Chromoblastomycosis associated with in a carpenter handling exotic woods
Published Web Location
https://doi.org/10.5070/D34tb1r39jMain Content
Chromoblastomycosis associated with Fonsecaea pedrosoi in a carpenter handling exotic woods
Nuno Menezes1, Paulo Varela1, Antónia Furtado2, Antonio Couceiro2, Ismália Calheiros3, Laura Rosado4, Gioconda Mota1, Armando Baptista1
Dermatology Online Journal 14 (2): 9
1. Serviço de Dermatologia do Centro Hospitalar V.N. de Gaia (CHVNG), Portugal2. Serviço de Anatomia Patológica do CHVNG, Portugal
3. Serviço de Patologia Clínica do CHVNG, Portugal
4. Instituto de Saúde Dr. Ricardo Jorge, Portugal
Abstract
Chromoblastomycosis is a rare, hard to diagnose disease that arises mostly in the tropics, especially in humid areas, affecting mainly males and rural workers. It is characterized by verrucous plaques or nodules that are slow growing and attributed to infection by different pigmented (dematiaceous) fungi. Usually the infection develops after injury, being primarily located on the lower extremities. The authors present the case of a healthy, 60-year-old man observed with a one year history of an erythematous violaceous 5cm tumor located on the right thigh. A biopsy specimen for histopathology revealed single or clustered brown pigmented cells, with a single or double septum and thick cell walls. Cultural smears showed growth of Fonsecaea Pedrosoi. The patient was treated with oral itraconazole (200mg per day), with a good response and clinical cure in 6 months that left only an atrophic scar.
The term chromoblastomycosis was first used by Terra et al. in cases of polymorphic fungal disease of the lower limbs presenting with nodules or verrucous plaques with hyperkeratosis and acanthosis of the affected epithelial tissues, which could develop into complications such as ulceration, lymphedema and squamous cell carcinoma [1, 2]. Chromoblastomycosis is a subcutaneous mycosis caused by a variety of black or dematiaceous fungi [3] that assume the shape of sclerotic, muriform structures known as Medlar bodies [4].
The ethiologic agents belong to the Fonsecaea, Phialophora, Rhinocladiella and Cladosporium genera [4, 5]. The causal agents are normally found as saprophytes in the soil, wood and vegetation [3]. They are more frequent in the tropics but have a worldwide distribution [7]. The vast majority of cases are attributed to Fonsecaea pedroso [4, 6].
In Portugal there have been cases both of native [7] and imported [8] origin. Nowadays the number of cases in north Europe is growing because of the increasing number of steam bath facilities, which are normally made of tropical wood [9].
The inoculation usually occurs after trauma, usually in exposed areas, particularly in the lower limbs, therefore being more frequent in rural areas [1, 4, 10, 11]. The disease is more common among men 30-50 years old [4, 6].
If not diagnosed early the infection has a chronic evolution, being difficult to treat and with frequent recurrences. Sometimes complications such as bacterial super-infection can lead to lymphedema and elephantiasis and occasionally to squamous cell carcinoma at affected sites. Lymphatic and hematogenic dissemination and brain metastases have been observed rarely [1, 5, 12].
Clinical synopsis
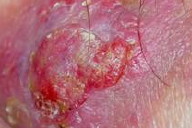
A previously healthy 60-year-old male carpenter who had never traveled outside Portugal was evaluated for a 5cm erythemato-violaceous tumoral lesion involving the external surface of the right thigh, with a one year evolution (Fig. 1).
 |  |
| Figure 1 | Figure 2 |
|---|
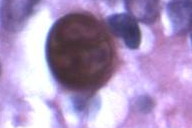
A skin biopsy was obtained, which showed intense pseudoepitheliomatous hyperplasia and a mixed dermal granulomatous response, with small neutrophil abscesses and organized granulomae of the giant-cell type, inside which groups of fungal cells were seen. The fungal cells were characteristically divided in several planes by thick septa (muriform or sclerotic cells) (Fig. 2).
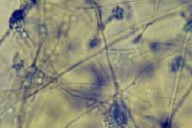
A skin biopsy fragment was then cultured in a Sabouraud medium with chloramphenicol and incubated at 25°C, showing growth of black colonies, whose microscopic exam showed numerous elliptical to oval spores with thick septa, long and oval conidia confined to the upper part of the cell, and multiple phialides with terminal collarette (Fig. 3). The diagnosis of chromoblastomycosis attributed to Fonsecaea pedrosoi was also confirmed by rRNA sequence analysis.
 |  |
| Figure 3 | Figure 4 |
|---|
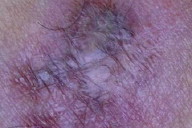
We started treatment with itraconazole PO 100mg bid reducing to 100mg per day after 3 months due to marked clinical improvement. After 5 months of treatment it was possible to see only a violaceous cicatricial area and treatment was suspended (Fig. 4). After a followup of 18 months the patient is still without any clinical recurrence.
