Cutaneous fusariosis developing in a post-irradiation site
Published Web Location
https://doi.org/10.5070/D35c85b73dMain Content
Cutaneous fusariosis developing in a post-irradiation site
WeiWei Dai DO1, Jennifer Warner Dharamsi MD2, Sameer Soliman BS2, Carlos Ricotti MD2, Rita Gander PhD3, Paul Bergstresser MD2, Joanna Chan MD2
Dermatology Online Journal 17 (5): 5
1. Department of Osteopathic Medicine, University Hospitals Richmond Medical Center, Case Western Reserve University, Cleveland,
Ohio2. Department of Dermatology, University of Texas Southwestern Medical Center, Dallas, Texas
3. Department of Pathology, University of Texas Southwestern Medical Center, Dallas, Texas. chan@post.harvard.edu
Abstract
Cutaneous fusariosis is an opportunistic mycosis in immunocompromised patients. We present a novel variation of an immunocompromised patient who developed fusariosis in a previously irradiated site. Irradiation led to atrophy, contraction, fibrosis, barrier disruption, and an altered dermal environment in which the infection developed. Significantly, this is the first case report of fusariosis in a previously irradiated site of an immunocompromised patient. Treatment included debridement and voriconazole.
Introduction
Fusarium species are emerging as the second most common opportunistic mycosis in immunocompromised patients because of the prophylactic use of antifungals for candidiasis following chemotherapy [1]. Skin and nails are common sites of infection, serving as portals for systemic disease [2]. The macerated skin surrounding ulcers is an ideal environment for colonization and invasion by Fusarium species and cutaneous fusariosis has been described previously in arterial and hypertensive leg ulcers [3, 4]. We present a case of cutaneous fusariosis that occurred within an ulcer at the site of prior ionizing radiation.
Case report
Patient Background
A 35-year-old woman with a history of congenital renal disease, status-post cadaveric renal allograft in 1994 presented to Parkland Memorial Hospital in December 2008 with a large non-healing painful ulceration on her left calf. She was receiving chronic immunosuppressive therapy with cyclosporine 100 mg twice daily and prednisone 5 to 10 mg daily. In 2005, the patient had been diagnosed with a poorly-differentiated primary cutaneous mixed B- and T-cell lymphoma on the left calf, for which she had undergone 34 treatments with local radiotherapy. Following irradiation, which had eradicated the tumor, the treated site healed fully, but developed an erythematous atrophic sclerotic plaque consistent with radiation dermatitis. This site remained asymptomatic and intact and the patient remained in good health for the next three years. In May 2008, she developed a self-described “insect bite” within the central aspect of the irradiated site, which enlarged gradually over several months to become a painful ulcer. She denied any preceding trauma to the irradiated site, nor any associated constitutional symptoms of fever, chills, or night sweats.
Objective Findings
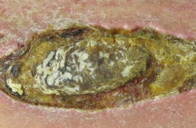 |
| Figure 1 |
|---|
| Figure 1. Ulcer on the left calf |
1. Description of pathology
Physical examination revealed a 10.5 cm x 3.8 cm x 0.4 cm well-circumscribed oval-shaped, verrucous ulceration with a rolled border on the left calf (Figure 1). The ulcer base was yellow and crusted with serous fluid drainage; no frank purulence or bullae were observed. The superior medial aspect of the ulcer demonstrated angulation with a satellite lesion. Surrounding the ulcer was a pink to violaceous, scaly indurated plaque. No lymphadenopathy was detected and the lower extremity was otherwise cool and non-tender to palpation.
2. Laboratory results
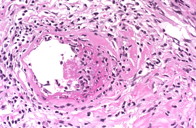
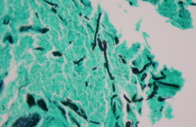
Initial laboratory examination revealed normal baseline laboratory levels aside from anemia (hemoglobin of 11.7 g/dL) and evidence of chronic kidney disease (creatinine of 1.8 gm/dL). Punch biopsy of the ulcer edge demonstrated extensive dermal and subcutaneous sclerosis with focal areas of necrosis and thrombosis of mid and deep dermal vessels. Superficially, a focal collection of neutrophils was present in the ulcer, with fungal hyphal elements visible in the crust itself (Figure 2). Periodic acid-Schiff staining revealed septate fungal hyphae penetrating the endothelium of dermal blood vessels and extending into the subcutaneous fat, reflecting the pathogenic nature of the organism (Figure 3). Fite stain was negative for acid fast bacilli. Colonies of Fusarium species were cultured from the tissue biopsy; however, multiple fungal blood cultures were negative. Osteomyelitis was excluded by magnetic resonance imaging and nuclear scintigraphy.
3. Treatment
A diagnosis of cutaneous fusariosis was made, and the patient underwent surgical debridement of the wound. Per recommendations of the infectious disease consultant, intravenous voriconazole 6mg/kg twice daily was started and was continued for 3 weeks. Over the course of her hospitalization, the patient did well and never developed signs or symptoms of systemic fusariosis. Upon discharge, she was switched to oral voriconazole 200mg twice daily. For nearly two years after this admission, her ulcer persisted, warranting several re-admissions for bacterial superinfection and requiring a total of 8 surgical debridements. Under guidance of the infectious disease team, she remained on twice daily oral voriconazole for one year. However, during this time, the patient remained lost to follow-up with dermatology and repeat biopsy and wound culture were not performed, Therefore, it is not known whether the non-healing nature of her ulcer was related to persistent fusarial infection. At one point, amputation was considered, but this was avoided after successful muscle flap grafting over the ulcer site by the plastic surgery team in April 2010. Since this procedure, her ulcer has now healed almost completely and the patient has improved ambulation and limb functionality. The patient continues to be followed regularly by the plastic surgery and wound care teams.
