Oral melanosis after tyrosine kinase inhibition with Imatinib for chronic myelogenous leukemia: Report of a case and review of the literature
Published Web Location
https://doi.org/10.5070/D3498194bxMain Content
Oral melanosis after tyrosine kinase inhibition with Imatinib for chronic myelogenous leukemia: Report of a case and review
of the literature
Michelle Wong DDS1, Shachar Sade MD FRCPC2, Melanie Gilbert DMD1, Hagen B E Klieb DMD MSc FRCDC1,2
Dermatology Online Journal 17 (5): 4
1. University of Toronto, Sunnybrook Health Sciences Center, Department of Dentistry2. University of Toronto, Sunnybrook Health Sciences Center, Department of Anatomic Pathology. hagen.klieb@sunnybrook.ca
Abstract
Dermatologic manifestations from therapy with imatinib are well known and frequently include hypopigmentation, and less commonly, hyperpigmentation. There have been few reports of oral hyperpigmentation. We present a case of palatal melanosis related to imatinib therapy for chronic myelogenous leukemia. This case is reported to add to the sparse literature concerning mucosal reactions related to this medication.
Introduction
Imatinib mesylate (Gleevec®) is a tyrosine kinase inhibitor that targets the constitutively active BCR-ABL protein and c-Kit. It is an important component of treatment for Philadelphia chromosome-positive chronic myelogenous leukemia and gastrointestinal stromal tumors. Dermatologic side effects are well known and diverse but hypopigmentation is a frequent event [1]. Cutaneous hyperpigmentation, on the contrary, appears to be a much rarer phenomenon. We present a case of palatal hyperpigmentation after therapy with imatinib. The differential diagnosis for diffuse oral melanosis is discussed. Whereas a diverse array of disease processes must be considered, we suggest clinicians recognize oral melanosis related to imatinib therapy.
Case report
 |
| Figure 1 |
|---|
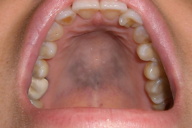
| Figure 1. Initial presentation of the patient with diffuse slate-gray discoloration of the hard palate |
A 43-year-old woman was referred by her dentist for assessment of asymptomatic palatal pigmentation that was identified approximately three-months following imatinib therapy for chronic myeloid leukemia (CML). No such pigmentation was identified prior to onset of therapy. She was otherwise healthy and did not smoke cigarettes. There was no history of palatal trauma or chronic irritation. She had used no other medication; medications associated with oral melanosis were specifically denied. Hyper- or hypo-pigmentation affecting other muco-cutaneous sites was also denied.
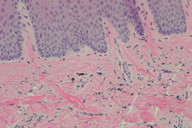
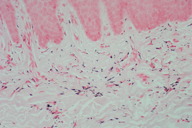
On examination, there was no palpable cervical lymphadenopathy. There was a diffuse slate-grey discoloration affecting the hard palate (Figure 1). A palatal biopsy was performed and showed fine, brown, granular particles within the superficial lamina propria consistent with melanin (Figure 2); this was confirmed on Fontana-Masson staining (Figure 3). There was no significant inflammation or hemorrhage and Prussian blue staining for iron was negative. Immunohistochemistry for c-Kit (CD117) showed focal reactivity of mesenchymal cells. The epithelium was unremarkable and there was no melanosis in the basal cell layer.
These findings were consistent with imatinib-induced melanosis. Neither active treatment nor modification of her current medication was recommended.
Discussion
Diagnostic considerations for oral melanosis are diverse and include physiologic pigmentation, systemic diseases (Addison disease, hemochromatosis), post-traumatic or inflammatory pigmentation, smoking-related pigmentation, as well as melanocytic neoplasms and Kaposi Sarcoma. Hereditary conditions to consider, albeit rarely, are Peutz-Jegher Syndrome, neurofibromatosis, and McCune-Albright Syndrome. Ecchymosis could also be considered for a patient with a myeloproliferative disorder.
Numerous medications can induce oral hyperpigmentation, including antibiotics (tetracycline), antimalarials, chemotherapeutics, hormones, and tranquilizers. The mechanism is variable and may be related to melanin deposition, pigmented medication metabolites, chelation of hemosiderin, and synthesis of other pigments [2].
