Papular mucinosis (discrete papular lichen myxedematosus)
Published Web Location
https://doi.org/10.5070/D34g03r972Main Content
Papular mucinosis (discrete papular lichen myxedematosus)
Jennifer Bragg MD, Anthony C Soldano MD, Jo-Ann M Latkowski MD
Dermatology Online Journal 14 (5): 14
Department of Dermatology, New York UniversityAbstract
A 56-year-old woman presented with small, skin-colored papules on the trunk and thighs. Histopathologic findings were consistent with papular mucinosis. Laboratory evaluation did not show an associated paraproteinemia. Treatment with topical glucocorticoids was unsuccessful. Papular mucinosis, also known as localized lichen myxedematosus, has been categorized into 4 subtypes. The discrete papular form, as seen in our patient, is characterized by a few to multiple, 2-5-mm, skin-colored, firm, waxy, dome-shaped papules on the trunk and proximal aspects of the extremities. By definition there is no associated paraproteinemia, but this form may be associated with human immunodeficiency virus infection. Focal or diffuse mucinous deposits are seen on histopathologic examination. The usual clinical course is slow cutaneous progression without spontaneous resolution. Treatment is empiric and is usually unsuccessful.
Clinical synopsis
The patient presented to NYU Dermatologic Associates for a second opinion regarding a previously biopsied skin eruption. The eruption began 3 years ago as barely palpable, asymptomatic papules under the left breast, which then spread to involve the chest and abdomen. More recently she also has noticed lesions on her upper thighs. Treatment with a topical glucocorticoid for 4 months had no effect. Past medical history includes hyperlipidemia. Her only medication is simvastatin. A review of systems was negative.
Physical examination
Numerous, 2-4-mm, skin-colored, firm, dome-shaped, waxy papules were scattered over the central chest, inframammary folds, abdomen, and upper anterior thighs. There was no involvement of the distal extremities or face. The patient is not obese.
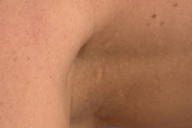 | 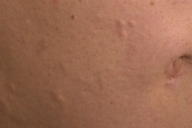 |
| Figure 1 | Figure 2 |
|---|
Laboratory data
A complete blood count, basic metabolic panel, liver function tests, and antinuclear antibody test were normal. A human immunodeficiency virus test was negative. The cholesterol and low-density lipoprotein levels were elevated (254 mg/dl and 158 mg/dl, respectively), but the high-density lipoprotein and triglyceride levels were normal. Thyroid stimulating hormone level was 4.90 uu/ml (normal range 0.30 to 4.70). Urine protein quantitative level was 6 mg/dl, and a random urine creatinine level was 106 mg/dl. Serum protein electrophoresis showed a slight increase in the beta region, but the immunofixation showed no qualitative abnormality.
Histopathology
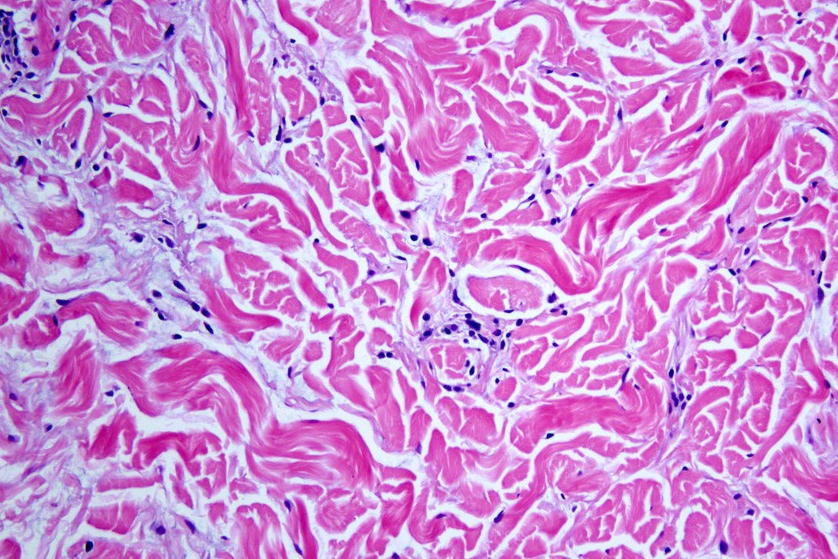 |
| Figure 3d |
|---|
There is a sparse, superficial and deep, perivascular lymphocytic infiltrate. There are increased spaces between dermal collagen fibers due to increased connective-tissue mucin deposition with focal areas of subtle increased number of fibroblasts. This mucin deposition is highlighted with a colloidal iron stain (not shown).
Comment
Papular mucinosis, also known as lichen myxedematosus (LM), was first described in 1906 [1]. In 1953, the condition was categorized into four subtypes: generalized lichenoid papular eruption, discrete papular form, localized-to-generalized lichenoid plaques, and urticarial plaques and nodular eruptions [2]. In 2001, the classification was revised into three subtypes: generalized papular and sclerodermoid (scleromyxedema), localized, and atypical forms. They subdivided the localized form into discrete papular LM, acral persistent papular mucinosis, juvenile and adult variants of self-healing papular mucinosis, papular mucinosis of infancy, and nodular LM (see table) [3]. In 2006, a self-healing variant was included in the discrete papular LM category [4]. According to these criteria, all forms require mucin deposition and the absence of thyroid disease. A diagnosis of the generalized form requires fibroblast proliferation and fibrosis on histopathologic examination and an associated monoclonal gammopathy. Diagnosis of the localized form requires the absence of a monoclonal gammopathy. The atypical subtypes include scleromyxedema without monoclonal gammopathy, localized LM with monoclonal gammopathy or systemic symptoms other than human immunodeficiency virus infection, localized LM with mixed features of different subtypes, and other not well-specified cases [3, 4]. All forms of lichen myxedematosus present with firm, dome-shaped, waxy papules, nodules, or plaques. Scleromyxedema, the most common form, is a widespread symmetric eruption that involves the face as well as the trunk and extremities. It is almost always associated with paraproteinemia and may be associated with other systemic disorders, which include multiple myeloma, lymphoma, and leukemia. Other organs may be involved, which include the muscles, joints, central and peripheral nervous system, lungs, and kidneys. Localized LM is confined to a few sites and is not associated with systemic findings, although a few cases have been reported in patients with human immunodeficiency virus infection (12 with discrete papular LM and two with the acral persistent subtype) [5, 6]. An association with obesity was noted in two cases, in which papules developed on the abdomen and resolved with subsequent weight loss [7].
