A review of thalidomide's history and current dermatological applications
Published Web Location
https://doi.org/10.5070/D35fk5w0qvMain Content
A review of thalidomide's history and current dermatological applications
Anthony J Perri III, and Sylvia Hsu MD
Dermatology Online Journal 9 (3): 5
From the Department of Dermatology, Baylor College of Medicine, Houston, TX Abstract
Once abandoned because of devastating teratogenic effects, thalidomide has reemerged as an alternative treatment in many dermatologic diseases. In 1998, thalidomide became FDA approved for the acute treatment and suppression of the cutaneous manifestations of erythema nodosum leprosum (ENL). ENL is a systemic disorder that typically occurs after several years of antileprosy treatments, usually for lepromatous leprosy. Off-label uses for thalidomide include: aphthous stomatitis, Behçet disease, pyoderma gangrenosum, chronic discoid lupus erythematosus, systemic lupus erythematosus, lichen planus, prurigo nodularis and sarcoidosis. This review examines the background, pharmacokinetics, mechanism of action, side-effects, and indications of thalidomide.
Background
In 1956, thalidomide was introduced by Chemie Grunenthal, a West German pharmaceutical company, as a sedative and was marketed under the name of Contergan. [1, 2, 3, 4] By 1958, it permeated the world market, and its use expanded from simply combating insomnia. Pregnant women frequently treated their nausea of pregnancy with thalidomide. [3] Soon thalidomide's teratogenic effects became apparent, and it was subsequently withdrawn from the market in 1961. Fortunately, thalidomide had never been approved in the United States because of concerns about the development of peripheral neuropathies. [1, 3] Thalidomide resurfaced in 1965 when Sheskin, a dermatologist from Israel, made a fortuitous discovery while treating his leprosy patients with thalidomide. He was prescribing thalidomide for its sedative properties, but he noticed that those with erythema nodosum leprosum had resolution or improvement of their skin lesions within 2 days of beginning treatment. This discovery has led to continued interest and research into thalidomide's immunomodulatory activities. [3]
Pharmacokinetics
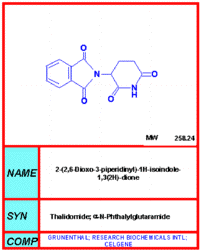 |
| Figure 1 |
|---|
Thalidomide is a derivative of glutamic acid. It consists of a left-sided phthalimide ring and a right-sided glutarimide ring (Fig. 1). The left-sided phthalimide ring is thought to be responsible for the teratogenic effects of thalidomide. [2] The right-sided glutarimide ring is similar in structure to other hypnotic drugs. It is believed that this portion of thalidomide mediates its sedative properties. [1, 3, 5] Thalidomide's R and L enantiomers rapidly interconvert so it can be administered as a racemic mixture. [1, 3]
Thalidomide is slowly absorbed from the gastrointestinal tract. Peak plasma concentrations occur approximately 2.9 to 5.7 hours after the dose is administered. [3] Food does not affect absorption or peak concentrations. However, a high-fat meal can delay the time to peak concentrations by several hours. [5] The drug distributes extensively throughout the bodily fluids and tissues. [6] It has been found in semen; thus male patients on thalidomide must use birth control if engaging in sexual activity with a female partner of childbearing potential. [7] It is unclear whether breast milk contains thalidomide, so it is recommended that women do not breast feed while taking this drug. The drug is mainly metabolized through a nonenzymatic pathway in which it undergoes spontaneous hydrolysis within the blood and tissues. [1] Although not hepatically metabolized, a minute quantity of the drug is metabolized by the hepatic cytochrome P450 system. [1, 3, 5] The mean elimination time is 5-7 hours, and it is not excreted renally; less than .7 percent of the drug is found in the urine. [1]
Mechanism of action
