Actinomycosis presenting as a large facial mass
Published Web Location
https://doi.org/10.5070/D35493b2d7Main Content
Actinomycosis presenting as a large facial mass
Holly L Bartell MD, Michael L Sonabend MD, Sylvia Hsu MD
Dermatology Online Journal 12 (2): 20
Department of Dermatology, Baylor College of Medicine, Houston, Texas. shsu@bcm.eduClinical synopsis
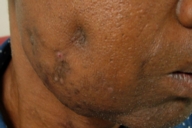 | 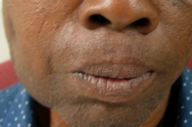 |
| Figure 1 | Figure 2 |
|---|
A 58-year-old previously healthy man presented to the emergency center with a 12-month history of progressive right-sided facial swelling. The patient reported only mild pain exacerbated by eating and difficulty opening his mouth. He denied fever, chills, or weight loss. He had a 40-pack per year history of smoking tobacco and consumed 40 ounces of beer per day. The patient had no significant past medical history, and denied diabetes, hypertension, or cancer. He had lived his entire life in Texas, and worked as a farmer. Physical examination revealed an 8 × 8 cm area of facial swelling and indurated nodularity in the right parotidomasseteric region. There was no skin breakdown or drainage at his initial visit. No fluctuance was noted; however, there was firm induration surrounding the mass as well as slight erythema of the overlying skin. A small right tonsillar mass was observed along with poor dentition. The rest of the head and neck exam was normal. The patient was admitted for further work up of the facial mass. Laboratory workup revealed an erythrocyte sedimentation rate of 37 mm/hr. Other laboratory values, a urinalysis, and chest x-ray were all within normal limits. An excisional biopsy by an otolaryngologist revealed only dense fibrosis of the dermal and subcutaneous tissues. A computerized axial tomography scan revealed right sided soft-tissue swelling, small focal micro-abscesses, dental caries, and a right tonsillar mass. No neoplastic process was identified on laryngoscopic guided biopsies. The patient was discharged with outpatient followup by both ENT and the dermatology service. At his dermatology visit, which was approximately 1 month after his initial presentation, the patient was noted to have a few small sinus tracts without obvious drainage and a new indurated red papule. Two 6 mm punch biopsies of the sinus tract as well as from the new papule were obtained, and each was split for routine histology and tissue culture. Routine histology revealed chronic granulomatous inflammation with a mixed inflammatory infiltrate. Clusters of thin filamentous bacteria within a large granule were noted in the deep dermis with a surrounding neutrophilic abscess. The bacteria were morphologically compatible with an Actinomyces spp. Culture confirmed the diagnosis by growing Actinobacillus actinomycetemcomitans. The patient was started on oral amoxicillin and doxycycline with close outpatient followup by the otolaryngology, dermatology, and infectious disease services.
Discussion
Actinomycosis is a chronic bacterial infection attributed to Actinomyces spp., most commonly Actinomyces israelli [1]. This Gram-positive, anaerobic organism is a normal inhabitant of the human oropharynx, gastrointestinal tract, and female genital tract [2]. Actinomycetes are usually non-virulent in nature, but a disruption of the protective mucosal barrier, and alteration of the resident microbial flora play a crucial role in infection [3]. Infections most commonly occur in the cervicofacial, abdominopelvic, and thoracic regions. CNS involvement has been documented and can occur secondary to hematogenous spread or by direct extension [4].
Cervicofacial actinomycosis, also known as lumpy jaw, is the most common variant encountered and accounts for 55 percent of cases [5]. Poor dental hygiene, dental disease, and dental procedures are common predisposing factors. Cervicofacial actinomycosis presents with palpable nodularity, soft tissue swelling, and fibrosis involving the submandibular or perimandibular region. Abscess formation is common and may extend to involve the overlying tissue forming draining sinus tracts, as occurred in our patient. Histologic examination shows a diffuse mixed infiltrate with granulation tissue, microabscesses, and sinus tracts [6]. The most helpful diagnostic feature is the presence of sulfur granules. Sulfur granules can occasionally be seen clinically as 1-mm yellow grains that resemble sand. Histologically, they contain numerous intertwined filamentous Gram-positive bacteria [7]. Isolating the Actinomyces spp. in culture of a clinical specimen is the most accurate method of diagnosis, but Gram stain of a crushed sulfur granule or histology with the characteristic features of a granule could suffice.
