White sponge nevus in a patient with EEC syndrome
Published Web Location
https://doi.org/10.5070/D3482528ntMain Content
White sponge nevus in a patient with EEC syndrome
Gisele Silva Dalben PhD, Helize A Cursino DDS, Bruno A Barbosa MSc, Beatriz Costa PhD, Alberto Consolaro PhD
Dermatology Online Journal 16 (5): 7
Hospital for Rehabilitation of Craniofacial Anomalies, University of São Paulo. gsdalben@usp.brAbstract
A 10-year-old boy, affected by cleft lip and palate and the Ectrodactyly-Ectodermal Dysplasia-Clefting (EEC) syndrome, presented with hypodontia, microdontia, enamel hypoplasia and caries, as well as white macules and plaques on the buccal mucosa bilaterally. Verrucous plaques were noted on the right side and a smooth diffuse white macule was present on the left side. Samples of each were taken by incisional biopsy. Microscopic analysis revealed oral mucosa comprised of hyperplastic stratified squamous epithelium with either hyperparakeratosis or lack of keratinization. There was extensive hydropic degeneration and sparse cells with perinuclear eosinophilic condensation were identified. The underlying fibrous connective tissue exhibited blood vessels with mild subepithelial mononuclear inflammatory infiltrate, bundles of skeletal striated muscle fibers, and areas of hemorrhage at deeper regions. White sponge nevus was diagnosed. To our knowledge, no other case of white sponge nevus has been reported in a patient with EEC syndrome. Considering the low prevalence of this disorder and also of the EEC syndrome, future studies on the etiology of the white sponge nevus might look at the common etiopathogenic aspects of both the white sponge nevus and the EEC syndrome.
Introduction
The clinical characteristics of the white sponge nevus were first described by Hyde in 1909, followed by Cannon in 1935, who discussed the clinical and histological aspects of the disorder and suggested the term “white sponge nevus” (WSN) [1].
The WSN is one among the several white lesions of the oral mucosa. It is an autosomal dominant disorder characterized by asymptomatic white spongy plaques of the oral mucosa; it may also affect the nasal, esophageal, laryngeal, vaginal, and anal mucosa. In the oral mucosa, the lesions are usually bilateral and located on the buccal mucosa, followed by the lips, tongue, alveolar mucosa, and mouth floor. It rarely affects the hard and soft palate and the tonsils. The plaques usually appear in the first decade of life and the condition affects both genders similarly. The size of lesions varies between individuals and at different periods in the same individual. The WSN lesions are benign and may alternate with periods of remission and exacerbation [1-7].
The differential diagnosis of WSN should include various other white lesions. Leukoplakia, chemical burns, trauma, tobacco use, and syphilis may occur in isolation and do not have a genetic etiology. The clinical and histological appearance of these are very different from the WSN; thus they are easily differentiated, especially when there is no family history and an associated environmental cause is reported. The WSN may also be confused with candidiasis. However, biopsy samples, cultures of microorganisms, and the response to antifungal drugs allow their differentiation. The lesions of morsicatio buccarum, pachyonychia congenita, hereditary benign intraepithelial dyskeratosis, Darier disease, congenital dyskeratosis, lichen planus, and lupus erythematosus may be similar to the lesions of WSN. Except for lichen planus and lupus erythematosus, which may be restricted to the oral cavity, the associated findings outside the oral cavity allow their differentiation from WSN [1, 2, 7, 8, 9].
Usually, there is no need for treatment of WSN because of the benign and asymptomatic nature [7, 9]. When requested by the patient, treatment with penicillin has been reported to produce remission of the lesions [2]; treatment with vitamins, anti-histaminic drugs, or mouthrinses have no benefit [2, 4].