The exact mechanism of action of thalidomide has not yet been clearly delineated; however, it is believed to be related to its anti-inflammatory, immunomodulatory and anti-angiogenic properties. [1, 3, 5, 8] Its anti-inflammatory properties include the inhibition of chemotaxis of both lymphocytes and neutrophils. Additionally, the drug inhibits phagocytosis by neutrophils and macrophages. [1, 3, 5, 8, 9] In terms of immunomodulatory properties, thalidomide decreases the levels of the cytokines, TNF-α and IFN-γ. Thalidomide's effect on TNF-α is mediated by accelerating the degradation of its mRNA. [1, 3, 5, 8, 9] It does not completely eliminate the mRNA of TNF-α but decreases the half-life from 30 minutes to 17 minutes. [8] In erythema nodosum leprosum, TNF-α and IFN-γ are usually elevated and clinical improvement of patients treated with thalidomide corresponds with the reduction of these two cytokines. [1] Also, thalidomide has been found to decrease the helper T-cell to suppressor T-cell ratio. [1, 3, 5] This observation also has implications on the treatment of erythema nodosum leprosum because helper T-cells are usually elevated in the skin lesions of these patients during acute episodes. [10] The drug has also been shown to enhance TH2 cytokines like IL-4 and IL-5. [1, 3, 5, 8] Finally, thalidomide's antiangiogenic properties have been correlated with its ability to treat some forms of cancer. [1]
Side-effects and drug interactions
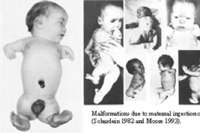 |
| Figure 2 |
|---|
Teratogenicity is thalidomide's most severe toxicity, and it is labeled as pregnancy category X. It is lipid soluble and readily crosses the placenta, so it should never be taken by pregnant women or those who could become pregnant. [5] Even one dose of a 50 mg capsule can cause severe birth defects. The teratogenic risk is highest during the critical period, which is days 20-40 of gestation or days 35-50 after the last menstrual period. [3] The risk of additional, potentially severe birth defects outside the critical period is unknown but may be significant. Therefore, women should not use this drug any time during pregnancy. Phocomelia is a very common birth defect seen with thalidomide use. It is characterized by defective, shortened limbs resulting in flipper hands and feet (Fig. 2). [8, 11] In more severe cases, the complete absence of limbs can occur. Additionally, the fetus can develop external ear abnormalities, hypoplastic or completely absent bones, facial palsy, eye abnormalities, and gastrointestinal and genitourinary tract malformations. [1, 8, 11] Approximately 40 percent of exposed fetuses die at or shortly after birth, with bowel atresia being the most common cause of death. [11] There are over 24 different proposed mechanisms for the teratogenicity. Currently, the most widely held theories involve either thalidomide's anti-angiogenic effect or its direct toxic effect on the segmental sensory neurons, with resulting limb deformities. [10] The McCredie-McBride hypothesis states that neural tissue normally has an inductive effect on the development of the limb, and because neural tissue is damaged by thalidomide, the limb bud subsequently becomes malformed. [3, 10]
|
To prevent pregnancy while taking thalidomide, Celgene requires all physicians prescribing the drug to follow the Enhanced STEPS program (System for Thalidomide Education and Prescribing Safety). [Website: http://www.celgene.com/steps/] Thalidomide can be prescribed only 28 days at a time with no refills. A prescription is valid for only 1 week. During the first month, it is recommended to obtain a pregnancy test within 24 hours before writing a prescription as well as once a week. After the first month, monthly pregnancy tests can then be administered within 24 hours before writing the prescription. No pregnancies have occurred while a patient has been on thalidomide since it again became available in 1998. If a pregnancy does occur, the FDA and Celgene should be contacted and the patient should be seen by an obstetrician specializing in reproductive toxicity.
