Pruritic urticarial papules and plaques of pregnancy wholly abated with one week twice daily application of fluticasone propionate lotion: A case report and review of the literature
Published Web Location
https://doi.org/10.5070/D34cf4j596Main Content
Pruritic urticarial papules and plaques of pregnancy wholly abated with one week twice daily application of fluticasone propionate
lotion: A case report and review of the literature
Noah Scheinfeld MD
Dermatology Online Journal 14 (11): 4
Department of Dermatology, Columbia University, New York, NY. NSS32@columbia.eduAbstract
Pruritic urticarial papules and plaques of pregnancy (PUPPP) is among the most common dermatoses of pregnancy. Most reports of the effective treatment of PUPPP involve high potency topical corticosteroids or oral steroids. Many authorities have noted cases of PUPPP whose resolution followed parturition. A few have noted that PUPPP can arise and resolve the third trimester. A 36-year-old prima gravida at 38 weeks of gestation presented with a 2-week history of a pruritic papular abdominal eruption. She used fluticasone propionate 0.05 percent lotion twice a day. One week after starting this medication, the pruritus had resolved and the erythema/urticaria had abated; the pigmentary alteration had improved, but still remained. The PUPPP did not return after parturition. PUPPP can abate entirely during pregnancy. Fluticasone propionate 0.05 percent lotion, a class 5 (low-medium potency) corticosteroid, has a benign side effect profile and should be considered for the treatment of PUPPP during pregnancy.
Case
 |  |
| Figure 1 | Figure 2 |
|---|---|
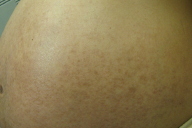
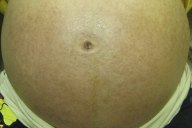
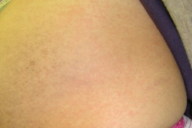
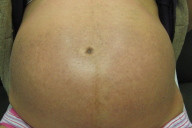
| Figure 1. Flank with urticarial plaques and erythema Figure 2. Abdomen and thighs with urticarial plaques and erythema | |
A 36-year-old Asian woman (G1, P0) presented at 38 weeks of gestation with a pruritic rash of 2 weeks duration. She stated the rash began on her abdomen and had progressed to her upper thighs over the previous week. Her pregnancy had been otherwise uneventful. She denied other systemic symptoms. Routine screening tests, including blood counts, human immunodeficiency virus (HIV), syphilis, and standard immune serologies were within normal limits. On the skin of her abdomen, numerous confluent, erythematous, urticarial macules and papules that coalesced into urticarial plaques were observed (Figs. 1 & 2). Striae were involved, but there was periumbilical sparing. The eruption also involved her upper thighs. She noted that the pruritus was preventing her from sleeping and that her obstetrician had not prescribed her any medication. She also noted that the papules and macules were stable and did not disappear in one location and appear in another. A putative diagnosis of pruritic, urticarial papules and plaques of pregnancy was made and the patient was prescribed fluticasone propionate lotion, 0.05 percent to be used twice-a-day. One week later, having used the medication as directed, the pruritus and erythema had abated altogether (Figs. 3 & 4), although brown, macules remained. The patient was told to stop using the fluticasone propionate lotion and one week later when the patient gave birth, the eruption had not returned. There was no recurrence post partum. Eight weeks post partum the pigmentary alteration began to fade.
Review
Pruritic urticarial papules and plaques of pregnancy (PUPPP) (known as the polymorphic eruption of pregnancy (PEP) in the United Kingdom), manifests as its names suggest with pruritic, polymorphic erythema, urticarial papules, vesicles, urticarial plaques, and/or erythema multiforme-like target eruptions. PUPPP is one of the most common dermatoses of pregnancy [1]. Aronson [1] divided PUPPP into three categories: (1) mainly urticarial papules and plaques, (2) non-urticarial erythema, papules, or vesicles, and (3) combinations of the type 1 and type 2. However, the success of particular therapies has not been related to the morphologic classifications.
The eruption of PUPPP most commonly manifests in the abdominal striae (periumbilical striae distensae) and can spread to the thighs. Less commonly, PUPPP manifests on the upper arms and even on the hands. One case described a patient with PUPP with extensive palmoplantar involvement [2]. Pruritic urticarial papules and plaques of pregnancy can involve the face.
Pruritic urticarial papules and plaques of pregnancy manifests in women and only occurs during and after the time of pregnancy. The group that accounts for most cases of PUPPP is primagravidas late in their third trimesters. PUPP is reported to develop in 0.5 percent of pregnancies; It can occur in women in the immediate postpartum period and last for up to about 6 weeks [3]. Pruritic urticarial papules and plaques of pregnancy uncommonly manifests after the first pregnancy (5-10%). A case report regarding PUPPP noted a case in a neonate and his mother but this report has not been widely noted [4].
The prognosis in terms of long-term morbidity and mortality of PUPPP are excellent. Pruritic urticarial papules and plaques of pregnancy generally appears to be unassociated with fetal or maternal morbidity and mortality. When PUPPP resolves, even in dark skinned patients, seldom is there any lasting, post-inflammatory pigment change or scarring of the skin.