Historically, treatment of actinomycosis was high dose penicillin administered over a prolonged period. Typically, intravenous penicillin was given for a 2-6 week course, followed by a 6-12 month course of oral penicillin or amoxicillin [8]. Recently, though, many reports document short-term antibiotic therapy to be adequate, especially in cervicofacial actinomycosis [9, 10, 11, 12, 13, 14, 15]. Sudhaker et al. reported successful cases using short-term therapy. One patient achieved complete resolution of infection after surgical incision and drainage followed by 4-days intravenous ampicillin-sulbactam, then 5 days of oral amoxicillin-clavulanic acid. They also reported success in another patient after only 2 weeks of intravenous penicillin G followed by 6 weeks of oral penicillin therapy [16]. In the case of a patient with a penicillin allergy, suitable alternatives include one of the tetracyclines, erythromycin, clindamycin, or a cephalosporin [17]. Because of the potential for relapse of infection, prolonged observation of patients after treatment is necessary to detect the possibility of recurrence.
As this case demonstrated, infection with actinomycosis can mimic a neoplastic process by presenting with a large mass. Without a high index of suspicion, the diagnosis can easily be overlooked. When considering a nodular mass, along with neoplastic processes, the possibility of infections with actinomycetes, fungi, and mycobacteria should be entertained. The clues to diagnosis in this case were the poor dentition, small draining sinuses, and an indurated papule at the edge of the swelling. Biopsies should be carefully directed to areas likely to have active infection and not the remaining fibrosis. It is important to make the correct diagnosis, since a short course of antibiotics can be curative and spare the patient extensive testing and surgery.
References
1. Schaal KP, Lee HJ. Actinomycete infections in humans-a review. Gene 1992; 115:201-11.2. Peabody J, Seabury J. Actinomycosis and nocardiosis. J Chronic Dis 1957;5:374- 403.
3. Pulverer G, Schutt-Gerowitt H, Schaal KP. Human cervicofacial actinomycoses: microbiological Data for 1997 cases. Clin Infect Dis 2003:37:490-7.
4. Smego RA Jr. Actinomycosis of the central nervous system. Rev Infect Dis 1987 Sep-Oct;9(5):855-65.
5. Russo TA. Agents of actinomycosis. In:Mandell GL, Bennett JE, Dolin R, eds. Principals and practice of infectious diseases. 5th ed. Philadelphia: Churchill Livingston, 2000:2645-54.
6. Rapini, RP. Bacterial Diseases In: Practical Dermatopathology. New York: Mosby. 2005; 170-171.
7. Hotchi M, Schwartz J. Characterization of actinomycotic granules by architecture and staining methods. Arch Pathol 1972; 93:392-400.
8. Peabody JW Jr, Seabury JH. Actinomycosis and nocardiosis:a review of basic differences in therapy. Am J Med 1960;28:99-115.
9. Dobson L, Cutting WC. Penicillin and sulfonamides in the therapy of actinomycosis, JAMA 1945; 128:856-63.
10. Walker JM, Hamilton JW. The treatment of actinomycosis with penicillin. Ann Surg 1945; 121:373-84.
11. Nichols DR, Herrell WE. Penicillin in the treatment of actinomycosis. J Lab Clin Med 1948; 33:521-5.
12. Bates M, Cruickshank G. Thoracic actinomycosis. Thorax 1957; 12: 99-124.
13. Spilsbury BW, Johnstone FR. The clinical course of actinomycotic infections. Can J Surg 1962; 5:33-48.
14. Martin MV. The use of oral amoxycillin for the treatment of actinomycosis. Br Dent J 1984; 156:252-4.
15. Nielsen PM, Novak A. Acute cervico-facial actinomycosis. Int J Oral Maxillofac Surg 1987; 16:440-4.
16. Sudhakar SS, Ross JJ. Short-term treatment of Actinomycosis: Two Cases and a Review. Clin Infect Dis 2004; 38:444-7.
17. Tanaka-Bandoh K, Watanabe K, Kato N, Ueno K. Susceptibilities of Actinomyces species and Propionibacterium propionicus to antimicrobioal agents. Clin Infect Dis 1997; 25(suppl 2):S262-3.
© 2006 Dermatology Online Journal