Thalidomide does not have any acute toxicity; a fatal overdose is virtually impossible. [5] Peripheral neuropathy is probably the most limiting factor when prescribing thalidomide. Some studies have shown a dose dependence of the peripheral neuropathy, with it typically occurring after a cumulative dose of 40-50 grams. Other studies refute this finding, showing neuropathy beginning at a cumulative dose of just 3-6 grams in some patients. [1] On electron microscopy, thalidomide induced neuropathies demonstrate an increase in the number of thin, unmyelinated axons and a decrease in the number of thick, myelinated fibers. [2] This morphological finding may account for signs and symptoms of thalidomide-induced neuropathy. Patients may complain of tingling in their hands and feet, muscle cramps, and proximal lower limb weakness. [1] On neurological exam, a symmetric sensory loss in the feet and hands is usually found. Typically, it is more prevalent in the feet and is more a sensory than a motor neuropathy. [1, 3] Thalidomide must be discontinued if any signs or symptoms of neuropathy develop and can be restarted only if the symptoms of and tests for neuropathy return to baseline. In one retrospective study from France, subjective symptoms of neuropathy always preceded the objective clinical signs. [12] When following patients on thalidomide, it is imperative to discuss the symptoms of neuropathy and urge them to immediately report any findings. It is recommended to obtain a baseline neurologic exam performed by a neurologist with followup examinations monthly for the first 3 months and then every 6 months. It has also been suggested that patients receive a baseline electrophysiologic exam because this test has a wide range of normative values. Thus, a patient can be monitored for any deviation from his baseline findings. [13] Measurement of SNAP (sensory nerve action potential) amplitudes of the sural, median, and radial nerves at baseline and every 6 months, or with each 10 g. increase in cumulative dose is recommended. [14] More frequent testing is needed if there is a greater than 30 percent decrease in the SNAP amplitude. [3, 9, 14] The drug should be stopped with a 40 percent decrease and restarted only if the SNAP amplitudes return to normal. [3, 14]
Somnolence, thalidomide's first indication, is now one of its most common side-effects. The incidence has been reported in as high as 11 percnet and commonly occurs when doses are over 200 mg per day. This side-effect can be managed by giving it as a single dose at night and warning patients not to engage in activities where somnolence could be hazardous. Also if the dosage must be increased, it should be slowly increased by 100-200 mg per day every 1-2 weeks. It is believed that thalidomide's sedative properties arise from a different mechanism than that seen with other hypnotic drugs. Thalidomide does not cause incoordination or respiratory depression. [1, 3] It activates the sleep center in the forebrain, unlike barbiturates, which act on the reticular activating system resulting in sedation as well as loss of consciousness, muscle relaxation, and reflex suppression. [1, 3, 5]
Constipation can occur in up to a third of patients. [1] It is usually mild but bloating can develop and relief may not be provided with medications. Thus, this side-effect can lead to noncompliance with thalidomide therapy.
A hypersensitivity reaction has also been noted. [1] Typically, it presents 2-10 days after the onset of treatment as a morbilliform eruption on the trunk and proximal extremities, sparing the groin. It can easily be treated with antihistamines, but, if the drug is continued, some patients can worsen and develop fever, tachycardia, and hypertension.
Neutropenia is a very rare side-effect occurring in less than 1 percent of patients. Its incidence is higher in HIV patients, but it is reversible when the drug is discontinued. Patients are monitored with a baseline complete blood-cell count with differential and the drug is not begun if the absolute neutrophil count is less than 750/mm3. The values should be monitored monthly and more often in HIV patients.
Thalidomide has been tested in vitro and ex vivo (animal systems) and has been shown to be nonmutagenic. However, long-term carcinogenicity tests have not been done.