Discrete papular LM is the most common form of the localized disease. Patients present with a variable number of asymptomatic, 2-5-mm papules that are distributed symmetrically over the trunk and limbs, with sparing of the face and distal aspects of the extremities. The underlying skin is not indurated. The lesions progress slowly to involve larger areas, do not progress to scleromyxedema, and do not resolve. Acral persistent papular mucinosis is characterized by multiple papules on the dorsal aspects of the hands. It is observed in women more frequently than it is in men. In papular mucinosis of infancy (also known as cutaneous mucinosis of infancy), the papules typically occur on the elbows and upper arms. In nodular LM, also known as atypical tuberous myxedema of Jadassohn-Dosseker, nodules rather than papules appear on the trunk and extremities. Our patient's findings meet the criteria for discrete papular LM except for a slightly elevated thyroid stimulating hormone level, which is probably unrelated.
Histopathologic findings in all forms of LM are characterized by dermal mucin comprised of acid glycosaminoglycans. The mucin in scleromyxedema is diffuse and is accompanied by collagen deposition and an increased number of large fibroblasts. In the discrete papular form, the mucin may be focal or diffuse, with no increase in collagen and fewer fibroblasts [8]. The etiology of LM has not been elucidated. It is thought to be a reactive process, but the initiating event and the factors involved are unclear. Paraproteins may play a role in the generalized form, but other triggers must be involved in the localized form. Other factors that might be involved include autoantibodies and cytokines, such as interleukin-1, tumor necrosis factors, and transforming growth factor fl [8].
Treatment for LM is based on anecdotal reports only. For scleromyxedema, treatments that have shown some clinical improvement in isolated cases include topical, intralesional, and systemic glucocorticoids; PUVA photochemotherapy; retinoids; plasmapheresis; extracorporeal photochemotherapy; dermabrasion; melphalan; granulocyte colony-stimulating factor; cyclosporine; and interferon alfa [3]. A recent case series reported a beneficial response to thalidomide in three patients [9]. For discrete papular LM which may be disfiguring but is not life-threatening, topical treatments can be tried but are usually unsuccessful. One patient was successfully treated with intralesional glucocorticoid injections and flurandrenolide-impregnated tape[10], and another responded to carbon dioxide laser and intralesional glucocorticoids [11]. Pimecrolimus ointment reduced pruritus in one patient, but the lesions persisted [12]. Some cases also resolve spontaneously (previously designated self-healing papular mucinosis) [3, 13]. Our patient did not respond to topical glucocorticoids, and intralesional injections are not feasible owing to the large number of lesions.
References
1. Dubreuilh W. Fibromes miliaries folliculaires: sclerodermie consecutive. Arch Dermatol Syphilol 1906;7:569 (as cited in Sulit DJ, et al. Discrete papular form of lichen myxedematosus: a case report and review of the literature. Cutis 2005;75:1052. Montgomery H, Underwood LJ. Lichen myxedematosus (differentiation from cutaneous myxedemas or mucoid states). J Invest Dermatol 1953;20:213
3. Rongioletti F, Rebora A. Updated classification of papular mucinosis, lichen myxedematosus, and scleromyxedema. J Am Acad Dermatol 2001;44:273
4. Rongioletti F. Lichen myxedematosus (papular mucinosis): new concepts and perspectives for an old disease. Semin Cutan Med Surg 2006;25:100
5. Rongioletti F, et al. Cutaneous mucinoses and HIV infection. Br J Dermatol 1998;139:1077
6. Depaire-Duclos F, et al. Papular mucinosis with rapid spontaneous regression in an HIV-infected patient. Eur J Dermatol 1998;8:353
7. Saez-Rodriguez M, et al. Localized lichen myxoedematosus (papular mucinosis) associated with morbid obesity: report of two cases. Br J Dermatol 2003;148:165
8. Rongioletti F, Rebora A. Cutaneous mucinoses: microscopic criteria for diagnosis. Am J Dermatopathol 2001;23:257
9. Sansbury JC, et al. Treatment of recalcitrant scleromyxedema with thalidomide in 3 patients. J Am Acad Dermatol 2004;51:126
10. Reynolds NJ, et al. Discrete papular mucinosis responding to intralesional and topical steroids. Arch Dermatol 1992;128:857
11. Kaymen AH, et al. The use of carbon dioxide laser in lichen myxedematosus. J Dermatol Surg Oncol 1989;15:862
12. Sulit DJ, et al. Discrete papular form of lichen myxedematosus: a case report and review of the literature. Cutis 2005;75:105
13. Sperber BR, et al. Self-healing papular mucinosis in an adult. J Am Acad Dermatol 2004;50:121
© 2008 Dermatology Online Journal

