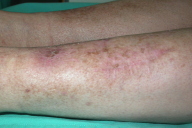
After 2 months of treatment, there was no improvement and new lesions had appeared on the same leg. New tissue samples for exhaustive microbiological studies were collected and allowed the isolation of Pseudallescheria boydii (confirmed in 2 samples). The patient underwent surgical excision of the lesions combined with voriconazole 400 mg/day for two months, with a good response (Fig. 4). The only side effect was a transient decrease in renal function associated with increased tacrolimus levels, due to interaction with voriconazole, which completely reversed after dose adjustments.
No recurrence was seen after 2.5 years of follow-up.
Discussion
The variety of agents and possible manifestations of infections in immunocompromised patients requires an tissue biopsies for histopathology and culture. The present case is a good example that highlights some important issues.
First, the clinical picture of P. boydii infection is quite variable and can include a sporothricoid-like distribution of lesions. This has been reported in the literature, but is more commonly associated with other agents (namely sporotrichosis and some atypical mycobacterial infections) [3, 7].
Second, the morphological characteristics of P. boydii in tissue biopsies include the formation of septate hyphae branching at 45º angles, morphologically similar to Aspergillus [2, 3, 5, 8] and, to a lesser extent, Fusarium [8, 9]; this can be a pitfall in the diagnosis. We highlight here the importance of the use of repeated cultures (or other techniques, when available), especially in cases of therapeutic failure, as in our patient.
Finally, P. boydii is frequently resistant to multiple antifungal agents, including those usually used in the treatment of systemic mycoses in immunocompromised hosts, most notably amphotericin B [2, 3, 5, 6]. In vitro susceptibility studies have found voriconazole to be the most effective drug, followed by posaconazole [10, 11]. Accumulated clinical experience seems to support voriconazole efficacy, with several reports of good response to this drug. However, to our knowledge, controlled studies are lacking [9,12, 13, 14]. Surgical treatment, whenever possible, seems to be of value: there are several reports of successful treatment combining antifungal and surgery, for cutaneous but also extracutaneous disease [14, 15, 16].
In conclusion, the accurate diagnosis and early treatment are extremely important in the management of fungal infections in immunocompromised patients, in order to avoid dissemination of the disease and a consequent unfavorable prognosis.
References
1. Virgili A, Zampino MR, Mantovani L. Fungal skin infections in organ transplant recipients. Am J Clin Dermatol. 2002;3:19-35. [PubMed]2. O'Brian TA. Pseudallescheriasis in the 21st century. Expert Rev Anti Infect Ther. Medsurg Nurs. 2005;3:765-73. [PubMed]
3. Panackal AA, Marr KA. Scedosporium/Pseudallescheria infections. Semin Respir Crit Care Med. 2004;25:171-81. [PubMed]
4. Chaveiro MA, Vieira R, Cardoso J, Afonso A. Cutaneous infection due to Scedosporium apiospermium in an immunosuppressed patient. J Euro Acad Dermatol Venereol. 2003;17:47-9. [PubMed]
5. Bernstein EF, Schuster MG, Stieritz DD, Heuman PC, Uitto J. Disseminated cutaneous Pseudallescheria boydii. Br J Dermatol. 1995;132:456-60. [PubMed]
6. Castiglioni B, Sutton DA, Rinaldi MG, Fung J, Kusne S. Pseudallescheria boydii (anamorph Scedosporium apiospermum) infection in solid organ transplant recipients in a tertiary medical centre and review of the literature. Medicine. 2002;81:333-48. [PubMed]
7. Hagari Y, Ishioka S, Ohyama F, Mihara M. Cutaneous infection showing sporotrichoid spread caused by Pseudallescheria boydii (Scedosporium apiospermum): Successful detection of fungal DNA in formalin-fixed, paraffin-embedded sections by seminested PCR. Arch Dermatol. 2002;138:271-2. [PubMed]
8. Hayden RT, Isotalo PA, Parret T, Wolk DM, Qian X, Roberts GD, Lloyd RV. In situ hybridization for the differentiation of Aspergillus, Fusarium, and Pseudallescheria species in tissue section. Diagn Mol Pathol. 2003;12:21-6. [PubMed]
9. Montejo M, Muñiz ML, Zárraga S, Aguirrebengoa, Amenabar JJ, López-Soria L, Gonzalez R. Case reports. Infection due to Scedosporium apiospermum in renal transplant recipients: a report of two cases and literature review of central nervous system and cutaneous infection by Pseudallescheria boydii/Sc. apiospermum. Mycoses. 2002;45:418-27. [PubMed]
10. Gil-Lamaignere C, Roilides EMosquera J, Maloukou A, Walsch TJ. Antifungal triazoles and polimorphonuclear leukocytes synergize to cause increased hyphal damage to Scedosporium prolificans e Scedosporium apiospermum. Antimicrob Agents Chemother. 2002;46:2234-37. [PubMed]
11. Gilgado F, Serena C, Cano J, Gené J, Guarro J. Antifungal susceptibilities of the species Pseudallescheria boydii complex. Antimicrob Agents Chemother. 2006;50:4211-13. [PubMed]
12. Rogasi PG, Zanazzi M, Nocentini J, Fantoni E, Trotta M, Faggi E, Fontanelli A, Bertoni E, Salvadori M, Leoncini F. Disseminated Scedosporium apiospermum infection in renal transplant recipient: Long-term successful treatment with voriconazole: A case report. Transpl Proceed. 2007;39:2033-35. [PubMed]
13. Lainščak M, Hočevar A, Logar D, Beović B, Matos T, Tomšič M. Subcutaneous infection with Pseudallescheria boydii in an immunocompromised patient. Clin Rheumatol. 2007;26:1023-4. [PubMed]
14. Chakraborty A, Workman MR, Bullock PR. Scedosporium apiospermum brain abcess treated with surgery and voriconazole. J Neurosurg (Pediatrics 1). 2005;103:83-7. [PubMed]
15. Ginter G, Petutschnig B, Pierer G, Soyer HP, Reischle S, Kern T, Hoog S. Case report: Atypical cutaneous pseudallescheriosis refractory to antifungal agents. Mycoses. 1999;42:507-11. [PubMed]
16. Lopes JO, Alves SH, Benevenga JP, Salla A, Khmonhan C, Silva CB. Subcutaneous Pseudallescheriasis in a renal transplant recipient. Mycopathologia. 1994;125:153-6. [PubMed]
© 2009 Dermatology Online Journal