Patients on thalidomide must employ some form of birth control, so it is important to note that it does not decrease the efficacy of birth-control pills. It should be used with caution with other drugs that are CNS depressants: barbiturates, chlorpromazine, alcohol, and reserpine. [1]
Indications
The only FDA-approved indication for thalidomide is the acute treatment and suppression of the cutaneous manifestations of erythema nodosum leprosum (ENL). ENL is a systemic disorder that typically occurs after several years of anti-leprosy treatments, usually for lepromatous leprosy. Most patients are afflicted with fever, weight loss, malaise, muscle weakness, clawed hands, progressive synovitis, nephritis, iritis, lympadenitis, plantar ulcers, arthralgias, and leukocytosis. [1] Additionally, they can develop painful, erythematous nodules of the skin and subcutaneous tissue, which are believed to be a vasculitis or panniculitis associated with immune-complex deposition and elevated levels of TNF-α and IFN-γ. [1] The skin lesions can lead to scarring and can resolve with post-inflammatory hyperpigmentation or hypopigmentation. In July 1998, thalidomide was approved as the treatment of choice for ENL. Although no trials have been conducted comparing its efficacy with corticosteroids or clofazimine, [5] response rates for thalidomide have been greater than 90 percent, with improvement seen within days and complete resolution within 2 weeks. [15] The other symptoms of ENL also responded rapidly. [8] The most common side-effects were somnolence and constipation. In these trials, all the adverse events were mild to moderate and none resulted in discontinuation of the drug. Long-term followup data have shown a reduction in the rates of ENL-induced reactional neuritis and polyneuritis. [8] Peripheral neuropathy was a very rare side-effect in patients with ENL. It has been suggested that, because ENL patients typically have a baseline neuropathy prior to beginning thalidomide, perhaps thalidomide-induced neuropathy is under reported. Also, it has been surmised that patients who show improvement on thalidomide for dermatological conditions may downplay their neuropathy. [13] If a moderate or severe neuropathy develops, steroids can be administered and tapered as the neuropathy improves. The recommended dosage is to begin with 100-400 mg at night and continue with this dose until the symptoms subside. [8] Thereafter, tapering by 50 mg every 2-4 weeks is recommended. [8] Patients can be maintained on 25-200 mg a day to prevent an ENL recurrence. [16]
Off-label uses
|
Thalidomide has been shown to be efficacious in the treatment of recurrent aphthous stomatitis. [1, 3, 8, 17] Aphthous stomatitis is the most common ulcerative disease of the oral mucosa and is characterized by localized painful, relapsing ulcers. [18] One mechanism by which thalidomide works in aphthous stomatitis is by inhibiting the increased chemotactic response of neutrophils. [5]
In a large multicenter, double-blinded, crossover study from France, over 50 percent of the patients using thalidomide had complete resolution of their aphthous ulcers. However, when they switched over to the placebo group, almost all of them had a recurrence of their ulcers. Thus, it was concluded that thalidomide's effect on apthous stomatitis is only suppressive. [19]
Behçet disease (BD) has also been treated with thalidomide. [1 3, 20, 21, 22] BD is a type of systemic vasculitis with recurrent oral and genital ulcers, skin lesions (papules, pustules, or erythema nodosum lesions), panuveitis, or arthritis. [21, 22] BD has a high mortality because of occasional pulmonary and central nervous system involvement. [23] There are several clinical studies and many case reports of thalidomide treatment for BD. A randomized, doubleblind placebo-controlled study from Turkey showed rapid healing of the orogenital ulcers while patients are on thalidomide. However, the ulcers return when thalidomide is discontinued. Thus, its effect on BD appears to be only suppressive. Patients should be titrated to the lowest efficacious maintenance dose once a desirable response has been attained. [22] Some studies have actually shown an increase in the occurrence of erythema nodosum, but one study has shown complete regression of erythema nodosum. [24]