Discussion
Optimal treatment protocols for cutaneous fusariosis are not yet established because of a dearth of clinical trials. Current guidelines reflect data from retrospective studies, case reports, and in vitro studies. Systematic antifungal susceptibility testing and speciation can assist in therapeutic management. Formerly published guidelines recommend high dose amphotericin B to treat F. solani/F. verticolloides and either high dose amphotericin B or voriconazole for management of other Fusarium species [5]. Amphotericin therapy is a viable option for fusariosis, with no statistically significant differences in the response rates for the lipid formulation (46%) compared to deoxycholate amphotericin B (32%) [6]. A recent retrospective pharmacoeconomics analysis of 18 peer-reviewed publications assessing various antifungal agents recommends voriconazole as the most cost-effective therapy for treatment of fungal infections, including fusariosis [7]. In another retrospective review of 73 patients with invasive fusariosis, 47 percent were treated successfully with voriconazole as monotherapy, with no significant clinical advantage among 13 patients who received combination therapy [8]. For patients refractory to amphotericin or intolerant of its adverse effects, posaconazole can be also be used as salvage therapy, with an overall success rate of 48 percent [9].
Fusarium species are filamentous fungi that are ubiquitous in the environment and can lead to several clinical manifestations. Localized superficial cutaneous infections can be seen in both immunocompetent and immunocompromised patients, whereas invasive and systemic infections are almost exclusively seen among immunosuppressed patients, especially those with neutropenia [2]. To our knowledge, fusarial infection occurring in the setting of radiation dermatitis has not been reported. Our patient had a localized fusarial infection, which occurred in the setting of chronic post-transplant immunosuppression, but normal neutrophil counts; her lack of neutropenia likely played a role in preventing dissemination of her infection. In addition to being on chronic immunosuppressive medications, we believe that the patient’s history of radiation played a key role in predisposing her to developing fusarial infection. In irradiated skin, upregulation of pro-inflammatory cytokines and growth factors leads to epidermal and dermal atrophy, endothelial damage, contraction, fibrosis, and barrier disruption [10]. These changes often manifest clinically as radiation dermatitis, which our patient exhibited prior to the onset of her ulcer. Epidermal atrophy and barrier disruption are major risk factors for the development of localized cutaneous fusariosis because the skin is more vulnerable to minor trauma, which can create a portal of entry for infection. Furthermore, dermal fibrosis and local vascular compromise may lead to poor wound healing and clearance of infection, which likely contributed to the chronic non-healing nature of her wound. This patient’s course illustrates a novel variation in cutaneous mycoses related to the potential contribution of radiotherapy in predisposing an immunocompromised patient to local infection.
References
1. Boutati EI, Anaissie EJ. Fusarium, a significant emerging pathogen in patients with hematologic malignancy: ten years' experience at a cancer center and implications for management. Blood. 1997 Aug 1;90(3):999-1008. [PubMed]2. Nucci M, Anaissie E. Fusarium infections in immunocompromised patients. Clin Microbiol Rev. 2007 Oct;20(4):695-704. [PubMed]
3. Willemsen MJ, De Coninck AL, Coremans-Pelseneer JE, Marichal-Pipeleers MA, Roseeuw DI. Parasitic invasion of Fusarium oxysporum in an arterial ulcer in an otherwise healthy patient. Mykosen. 1986 Jun;29(6):248-52. [PubMed]
4. van Dijk E, van den Berg WH, Landwehr AJ. Fusarium solani infection of a hypertensive leg ulcer in a diabetic. Mykosen. 1980 Nov;23(11):603-6. [PubMed]
5. Jensen HE, Schonheyder HC, Hotchi M, Kaufman L. Diagnosis of systemic mycoses by specific immunohistochemical tests. APMIS. 1996 Apr;104(4):241-58. [PubMed]
6. Nucci M, Anaissie EJ, Queiroz-Telles F, Martins CA, Trabasso P, Solza C, Mangini C, Simoes BP, Colombo AL, Vaz J, Levy CE, Costa S, Moreira VA, Oliveira JS, Paraguay N, Duboc G, Voltarelli JC, Maiolino A, Pasquini R, Souza CA. Outcome predictors of 84 patients with hematologic malignancies and Fusarium infection. Cancer. 2003 Jul 15;98(2):315-9. [PubMed]
7. Al-Badriyeh D, Heng SC, Neoh CF, Slavin M, Stewart K, Kong DC. Pharmacoeconomics of voriconazole in the management of invasive fungal infections. Expert Rev Pharmacoecon Outcomes Res. 2010 Dec;10(6):623-36. [PubMed]
8. Lortholary O, Obenga G, Biswas P, Caillot D, Chachaty E, Bienvenu AL, Cornet M, Greene J, Herbrecht R, Lacroix C, Grenouillet F, Raad I, Sitbon K, Troke P. International retrospective analysis of 73 cases of invasive fusariosis treated with voriconazole. Antimicrob Agents Chemother. 2010 Oct;54(10):4446-50. [PubMed]
9. Raad II, Hachem RY, Herbrecht R, Graybill JR, Hare R, Corcoran G, Kontoyiannis DP. Posaconazole as salvage treatment for invasive fusariosis in patients with underlying hematologic malignancy and other conditions. Clin Infect Dis. 2006 May 15;42(10):1398-403. [PubMed]
10. Hymes SR, Strom EA, Fife C. Radiation dermatitis: clinical presentation, pathophysiology, and treatment 2006. J Am Acad Dermatol. 2006 Jan;54(1):28-46. [PubMed]
© 2011 Dermatology Online Journal