What are its symptoms?
The primary symptom of PUPPP is pruritus. Occasionally, the pruritus is not striking. Usually, however, the itch of PUPPP is very unpleasant (even unbearable) for patients and can interrupt their sleep and their lives during a time when they are already uncomfortable as they approach parturition.
Correlating factors?
The factors that correlate with PUPPP have been defined in part, but remain controversial. One commentator has stated that PUPPP is not related to maternal or fetal weight gain [5]. A French case-control study of 40 patients with PUPPP and 160 normal controls found that PUPPP had associations with multiple gestations, cesarean deliveries, and male fetuses, but no relationship to maternal or fetal weight gain [6]. An Israeli study linked PUPP to multiple pregnancies, hypertensive disorders, and induction of labor [7]. An English study found a 2:1 male:female fetus ratio in cases of PUPPP [8]. A study of 181 European patients found that multiple gestation pregnancies and excessive maternal weight gain were linked to PUPPP. However, fetal weight and sex, were not linked to PUPPP [9].
Clinical findings can help to distinguish PUPPP from other dermatoses of pregnancy. Despite PUPPP's pruritus, patients seldom manifest linear crusted erosions, which are the sign of excoriative scratching. This is in contrast to patients with cholestasis of pregnancy who commonly are found to have linear erosions secondary to scratching. Pruritic urticarial papules and plaques of pregnancy typically spares the periumbilical area, unlike herpes (pemphigoid) gestationis which typically involves the periumbilical area.
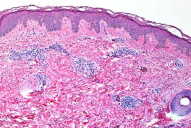
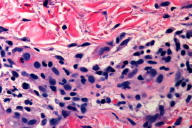
Histopathologic changes of PUPPP vary. Histologically, PUPPP demonstrates negative immunofluorescent studies and a mild lymphohistiocytic perivascular and interstitial infiltrate with eosinophils of variable quantities. There can be foci of spongiosis, slight upper epidermal edema, and occasionally, parakeratosis (Figs. 5 & 6). Although the infiltrate of insect bites can be wedge-shaped, the infiltrate of PUPPP is more diffuse. Urticaria spares the epidermis and can manifest with neutrophils.
Treatment of PUPPP
Many authorities and observers suggest a patient with PUPPP must typically wait for the end of the pregnancy for their PUPPP to resolve. One review even noted that PUPPP's regression can be delayed for up to 6 weeks postpartum [10]. That is, even delivery can fail to provide relief of symptoms immediately [11]. One commentator stated, in their experience, that PUPPP is self-limited, ending 4 weeks after it begins [12]. Callen [13] noted in 5/15 cases, PUPPP abated before the end of pregnancy. It seems likely that the resolution of PUPPP can be variable and may both antedate and post-date the end of pregnancy.
Medical therapy plays a role in the treatment of PUPPP. High potency topical or oral corticosteroids have been advocated to abate the eruption of PUPPP and were effective in 22/25 cases reported [14]. Several reports have noted the effectiveness of oral corticosteroids [15]. One report noted that PUPPP did not respond to oral corticosteroids in 1/5 cases [16]. Other writers have noted that antihistamines in combination with topical corticosteroids can help patients with PUPPP [17].
Delivery can put a patient with PUPPP on the path to recovery. One review noted that most of the "lesions [of PUPPP] spontaneously resolve after parturition" [18]. Some have even claimed that to control PUPPP labor had to be induced and the fetus delivered (although, induction according to one textbook of dermatological therapeutics, it is a disfavored treatment modality) [19].
The utility of low and medium potency steroids in the treatment of PUPPP is less clear. One report noted "low potency topical steroid cream, oral diphenhydramine, cool soaks, and emollients" failed to control a case of PUPPP that was only controlled with oral and intravenous corticosteroids and delivery [20]. One review of PUPPP has suggested that the symptoms of PUPPP can be controlled by betamethasone valerate 0.1 percent cream (a class 4 steroid), but no mention of clearing was mentioned in this review [21].
Fluticasone propionate 0.05 percent lotion is a treatment alternative that obstetricians and obstetrical dermatologists should consider adding to their armentarium. Fluticasone propionate 0.05 percent lotion is a class 5 (low-medium potency) corticosteroid and is indicated by the FDA for use in children as young as three months for atopic dermatitis [22]. Fluticasone propionate 0.05 percent lotion has not been shown to cause adrenal suppression with short-term use [23]. Fluticasone propionate lotion is one of the newer types of topical corticosteroid preparations and has high topical anti-inflammatory effects as well as a low potential to cause adverse effects because of low systemic absorption, rapid metabolism, and rapid clearance. Fluticasone propionate does not cross the placenta. Fluticasone propionate 0.05 percent lotion can be refrigerated, adding to its antipruritic qualities, and it is easy to smooth on large surfaces such as the distended abdomen of a 38-week pregnant woman.