Pyoderma gangrenosum is a chronic skin disease that presents with painful nodules that can ulcerate. It is a diagnosis of exclusion when other causes of ulceration, such as infection and neoplasia, have been excluded. [25] There are no clinical trials or studies evaluating the efficacy of thalidomide for pyoderma gangrenosum, but multiple case studies suggest that thalidomide can be useful when other treatments have failed. [9, 25]
Thalidomide has also been useful in treating chronic discoid lupus erythematosus (CDLE). [1, 4, 26, 27] The gold standard of treatment for CDLE is avoidance of UV exposure, topical corticosteroids, and antimalarials. [4] However, when this approach fails, thalidomide has been shown to be effective in suppressing this disease. [27] CDLE has a high incidence of relapse when thalidomide is stopped, so it is suggested to administer it in a low maintenance dose over a long period of time. [4]
There have not been any controlled clinical trials for thalidomide and systemic lupus erythematosus (SLE), but numerous clinical studies have shown some efficacy. [28, 29] The majority of patients in these studies were refractory to previous treatment for SLE with topical and systemic corticosteroids, antimalarials and immunosuppressants. [28, 29] Of note, thalidomide has been shown to stimulate hair regrowth in patients with SLE-induced alopecia. [30] Also, thalidomide had some effect on improving the articular manifestations of SLE. [8, 30] In a case series by Stevens et al., it was noted that thalidomide does not significantly suppress systemic disease in SLE. Thus, one should not discontinue other lupus treatments. [31]
Thalidomide has been used in the treatment of lichen planus, but only case reports exist. [3] TNF-α has been implicated in mediating the effects of lichen planus, so thalidomide's ability to suppress this cytokine has led to its use in this disease. [3] In most case reports, a dose of 150-300 mg was used and complete healing was usually noted within 4 months of beginning treatment. [32, 33]
Prurigo nodularis (PN) is a very pruritic form of neurodermatitis manifested by erythematous or hyperpigmented nodules. Treatment for PN, such as intralesional corticosteroids, benoxaprofen, and PUVA has not been very efficacious as the result of frequent relapse. [34] There has been one clinical study and many case reports on the use of thalidomide for PN. [35, 36, 37] These published studies indicate that thalidomide might be effective in the treatment of PN, but efficacy needs to be evaluated in clinical trials. The mechanism of action of thalidomide on PN is believed to be its direct toxic effect on the proliferated neural tissues. [2,5] Thalidomide has no effect on prurigo simplex because it does not possess the same nerve hyperplasia seen in prurigo nodularis. [2]
Sarcoidosis has been treated with thalidomide, but there are only case reports detailing its use. Thalidomide has been proposed to decrease the granulomatous inflammation in sarcoidosis by inhibiting macrophage function and decreasing TNF-α and IFN-γ. [3]
Thalidomide has shown beneficial effects in both acute and chronic forms of graft-versus-host disease (GVHD) in animal models. It may act at an early stage in the antigen recognition pathway in down-regulating the lymphocyte response in GVHD. The case reports and clinical trials have not been conclusive, as some demonstrate efficacy while others do not. [5,38] A recent study showed that thalidomide can be beneficial in the treatment of chronic GVHD in patients refractory to prednisone and cyclosporin. [39] Conversely, Chao et al. performed a randomized, double-blind, placebo-controlled study with 59 patients to evaluate the efficacy of thalidomide as a prophylactic agent in the prevention of chronic GVHD. The treatment group received 200 mg of thalidomide twice a day beginning 80 days following allogeneic bone-marrow transplantation. The treatment group actually developed chronic GVHD more often than the placebo group. [40] Additionally, severe cutaneous ulceration, skin rashes, and neutropenia have been reported when thalidomide has been used to treat chronic GVHD. [41, 42]
Thalidomide can have deleterious effects when used to treat acute severe drug reactions. For instance, thalidomide resulted in an increased mortality when it was used to treat toxic epidermal necrolysis (TEN) in a randomized, double-blind, placebo-controlled study by Wolkenstein et al. In their study, the thalidomide treatment group had an 83 percent mortality (10 of 12 patients) after receiving 400 mg a day of thalidomide for 5 days; the placebo group experienced a 30 percent mortality (3 of 10 patients). Thus, thalidomide appears to have harmful effects when used to treat TEN. [43, 44]
