Vesiculo-bullous dermatomyositis: Report of three cases
Published Web Location
https://doi.org/10.5070/D3492472wjMain Content
Vesiculo-bullous dermatomyositis: Report of three cases
Katsuyuki Nishigori, Toshiyuki Yamamoto, Hiroo Yokozeki
Dermatology Online Journal 15 (4): 6
Department of Dermatology, Tokyo Medical and Dental University, Tokyo, Japan. toyamade@fmu.ac.jpAbstract
Dermatomyositis (DM) presents with various types of skin lesions, but vesicle formation is rare. We herein present three female patients with DM who formed vesicles on the trunk and extremities. In all cases the bullous lesions appeared in parallel with increased disease activity of DM. In one case, only one large bulla within an erythematous background was found on the abdomen; the other two cases presented with a number of vesicles. Histological examination revealed marked subepidermal edema with mucin deposition in the upper dermis in all cases. Breast carcinoma and malignant lymphoma were associated in one case each. However, investigations did not reveal any malignancies in the third case. Prior reports suggest that internal malignancy is much higher in patients with vesiculo-bullous DM than in those unassociated with bullous lesions. Clinicians should be aware of vesiculo-bullous type DM.
Introduction
Dermatomyositis (DM) is an autoimmune inflammatory disease involving the skin and the muscle. The characteristic cutaneous manifestations of DM include an eyelid eruption described as heliotrope and Gottron papules on the dorsal fingers. In addition, periungual telangiectasia, poikiloderma, subcutaneous calcinosis, panniculitis, mucinosis, seborrheic dermatitis-like facial erythema, and blister formation are occasionally or less frequently seen. Several cases of vesiculo-bullous DM have been reported in the English literature [1, 2, 3, 4, 5]. We describe herein three patients with DM who developed vesicles and/or bullae during the course. All the patients were females; breast cancer and malignant lymphoma was associated in one case each.
Cinical synopsis
 |
| Figure 1 |
|---|
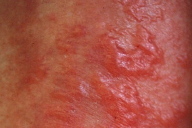
| Figure 1. Close-up view of the bullae in Case 1 |
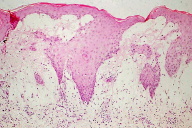
Case 1. A 55-year-old woman had noticed tenderness of the breast for four years prior to presentation and was diagnosed with breast cancer one month earlier. Also, she recognized muscle weakness of the upper limbs these 6 months. Itchy erythema appeared on her face two months before, which gradually exacerbated to involve her trunk and extremities. On physical examination, edematous erythema on the upper eyelids and Gottron papules on the fingers were observed. A number of red papules were noted along with linear erythema on her back. Proximal muscle weakness in her upper limbs was present. Laboratory findings revealed elevation in creatine kinase (CK) level (462 IU/l; normal <200 IU/l), aldolase level (7.8 U/l; normal <5.0 U/l), and erythrocyte sedimentation rate (ESR) (33 mm/hr). She had a positive antinuclear antibody (1:160, homogeneous & speckled). Magnetic resonance imaging examination of her proximal muscles of upper limbs showed a diffuse inflammatory reaction; electromyogram of her left deltoid muscle showed myopathic changes and a muscle biopsy exhibited mild cellular infiltrates. She was diagnosed with dermatomyositis. Because she was planning to have surgery for her breast cancer, systemic administration of prednisolone was postponed. Just after her breast operation, a number of bean-sized, tense vesicles appeared on a diffuse erythematous background on the forearms and thighs (Fig. 1). A biopsy specimen revealed subepidermal vesicles, edema and perivascular infiltration of mononuclear cells in the upper dermis (Fig. 2).
 |
| Figure 2 |
|---|
| Figure 2. Histology showing subepidermal bulla (H&E) |
Alcian blue and colloidal iron stain both showed positive findings in the upper dermis indicating the deposition of mucin. Direct immunofluorescence studies showed negative findings at the basement membrane zone. Although vesicle formation had decreased after the operation, she developed methicillin-resistant staphylococcus aureus (MRSA) sepsis originated from the surgical wound. Although the infection responded to vancomycin infusion therapy, muscle soreness and weakness got worse. Oral prednisolone (60 mg/day) was not effective, but steroid pulse therapy with methylprednisolone (1000 mg/day x 3 times) followed by oral prednisolone (80 mg/day) improved the myositis and cutaneous eruption. Thereafter, oral prednisolone was gradually tapered, without relapse over 2 years.
 |
| Figure 3 |
|---|
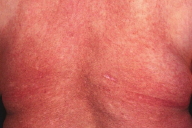
| Figure 3. Diffuse erythema on the trunk of Case 2 |
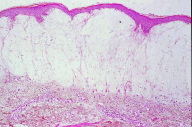
Case 2. A 55-year-old female suspected as having dermatomyositis was referred to our department for further investigation and treatment. On physical examination, she exhibited erythema of the eyelids, nail fold erythema, and diffuse erythema on the trunk (Fig. 3). Laboratory examination showed increased levels of ESR, CK, AST, ALT, lactate dehydrogenase (LDH), aldolase and myoglobin. Antinuclear antibody was positive (1:1280, discrete), but Jo-1, anti-SS-A, and SS-B antibodies were all negative. Examination by electromyography showed a myopathic pattern. A muscle biopsy from her left biceps showed cellular infiltrates between the muscle fibers and around the vessels. During admission, she developed bullae on erythema on the abdomen. A biopsy revealed subepidermal vesicles, marked edema of the upper dermis, and mild infiltration of inflammatory cells (Fig 4a). Colloidal iron and alcian blue stains revealed positive findings for mucin (Fig. 4b). Direct immunofluorescence studies showed negative findings. She was effectively treated with oral prednisolone. She has been followed up for over 10 years, but workup showed neither lung fibrosis nor internal malignancy.