The Ectrodactyly-Ectodermal Dysplasia-Clefting (EEC) syndrome, associated with the chromosomal region 7q11.2-q21.3, is characterized by malformation of the limbs with ectrodactyly (“lobster claw”), signs of ectodermal dysplasia (keratoconjunctivitis, lacrimation, photophobia, sparse hair), hypopigmentation of the hair and skin, cleft lip and/or palate in most cases, conductive hearing loss (in nearly 30 percent of cases) and possible mental retardation [10, 11, 12, 13]. Ectrodactyly affects the hands and feet in 90 percent of cases and may be detected prenatally by ultrasound examination [11, 12], yet milder alterations and even normal limbs have already been described [10]. Microcephaly and mental retardation may be present in a few cases [11, 12, 13]. The wide phenotypic variability highlights the need for a detailed investigation of mild signs in suspected cases. Cleft lip and palate frequently occurs in association with limb malformations, indicating that there may be common factors between the limbs and face during embryonic development [10].
This paper describes the occurrence of white sponge nevus in the buccal mucosa bilaterally in a patient with EEC syndrome.
Case report
 |  |
| Figure 1 | Figure 2 |
|---|
 |
| Figure 3 |
|---|
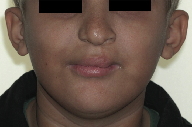
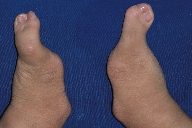
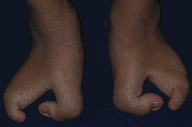
The 10-year-old boy was the second child of unrelated parents without a family history of malformations. The patient was delivered by an uneventful Cesarean section at term, weighing 3.165 kg and measuring 50 cm. The patient attended the Hospital for Rehabilitation of Craniofacial Anomalies, University of São Paulo (HRAC-USP), for the first time at the age of 3 months and 20 days. The physical examination revealed complete unilateral left cleft lip and palate, obstruction of the lacrimal duct, clear and sparse hair, dry hair and skin (Figure 1), oligodactyly of the hands bilaterally with syndactyly of the second and third fingers (Figure 2), and ectrodactyly of the feet bilaterally (Figure 3). The patient was examined by a geneticist and was diagnosed as having the EEC syndrome based on the clinical findings.
 |  |
| Figure 4a | Figure 4b |
|---|
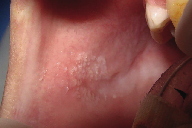
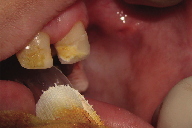
The intraoral examination at 10 years of age revealed hypodontia, microdontia, enamel hypoplasia and caries, and white lesions on the buccal mucosa bilaterally. There were verrucous plaques on the right side and a smooth diffuse white macule on the left side (Figures 4a and 4b). The mother reported that the child had the habit of biting the cheeks. No similar lesions were observed in other regions of the body.
Incisional biopsy was performed using a blade (elliptic incision on the left side) and punch (right side), under general anesthesia, together with secondary cheiloplasty. The suspected diagnoses were leukoedema, white sponge nevus, or morsicatio buccarum.
 |  |
| Figure 5 | Figure 6 |
|---|
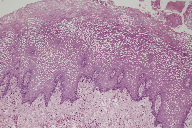
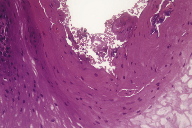
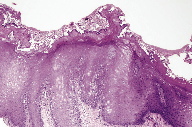
The microscopic diagnosis was white sponge nevus because of the presence of oral mucosa made up of hyperplastic stratified squamous epithelium with areas of hyperparakeratosis and non-keratinized areas, with extensive hydropic degeneration (Figure 5); some cells showed perinuclear eosinophilic condensation. Microbial biofilms were observed on the surface (Figure 6) and there was separation of the most superficial epithelial layers, compatible with morsicatio buccarum associated with the white sponge nevus (Figure 7). The underlying fibrous connective tissue exhibited blood vessels with mild subepithelial mononuclear inflammatory infiltrate, bundles of skeletal striated muscle fibers, and areas of hemorrhage at deeper regions (Figure 8).
 | 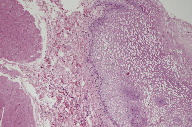 |
| Figure 7 | Figure 8 |
|---|
Discussion
Hypodontia, microdontia, enamel alterations, and taurodontism are commonly observed in patients with EEC syndrome [11, 13, 14, 15]. Other features include xerostomia, with possible atresia or agenesis of the parotid duct [11, 12]. The combination of these factors predisposes these patients to dental caries; thus, the need for excellent dental hygiene and regular follow-up should be emphasized; the prescription of artificial saliva may be needed. The high caries prevalence may lead to early tooth loss. When associated with cleft lip and palate, this may inhibit the growth of the middle facial third and require surgical correction. The need of orthognathic surgery seems to be more frequent compared to patients with non-syndromic cleft lip and palate [16].