HIV has been treated with thalidomide because it does have some proven anti-retroviral effects assiciated with its inhibition of TNF-α production. [45] TNF-α stimulates a cellular transcription factor that induces the expression of HIV from chronically infected cell lines. [45] Blocking of TNF-α- stimulated HIV replication by thalidomide has been demonstrated both in vitro and ex vivo. [45] Thalidomide has also shown some potential in AIDS-related conditions such as cachexia and aphthous ulcers. [45, 46, 47, 48, 49] Several clinical trials have revealed a beneficial effect on Kaposi sarcoma when treated with thalidomide. [50] However, thalidomide should be used cautiously in HIV patients until further studies are done to determine the effect of an apparent increased viral load caused by this drug. [5]
Thalidomide has shown promise in the treatment of Jessner lymphocytic infiltration of the skin. In 1995, an open-label study found that those treated with thalidomide had a significantly greater complete response rate than the placebo group. [51]
Langerhans-cell histiocytosis is a disease in which Langerhans cells proliferate and can infect many organ systems. There have been case reports of patients demonstrating complete response when treated with thalidomide. [52]
There has been limited experience with thalidomide in pemphigoid disorders; just a small series and a case report have been published. Duong et al. reported the use of thalidomide in a patient with cicatricial pemphigoid, an autoimmune blistering disorder affecting the mucous membranes and occasionally the skin. Cicatricial pemphigoid is usually treated with dapsone or immunosuppressants. Because the patient's oral and cutaneous lesions had not responded to these treatments, he was treated with 100 mg of thalidomide a day and improvement was noted after just 5 months. The disease remained stable after a taper of the thalidomide. [53]
Thalidomide has also been used outside of dermatology in the treatment of various cancers, utilizing its immunomodulatory and anti-angiogenic properties to alter the tumor micro-environment. It has been used in various hematologic malignancies and solid tumors, such as renal-cell carcinoma. There are ongoing trials for multiple myeloma. [8]
The rediscovery of thalidomide has given physicians an effective FDA-approved treatment for erythema nodosum leprosum. Although teratogenicity and neurotoxicity must always be kept in mind, thalidomide appears to be a useful drug in a variety of inflammatory diseases, and even cancers. Further studies are needed to delineate fully the benefits of this useful drug.
References
1. Tseng S, Pak G, Washenik K, et al. Rediscovering thalidomide: A review of its mechanism of action, side effects, and potential uses. J Am Acad Dermatol. 1996;35(6):969-79. PubMed2. Grosshans E, Illy G. Thalidomide therapy for inflammatory dermatoses. Int J Dermatol. 1984;23(9):598-602. PubMed
3. Stirling DI. Thalidomide and its impact in dermatology. Semin in Cutan Med Surg. 1988;17(4):231-42. PubMed
4. Ordi-Ros J, Cortes F, Cucurull E, et al. Thalidomide in the treatment of cutaneous lupus refractory to conventioal therapy. J Rheumatol 2000;27(6):1429-33.
5. Radomsky CL, Levine N. Thalidomide. Dermatol Clinics. 2001;19(1):87-103.
6. Teo SK, Harden JL, Burke AB, et al. Thalidomide is distributed into human semen after oral dosing. Drug Metab Dispos. 2001;29(10):1355-57.
7. Nasca MR, O'Toole EA, Palicharla P, et al. Thalidomide increases human keratinocyte migration and proliferation. J Invest Dermatol. 1999;113(5):720-4.
8. Calabrese L, Fleischer AB. Thalidomide: current and potential clinical applications. Am J Med. 2000;108(6):487-95.
9. Federman GL, Federman DG. Recalcitrant pyoderma gangrenosum treated with thalidomide. Mayo Clin Proc. 2000;75(8):842-4.
10. Stephens TD. Proposed mechanisms of action in thalidomide embryopathy. Teratology. 1988;38:229-39.
11. McBride WG. Thalidomide embryopathy. Teratology. 1977;16(1):79-82.
12. Ochonisky S, Verroust J, Bastuji-Garin S, et al. Thalidomide neuropathy incidence and clinico-electrophysiologic findings in 42 patients. Arch Dermatol. 1994;130(1):66-9.