 |  |
| Figure 4a | Figure 4b |
|---|---|
| Figure 4a. Subepidermal bulla (H&E) Figure 4b. Mucin deposition below the bulla (colloidal iron stain) | |
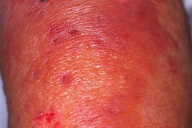 |
| Figure 5 |
|---|
| Figure 5. Edematous erythema, bulla, and erosions on the upper extremities |
Case 3. A 63-year-old female was referred to our hospital, complaining of general fatigue and prominent muscle weakness which appeared 3 months before. She had been diagnosed with malignant lymphoma 5 years before and had been treated with chemotherapy. Thereafter, she was in remission for three years. Laboratory examination revealed increased ESR levels. Muscle enzyme levels were elevated: LDH; 1761 IU/l (normal <225 IU/l), CK; 4485 IU/l, aldolase; 15.9 U/l, myoglobin 270 ng/ml (normal <60 ng/ml). Antinuclear antibody was 1:40 (homogeneous & speckled), but anti-Jo-1 antibody was negative. An electromyogram of the deltoid muscle showed a myogenic pattern. A muscle biopsy specimen showed cellular infiltrates of mononuclear cells. She was referred to our department for cutaneous manifestations. On physical examination, she exhibited diffuse erythema on her back. Both forearms were lichenified. Symptoms of heliotrope rash on the eyelids, Gottron papules, and nail bed erythema were not present. In addition, several small vesicles and erosions were found on the lateral aspects of the upper extremities (Fig. 5). A skin biopsy revealed a subepidermal vesiculation. Colloidal iron stain was positive for mucin in the upper dermis. Direct immunofluorescence studies were negative. She was successfully treated with an initial dose of oral prednisolone at 60 mg/day; 4 months later she was under remission. She has had no relapse of her lymphoma.
Discussion
DM presents various skin lesions; however, vesicle formation is rare. In this article, we report three female patients with vesiculo-bullous DM. The histology in all three women showed dermal mucin and subepidermal bullae. In Case 1, a relatively large bulla was noted on the abdomen and in Case 2, small vesicles were limited to the upper limbs. On the contrary, a number of vesicles and bullae were diffusely noted in Case 3. Thus, clinical variations of bullous lesions in association with DM are demonstrated. As was reported by Kubo et al. [1], vesicle formation is occasionally found, especially in Japanese DM patients.
The incidence of internal malignancy is reported to be much higher in patients with vesiculo-bullous DM than in those unassociated with bullous lesions [1]. In our Case 1, vesicle formation rapidly progressed involving most areas of the trunk and extremities at the time of her diagnosis of breast cancer. According to the report by Kubo et al [1], 19 cases were reviewed, of whom 6 cases were males and 13 cases were females. All except for one case had subepidermal bullae; intraepidermal bullae were recognized in one case. Our three cases had subepidermal bullae and had mucin deposition in the upper dermis. Marked subepidermal edema, mucin deposition and mechanical stress have been cited as major causes of subepidermal bulla. Our cases also suggest that marked mucin deposition and edema may lead to the formation of vesicles/bullae. In one of our two cases associated with malignancy, vesicles appeared at a time that correlated with the diagnosis of the underlying malignancy. In contrast, the other case developed a number of vesicles in spite of the fact that her malignant lymphoma was in remission; further investigations did not reveal other malignancies. Clinicians should be aware of the vesiculo-bullous type of DM.
Other vesicular diseases such as bullous lupus erythematosus, bullous pemphigoid, or dermatitis herpetiformis should be ruled out. There have been two reports in the literature documenting the coexistence of DM and bullous pemphigoid [6, 7]. The negative direct immunofluorescence tests in our patients aided in the exclusion of these other bullous entities.
References
1. Kubo M, Sato S, Kitahara H, Tsuchida T, Tamaki K. Vesicle formation in dermatomyositis associated with gynecologic malignancies. J Am Acad Dermatol 1996; 34: 391-4. [PubMed]2. McCollough ML, Cockerell CJ. Vesiculo-bullous dermatomyositis. Am J Dermatopathol 1998; 20: 170-4. [PubMed]
3. Glover M, Leigh I. Dermatomyositis pemphigoides: a case with coexistent dermatomyositis and bullous pemphigoid. J Am Acad Dermatol 1992; 27: 849-52. [PubMed]
4. Fujimoto M, Murakami T, Murata S, Ohtsuki M, Kawai T, Saitoh K, Nakagawa H. Acute onset vesiculo-bullous dermatomyositis associated with massive mucosal necrosis of the intestines. Clin Exp Dermatol 2002; 27: 718-20. [PubMed]
5. Zangrilli A, Papoutsaki M, Bianchi L, Teoli M, Chimenti S. Bullous dermatomyositis: A marker of poor prognosis and aggressive internal malignancy? Acta Derm Venereol 2008; 88: 393-4. [PubMed]
6. Ang P, Sugeng MW, Chua SH. Bullous dermatomyositis associated with nasopharyngeal carcinoma-A case report. Ann Acad Med Singapore 1999; 28: 855-7. [PubMed]
7. Tsukada Y, Kawase MK, Murashima A, Kitahora T, Hasimoto T, Komai A . Bullous pemphigoid associated with dermatomyositis successfully controlled with minocycline. Clin Exp Dermatol 2003; 28: 563-4. [PubMed]
© 2009 Dermatology Online Journal