Imatinib may be associated with cutaneous depigmentation although hyperpigmentation has also been described [1, 3, 4, 5]. Arora et al. found that in CML patients, 40.9 percent developed localized or generalized cutaneous depigmentation, whereas only 3.6 percent developed hyperpigmentation [4]. The pigmentation effects appear to be dose-related and reversible with discontinuation [1].
The postulated mechanisms of action of imatinib-induced cutaneous changes has been discussed elsewhere [1]. Effects on c-Kit are thought to be relevant as this receptor tyrosine kinase has regulatory roles in melanogenesis, melanocyte homeostasis, and pigmentation [1, 9]. Imatinib causes diffuse overstimulation of melanogenesis affecting muco-cutaneous sites, but the paradoxical association with both hypo- and hyper-pigmentation is difficult to explain. There is speculation that the medication may bind to different receptors in the skin that may either activate or inhibit melanogensis [1].
Oral manifestations from imatinib therapy are distinctly uncommon. Lichenoid reactions have been described [6] and there have been a few reports of solitary bluish-brown pigmentation affecting the hard palate [7, 8]. Hyperpigmentation has also been reported to affect the gingiva, perioral region, and teeth [9, 10]. c-Kit signalling may play a role in oral hyperpigmentation, and indeed, c-Kit is widely expressed and has been identified in mesenchymal cells of the oral cavity including the dental pulp [11].
Reports of oral melanosis related to imatinib therapy are rare but it is possible this is an underreported phenomenon given that the oral soft-tissues are less amenable to observation. Recognition, however, is important given the diverse differential diagnosis for oral melanosis.
References
1. Alexandrescu, D. T., Constatin, C. A., Farzanmehr, H., & Kauffman, C. L. (2008). Persistent cutaneous hyperpigmentation after tyrosine kinase inhibition with imatinib for GIST. Dermatology Online Journal , 14 (7), 7. [PubMed]2. Lerman MA, Karimbux N, Guze KA, Woo SB. (2009). Pigmentation of the hard palate. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. Jan;107(1):8-12. [PubMed]
3. McPherson T, Sherman V, & Turner R. (2009). Imatinib-associated hyperpigmentation, a side effect that should be recognized. Journal of European Academy of Dermatology and Venereology, 23 (1), 82-83. [PubMed]
4. Arora, B., Kumar, L., Sharma, A., Wadhwa, J., & Kochupillai, V. (2004). Pigmentary changes in chronic myeloid leukemia patients treated with imatinib mesylate. Annals of Oncology , 15 (2), 358-359. [PubMed]
5. Hasan, S., Dinh, K., Lombardo, F., Dawkins, F., & Kark, J. (2003). Hypopigmentation in an African patient treated with imatinib mesylate: A case report. Journal of National Medical Assocation , 95 (8), 722-724. [PubMed]
6. Ena, P., Chiarolini, F., Siddi, G. M., & Cossu, A. (2004). Oral lichenoid eruption secondary to imatinib (Glivec). Journal of Dermatological Treatment, 15, 253-255. [PubMed]
7. Lewis, D. M. (2009). Diffuse pigmentation of the palate. Oklahoma Dental Association Journal, 100 (8), 24-25. [PubMed]
8. Mattsson U, Halbritter S, Serikoff EM, Christerson L, Warfvinge G. (2011) Oral pigmentation in the hard palate associated with imatinib mesylate therapy: A report of three cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. May;111(5):e12-6 [PubMed]
9. Basso, F. G., Boer, C. C., Torrazan, M., Cintra, M. L., de Magahles, M. H., da Silva Santos, P., et al. (2009). Skin and oral lesions associated to imatinib mesylate therapy. Support Care Cancer , 17, 465-468. [PubMed]
10. Singh, N., & Bakhshi, S. (2007). Imatinib-induced dental hyperpigmentation in childhood chronic myeloid leukemia. Journal of Pediatric Hematology/Oncology, 29 (3), 208-209. [PubMed]
11. Gagari E, Rand MK, Tayari L, Vastardis H, Sharma P, Hauschka PV, Damoulis PD. (2006). Expression of stem cell factor and its receptor, c-kit, in human oral mesenchymal cells. Eur J Oral Sci. Oct;114(5):409-15. [PubMed]
© 2011 Dermatology Online Journal