13. Hess CW, Hunziker T, Kupfer A, et al. Thalidomide-induced peripheral neuropathy. A perspective clinical, neurophysiological and pharmacogenetic evaluation. J Neurol. 1986;233(2):83-9.
14. Powell RJ, Gardner-Medwin JMM. 1994. Guideline for the clinical use and dispensing of thalidomide. Postgrad Med J. 1994;70:901-04.
15. Iyer, CG, Languillon J, Ramanujam K, et al.WHO co-ordinated short-term double-blind trial with thalidomide in the treatment of acute lepra reactions in male lepromatous patients. Bull World Health Organ 1971;45(6):719-32.
16. Latapi F, Saul A, Gomez-Vidal M. Talidomia en el tratamiento de la reaccion leprosa. Gaceta Medica De Mexico 99(10):917-31, 1969.
17. Grinspan D. Significant response of oral aphthosis to thalidomide treatment. J Am Acad Dermatol. 1985;12(1):85-90.
18. Hutton KP, Rodgers RS. Recurrent aphthous stomatitis. Dermatol. 1987;5:761-68.
19. Revuz J, Guillaume JC, Janier M, et al. Crossover study of thalidomide vs. placebo in severe recurrent aphthous stomatitis. Arch Dermatol. 1990;126:923-7.
20. Rustin MH, Gilkes JJ, Robinson TW. Pyoderma gangrenosum associated with Behcet's disease: treatment with thalidomide. J Am Acad Dermatol. 1990;23(5):941-4.
21. Munro CS, Cox NH. 1988. Pyoderma gangrenosum associated with Behcet's syndrome-response to thalidomide. Clin Exp Dermatol. 1988;13(6):408-10.
22. Hamuryudan V, Mat C, Saip S, et al. Thalidomide in the treatment of the mucocutaneous lesions of the Behcet syndrome. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 1998;128(6):443-50.
23. Nadeau SE. Neurologic manifestations of systemic disease. Neurol Clin. 2002;20(1):151-78.
24. Hamza M, Hamzaoui K, Ayed K, et al. Thalidomide and cell-mediated immunity in Behcet's disease. Clin Rheumatol. 1987;6(4):608-9.
25. Farrell AM, Black MM, Bracka A, Bunker CB. Pyoderma gangrenosum of the penis. Br J Dermatol. 1998;138(2):337-40.
26. Warren KJ, Nopper AJ, Crosby DL. Thalidomide for recalcitrant discoid lesions in a patient with systemic lupus erythematous. J Am Acad Dermatol. 1998;39(2):293-5.
27. Kyriakis KP, Kontochristopoulos GJ, Panteleos DN. Experience with low-dose thalidomide therapy in chronic discoid lupus erythematosus. Int J Dermatol. 2000;39(13):218-22.
28. Atra E, Sato EI. Treatment of the cutaneous lesions of systemic lupus erythematous with thalidomide. Clin Exp Rheumatol. 1993;11(5):487-93.
29. Scolari F, Harms M, Gilardi S. Thalidomide in the treatment of chronic lupus erythematous. Dermatologica. 1982;165(4):355-62.
30. Walchner M, Meurer M, Plewig G, et al. Clinical and immunologic parameters during thalidomide treatment of lupus erythematous. Int J Dermatol. 2000;39(5):383-8.
31. Stevens RJ, Andujar C, Edwards CJ, et. al. Thalidomide in the treatment of the cutaneous manifestations of lupus erythematous: experience in sixteen consecutive patients. Br J Rheumatol. 1997;36(3):353-9.
32. Dereure O, Basset-Seguin N, Guilhou JJ. Erosive lichen planus: dramatic response to halidomide. Arch Dermatol. 1996;118(8):536.