Conclusion
The skin care of women during pregnancy is a complex matter. It is important to utilize therapies with the most benign side effect profile possible to safeguard mother and fetus. If indeed fluticasone propionate 0.05 percent lotion can reproducibly be shown to clear PUPPP, it could become another and an important treatment option considered for PUPPP.
References
1. Aronson IK, Bond S, Fiedler VC, Vomvouras S, Gruber D, Ruiz C. Pruritic urticarial papules and plaques of pregnancy: clinical and immunopathologic observations in 57 patients. J Am Acad Dermatol 1998;39:933-9. PubMed2. High WA, Hoang MP, Miller MD. Pruritic urticarial papules and plaques of pregnancy with unusual and extensive palmoplantar involvement. Obstet Gynecol. 2005;105:1261-4. PubMed
3. Buccolo LS, Viera AJ. Pruritic urticarial papules and plaques of pregnancy presenting in the postpartum period: a case report. J Reprod Med. 2005;50:61-3. PubMed
4. Uhlin SR. Pruritic urticarial papules and plaques of pregnancy. Involvement in mother and infant. Arch Dermatol. 1981;117:238-9. PubMed
5. Roger D, Vaillant L, Lorette G. Pruritic urticarial papules and plaques of pregnancy are not related to maternal or fetal weight gain. Arch Dermatol. 1990;126:1517. PubMed
6. Regnier S, Fermand V, Levy P, Uzan S, Aractingi S. A case-control study of polymorphic eruption of pregnancy. J Am Acad Dermatol. 2008;58:63-7. PubMed
7. Ohel I, Levy A, Silberstein T, Holcberg G, Sheiner E. Pregnancy outcome of patients with pruritic urticarial papules and plaques of pregnancy. J Matern Fetal Neonatal Med. 2006;19:305-8. PubMed
8. Vaughan Jones SA, Hern S, Nelson-Piercy C, Seed PT, Black MM. A prospective study of 200 women with dermatoses of pregnancy correlating clinical findings with hormonal and immunopathological profiles. Br J Dermatol. 1999;141:71-81. PubMed
9. Rudolph CM, Al-Fares S, Vaughan-Jones SA, Müllegger RR, Kerl H, Black MM. Polymorphic eruption of pregnancy: clinicopathology and potential trigger factors in 181 patients. Br J Dermatol. 2006;154:54-60. PubMed
10. Brzoza Z, Kasperska-Zajac A, Oleś E, Rogala B. Pruritic urticarial papules and plaques of pregnancy. J Midwifery Womens Health. 2007;52:44-8. PubMed
11. Matz H, Orion E, Wolf R. Pruritic urticarial papules and plaques of pregnancy: polymorphic eruption of pregnancy (PUPPP). Clin Dermatol. 2006;24:105-8. PubMed
12. Carruthers A. Pruritic urticarial papules and plaques of pregnancy. J Am Acad Dermatol. 1993;29:125. PubMed
13. Callen JP, Hanno R. Pruritic urticarial papules and plaques of pregnancy (PUPPP). A clinicopathologic study. J Am Acad Dermatol. 1981;5:401-5. PubMed
14. Yancey KB, Hall RP, Lawley TJ. Pruritic urticarial papules and plaques of pregnancy. Clinical experience in twenty-five patients, J Am Acad Dermatol 1984;10:473-480. PubMed
15. Lawley TJ, Hertz KC, Wade TR, Ackerman AB, Katz SI. Pruritic urticarial papules and plaques of pregnancy. JAMA. 1979;241:1696-9. PubMed
16. Cooper AJ, Fryer JA. Prurigo of late pregnancy. Australas J Dermatol. 1980;21:79-84. PubMed
17. Ahmed AR, Kaplan R. Pruritic urticarial papules and plaques of pregnancy. J Am Acad Dermatol. 1981;4:679-81. PubMed
18. Sherard GB 3rd, Atkinson SM Jr. Focus on primary care: pruritic dermatological conditions in pregnancy. Obstet Gynecol Surv. 2001;56:427-32. PubMed
19. Jurecka W. Pregnancy Dermatoses. In: Treatment of Skin Disease. 2nd Ed Lebwohl M, Heymann WR, Berth-Jones J, Coulson I eds. London, England: Mosby; 2006.
20. DE de Gaetano, HM de Gaetano. Pruritic urticarial papules and plaques of pregnancy: an unusual case. J Am Osteopath Assoc. 2002;102:44-6. available at www.jaoa.org/cgi/reprint/102/1/44
21. Ahmadi S, Powell FC. Pruritic urticarial papules and plaques of pregnancy: current status. Australas J Dermatol. 2005;46:53-9. PubMed
22. Eichenfield LF, Miller BH; Cutivate Lotion Study Group. Two randomized, double-blind, placebo-controlled studies of fluticasone propionate lotion 0.05% for the treatment of atopic dermatitis in subjects from 3 months of age. J Am Acad Dermatol. 2006;54:715-7. PubMed
23. Hebert AA, Friedlander SF, Allen DB. Topical fluticasone propionate lotion does not cause HPA axis suppression. J Pediatr. 2006 ;149:378-82. PubMed
© 2008 Dermatology Online Journal