Reports of malignant lesions in patients with EEC syndrome are rare. However, Balci et al. [17] published a report on a patient with EEC syndrome who presented with a malignant lymphoma with high expression of the gene p63, located at the chromosomal region 3q27, which led to death at 19 years of age.
It is known that mutations in the genes K4 and K13 (located on the chromosomal regions 12q13 and 17q21, respectively), which encode the mucosa-specific keratins, are the underlying cause of WSN [3, 8]. Even though the occurrence of white sponge nevus in this patient with EEC syndrome may be a coincidental finding, considering the aforementioned reports, future investigations or studies of case series using molecular biology techniques might elucidate the interaction between: 1. chromosomal regions 7q11.2-q21.3, involved in the EEC syndrome, 2. 3q27, related to lymphoma, and 3. 12q13 and 17q21, which encode the mucosa-specific keratins. It is known that the suprabasal keratinocytes of the buccal mucosa specifically express K4 and K13. A 3-base-pair deletion in the helix initiation peptide of K4 was identified by Rugg et al. [18] in affected members from two families presenting with white sponge nevus. Chao et al. [19] also reported a novel pathogenic mutation in this gene in an individual with white sponge nevus; the mutation could disrupt the stability of keratin filaments.
The WSN lesions in the present case did not detach upon scratching, in agreement with previous reports [7]. Despite this clinical aspect, WSN is frequently confused with candidiasis resistant to treatment. This difficulty may be related to the irregular surface of WSN, which favors secondary infection by yeasts [9].
Similar to previous reports, the surfaces of plaques were thick, corrugated, and soft. According to the literature, microscopically the plaques consist of parakeratinized epithelium with acanthosis, with alternate areas of hyperkatinization and non-keratinization. The cells in all epithelial layers may occasionally be vacuolated. The nuclei of cells are picnotic and the underlying connective tissue is normal. With regard to the pathogenesis, the epithelial cells do not desquamate, leading to epithelial hyperplasia. The underlying fibrous connective tissue is usually normal, without signs of inflammation and there are reports of spongiosis [1, 2, 4, 5, 6, 9]. Most of these characteristics were observed in this case, except for the presence of subepithelial inflammation. According to Jorgenson [2], the underlying fibrous connective tissue in WSN usually does not exhibit inflammatory infiltrate, unless the lesions are subjected to secondary trauma. In this case, considering the report of cheek biting, the subepithelial inflammation was probably related to the concomitant occurrence of morsicatio buccarum. This is a white area that results from repetitive, chronic frictional trauma, usually from raking of the teeth over the mucosa (morsus = bite) [20].
The WSN usually affects several individuals in a family, corroborating its autosomal dominant heredity [1, 2, 3, 4, 21]. However, in this case there was no history of other family members affected by WSN nor by the EEC syndrome.
To our knowledge, no other case of white sponge nevus in a patient with EEC syndrome has been reported so far. Considering the low prevalence of this disorder and also of the EEC syndrome, future studies on the etiology of the white sponge nevus might look at common etiopathogenic aspects of both the white sponge nevus and the EEC syndrome.