33. Perez Alfonzo R, Weiss E, Piquero Martin J, Rondon Lugo A. Generalized lichen planus with erosive lesions of the penis, treated with Thalidomide. Report of a case and review of the literature. Med Cutan Ibero Lat Am. 1987;15(4):321-6.
34. Young, Baik G, Hee JY, Sook JS. Thalidomide therapy on a case of prurigo nodularis. Ann Dermatol. 1993;5(2):117-20.
35. Herranz P, Pizarro A, De Lucas R, et al. 1998. Treatment of AIDs-associated prurigo nodularis with Thalidomide. Clin Exp Dermatol. 1998;23(5):233-5.
36. Winkelmann RK, Connolly SM, Doyle JA, Padhilha-Goncalves A. Thalidomide treatment of prurigo nodularis. Acta Derm Venereol. 1994;64(5):412-7.
37. Van den Broek H. Treatment of prurigo nodularis with thalidomide. Arch Dermatol. 1980;116(5):571-2.
38. Rovelli A, Arrigo C, Nesi F, et al. The role of thalidomide in the treatment of refractory chronic graft-versus-host disease following bone marrow transplantation in children. Bone Marrow Transplant. 1998;21(6):577-81.
39. van de Poel MH, Pasman PC, Schouten HC. The use of thalidomide in chronic refractory graft versus host disease. Neth J Med.2001;59(2):45-9.
40. Chao NJ, Parker PM, Niland JC, et al. Paradoxical effect of thalidomide prophylaxis on chronic graft-vs-host disease. Biol Blood Marrow Transplant. 1996;2(2):86-92.
41. Schlossberg H, Klumpp T, Sabol P, et al. Severe cutaneous ulceration following treatment with thalidomide for GVHD. Bone Marrow Transplant. 2001;27(2):229-30.
42. Parker PM, Chao NJ, Nademanee A, et al. Thalidomide as salvage therapy for chronic graft-versus-host disease. Blood. 1995;86(9):3604-9.
43. Wolkenstein P, Latarjet J, Roujeau JC, et al. Randomized comparison of thalidomide versus placebo in toxic epidermal necrolysis. Lancet. 1998;352:1586-89.
44. Ghislain PD, Roujeau JC. Treatment of severe drug reactions: Stevens-Johnson syndrome, toxic epidermal necrolysis and hypersensitivity syndrome. Dermatol Online J. 2002;8(1):5.
45. Ravot E, Lisziewicz J, Lori F. New uses for old drugs in HIV infection: the role of hydrozyurea, cyclosporin and thalidomide. Drugs. 1999;58(6):953-63.
46. Gagnon B, Bruera E. A review of the drug treatment of cachexia associated with cancer. Drugs. 1998;55(5):675-88.
47. Kaplan G, Thomas S, Fierer DS, et al. Thalidomide for the treatment of AIDS-associated wasting. AIDS Res Hum Retroviruses. 2000;16(14):1345-55.
48. Wanke C. Single-agent/combination therapy of human immunodeficiency virus-related wasting. Semin Oncol. 1998;25(2 suppl 6):98-103.
49. Peuckmann V, Fisch M, Bruera E. Potential novel uses of thalidomide: focus on palliative care. Drugs. 2000;60(2):273-92.
50. Krown SE. Management of Kaposi sarcoma: the role of interferon and thalidomide. Curr Opin Oncol. 2001;13(5):374-81.
51. Guillaume JC, Moulin G, Dieng MT. Crossover study of thalidomide vs placebo in Jessner's lymphocytic infiltration of the skin. Arch Dermatol. 1995;131(9):1032-5.
52. Mortazavi H, Ehsani A, Namazi MR. Langerhans' cell histiocytosis. Dermatol Online J. 2002;8(2):18.
53. Duong DJ, Moxley RT, Kellman RM, et al. Thalidomide therapy for cicatricial pemphigoid. J Am Acad Dermatol. 2002;47(2):S193-5.
© 2003 Dermatology Online Journal