Discussion
Our patient denied any travel to foreign countries or contact with people arriving from tropical countries. We think it is likely that the source of infection was the professional contact with tropical woods, probably from a minor trauma.
This represents one more case of this rare disease in developed countries (less than 10 cases reported in Portugal) [7, 10, 11] and to our knowledge the first to occur in a carpenter. Diagnosis is accomplished by Medlar bodies isolation either in pathologic or microbiological exams. They are defined as rounded, brown structures, with 5-10mm in diameter, thick wall and internal septation [5, 11, 13].
Clinically, the diagnosis is suggested by warty plaques, nodules, or tumors, located in the lower limbs of a patient living or arriving from a tropical country. Differential diagnosis must be done with other fungal infections (such as sporotrichosis or lobomycosis), cutaneous tuberculosis, Hansen disease, late syphilis, cutaneous leishmania, squamous cell carcinoma, amelanotic melanoma, and other dermatologic malignancies [4, 10].
The clinical suspicion should then be confirmed by pathologic, cultural and microscopic examinations [10]. Recent technology was developed that allows species confirmation with rRNA sequence analysis [6, 14].
Fonsecaea pedroso has the ability to produce melanin, which will be an immunogenic structure that together with the fungal antigens will induce a humoral and cellular response, which will be responsible by the epidermis pseudoepitheliomatous hyperplasia with transepidermal fungal elimination. This phenomenon is clinically seen by the observation of black dots at the surfaces of the lesion [7, 11, 13].
Treatment is difficult and not well established. Small lesions in hidden places can be treated by simple surgical excision with large margins followed by systemic treatment with an oral anti-fungal drug to avoid dissemination [4, 7]. More often, medical treatment is preferred with systemic anti-fungal agents. Fonsecaea pedrosoi, the most common pathogenic agent, is also more resistant to treatment, especially in protracted cases [15].
Usually, itraconazole (200-400mg/day PO for 6 months) is the first choice of therapy, with an 80-90 percent rate of success. This rate may increase if an association of 5-flucytosine is used [16, 17, 18]. Terbinafine, fluconazole, ketoconazole, and thiabendazole have been used [17, 18, 19, 20, 21, 22, 23]. Intravenous amphotericin B (up to 1mg/kg daily) is recommended in extensive cases [16]. Voriconazole efficacy is being tested, although its use is limited by serious side-effects [23].
Topical heat, cryotherapy, and CO2-laser ablation also have been described as treatment options [24, 25, 26, 27]. The more resistent situations can be treated with combination of agents like CO2 laser with topical heat [28]; combination of cryotherapy, shaving, oral 5-fluorocytosine, and oral amphotericin B [16] or itraconazole plus cryotherapy [29].
In our patient we had excellent results with slightly lower doses than those described in the literature (itraconazole 200mg/day for 3 months plus 100mg/day for more 2 months).
References
1. Terra F, Torres M, Fonseca Filho O, Arêa Leão AE. Novo tipo de dermatite verrucosa: micose por Acrotheca com associação de leishmaniose. Brasil Med 1922;36: 363-82. Minotto R, Bernardi CD, Mallmann LF, Edelweiss MI, Scroferneker ML. Chromoblastomycosis: A review of 100 cases in the state of Rio Grande do Sul, Brazil. J Am Acad Dermatol 2001;44: 585-92. PubMed
3. Vidal MS, de Castro LG, Cavalecate SC, Lacaz Cda S. Immunoprecipitation techniques and elisa in the detection of anti-Fonsecaea Pedrosoi antibodies in chromoblastomycosis. Rev. Inst. Med. Trop. S. Paulo 2003;45(6): 315-318. PubMed
4. Vieira MR, Martins ML. Micoses subcutâneas. Trab Soc Port Dermatol Venerol 2004;62 (2): 137-67.
5. Pang KR, Wu JJ, Huang DB, Tyring SK. Subcutaneous fungal infections. Dermatologic Therapy 2004;17(6): 523-31. PubMed
6. Tanabe H, Kawasaki M, Mochizuki T, Ishizaki H. Species identification and strain typing of Fonsecaea Pedrosoi using ribosomal RNA gene internal transcribed spacer regions. Nippon Ishinkin Gakkai Zasshi 2004;45(2):105-12. PubMed
7. Clerins I, Mariano A, Tellechea O, Reis JP, Velho R, Figueiredo A. romomicose por Exophiala Jeanselmei em doente transplantado renal. Trab Soc Port Dermatol Venerol 2000;58(2): 241-47.
8. Matos J, Figueiredo A, Eiriz E, Velho R, Baptista AP. Cromomicose (2 casos autóctones). Trab Soc Port Dermatol Venerol 1990;48: 43-47.
9. Verde SF, Nobre G, Oliveira A. Cromomicose, primeiro caso descrito em Portugal (oriundo de Angola). Trab Soc Port Dermatol Venerol 1976;34: 135-46.
10. Guerras Rodrigo F, Sequeira H. Micoses subcutâneas e sistémicas. Trab Soc Port Dermatol Venerol 2000;58 (4): 459-91.
11. McGinnis MR. Cromoblastomycoses and phaeohyphomycoses: New concepts, diagnosis and mycology. J Am Acad Dermatol 1983;8(1): 1-16. PubMed
12. Nobrega JP, Rosemberg S, Adami AM, Heins-Vaccari EM, Lacaz Cda S, de Brito T. Fonsecaea pedrosoi cerebral phaeohyphomycosis ("chromoblastomycosis"). First human culture-proven case reported in Brazil. Rev Inst Med Trop S. Paulo 2003;45(4): 217-220. PubMed
13. Brandt ME, Warnock DW. Epidemiology, clinical manifestations, and therapy of infections caused by dematiaceous fungi. J Chemother 2003;15 Suppl 2: 36-47. PubMed
14. Vidal MS, de Castro LG, Cavalecate SC, Lacaz Cda S. Immunoprecipitation techniques and ELISA in the detection of anti-Fonsecaea pedrosoi antibodies in chromoblastomycosis. Rev Inst Med Trop S. Paulo 2003;45(6): 315-18. PubMed
15. Poirriez J, Breuillard F, Francois N, Fruit J, Sendid B, Gross S, Dei-Cas E. A case of chromoblastomicosis treated by a combination of cryotherapy, shaving, oral 5-fluorocytosine and oral amphotericin B. Am J Trop Med Hyg 2000;63(1, 2): 61-63. PubMed
16. Hay R.J. Deep Fungal Infections. In: Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, editors. Dermatology in General Medicine, 6th ed. Mc Graw-Hill, New York, 2003, 2018-34.
17. Esterre P, Inzan CK, Ramarcel ER, Andriantsimahavandy A, Ratsioharana M, Pecarrere JL, Roig P. Treatment of chromomycosis with terbinafine preliminary results of an open pilot study. Br J Dermatol 1996, 134 (Suppl. 46): 33-36. PubMed
18. Sobera JO, Elewski BE; Fungal Diseases. In: Bolognia JL, Jorizzo JL, Rapini RP, editors. Dermatology, 1st ed, Londres. Mosby, 2003, 1171-98.
19. Vivas JRC, Torres-Rodríguez JM. Sensibilidad de hongos miceliares dematiáceos a diez antifúngicos empleando un método de difusión en agar. Rev Iberoam Micol 2001; 18: 113-17. PubMed
20. Mc Burney EI. Chromoblastomycosis treatment with ketoconazole. Cutis 1982;30(6): 746-8
21. Guerriero C, De Simone C, Tulli A. A case of chromoblastomycosis due to Phialophora verrucosa responding to treatment with fluconazole. Eur J Dermatol 1998;8(3): 167-8. PubMed
22. Olle-Goig JE, Domingo J. Case of chromomycosis treated with thiabendazole. Trans R Soc Trop Med Hyg 1983;77(6): 773-4. PubMed
23. Radford SA, Johnson EM, Warnock DW. In Vitro Studies of Activity of Voriconazole (UK-109,496), a New Triazole Antifungal Agent, against Emerging and Less-Common Mold Pathogens. Antimicrobial Agents and Chemotherapy 1997; 41: 841-843. PubMed
24. Tagami H, Ohi M, Aoshima T, Moriguchi M, Suzuki N, Yamada M. Topical heat therapy for cutaneous chromomycosis. Arch Dermatol 1979;115(6): 740-1. PubMed
25. Tagami H, Ginoza M, Imaizumi S, Urano-Suehisa S. Successful treatment of chromoblastomycosis with topical heat therapy. J Am Acad Dermatol 1984;10(4): 615-9. PubMed
26. Bonifaz A, Martinez-Soto E, Carrasco-Gerard E, Peniche J. Treatment of chromoblastomycosis with itraconazole, cryosurgery and a combination of both. Int J Dermatol 1997;36: 542-547. PubMed
27. Kuttner BJ, Siegle RJ. Treatment of chromomycosis with a CO2 laser. J Dermatol Surg Oncol 1986;12(9): 965-8. PubMed
28. Hira K, Yamada H, Takahashi Y, Ogawa H. Successful treatment of chromomycosis using carbon dioxide laser associated with topical heat applications. J Eur Acad Dermatol Venereol. 2002 May;16(3): 273-5. PubMed
29. Kullavanijaya P, Rojanavanich V. Successful treatment of chromoblastomycosis due to Fonsecaea pedrosoi by the combination of itraconazole and cryotherapy. Int J Dermatol 1995;34(11): 804-7. PubMed
© 2008 Dermatology Online Journal