References
1. Morris R, Gansler TS, Rudisill MT, Neville B. White sponge nevus – diagnosis by light microscopic and ultrastructural cytology. Acta Cytol 1988; 32:357-61. [PubMed]2. Jorgenson RJ, Levin S. White sponge nevus. Arch Dermatol 1981; 117:73-6. [PubMed]
3. Terrinoni A, Rugg EL, Lane EB, Melino G, Felix DH, Munro CS, McLean WHI. A novel mutation in the keratin 13 gene causing oral white sponge nevus. J Dent Res 2001; 80:919-23. [PubMed]
4. Martelli Jr H, Pereira SM, Rocha TM, Santos PLAN, Paula AMB, Bonan RF. White sponge nevus: report of a three-generation family. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007; 103:43-7. [PubMed]
5. Martelli Jr H, Santos PLAN, Souza LN, Melo Filho MR, Paula AMB. Nevo branco esponjoso – relato de caso clínico e revisão da literatura. J Bras Clin Odontol Int 2006; 10:128-31. [PubMed]
6. Quintella C, Janson G, Azevedo LR, Damante JH. Orthodontic therapy in a patient with white sponge nevus. Am J Orthod Dentofacial Orthop 2004; 125:497-9. [PubMed]
7. Sambucety OS, López PM, Prieto MAR, Gónzalez IR, Fernández MM. Lesiones blanquecinas en la mucosa oral. An Esp Pediatr 2001; 55:159-60. [PubMed]
8. Terrinoni A, Candi E, Oddi S, Gobello T, Camaione DB, Mazzanti C, Zambruno G, Knight R, Melino G. A glutamine insertion in the 1A alpha helical domain of the keratin 4 gene in a familial case of white sponge nevus. J Invest Dermatol 2000; 114:388-91. [PubMed]
9. Happle R, Manegold HG. Weisser Schleimhautnävus. Hautarzt 1979; 30:30-2. [PubMed]
10. Annerén G, Andersson T, Lindgren PG, Kjartansson S. Ectrodactyly-ectodermal dysplasia-clefting syndrome (EEC): the clinical variation and prenatal diagnosis. Clin Genet 1991; 40:257-62. [PubMed]
11. Gorlin RJ, Cohen Jr MM, Hennekan RCM. Oral clefting syndromes. In: Gorlin RJ, Cohen Jr MM, Levin LS, editors. Syndromes of the head and neck. Oxford: University Press; 2001. p. 877-911.
12. Johns Hopkins University. Online Mendelian Inheritance in Man. MIM Number: 129900. Accessed Oct 2nd, 2009.
13. Buss PW, Hughes HE, Clarke A. Twenty-four cases of the EEC syndrome: clinical presentation and management. J Med Genet 1995; 32:716-23. [PubMed]
14. Tanboga I, Pince S, Duzdar L. Dental management of a child with EEC syndrome. Int J Paediatr Dent 1992; 2:99-103. [PubMed]
15. Ulukapi I, Bilgin T, Yalcin S. EEC syndrome (ectrodactyly-ectodermal dysplasia-clefting): a clinical case report. ASDC J Dent Child 2001; 68:350-2. [PubMed]
16. Birgfeld CB, Glick P, Singh D, LaRossa D, Bartlett S. Midface growth in patients with ectrodactyly-ectodermal dysplasia-clefting syndrome. Plast Reconstr Surg 2007; 120:144-10. [PubMed]
17. Balci S, Engiz O, Okten G, Sipahier M, Gursu G, Kandemir B. A 19-year follow-up of a patient with type 3 ectrodactyly-ectodermal dysplasia-clefting syndrome who developed non-Hodgkin syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009; 108:e91-5. [PubMed]
18. Rugg EL, McLean WHI, Allison WE, Lunny DP, Macleod RI, Felix DH, Lane EB, Munro CS. A mutation in the mucosal keratin K4 is associated with oral white sponge nevus. Nature Genet 1995; 11:450-2. [PubMed]
19. Chao SC, Tsai YM, Yang MH, Lee JYY. A novel mutation in the keratin 4 gene causing white sponge nevus. Br J Dermatol 2003; 148:1125-8. [PubMed]
20. Woo SB, Lin D. Morsicatio mucosae oris – a chronic oral frictional keratosis, not a leukoplakia. J Oral Maxillofac Surg 2009; 67:140-6. [PubMed]
21. Jornet PL. White sponge nevus: presentation of a new family. Ped Dermatol 2008; 25:116-7. [PubMed]
© 2010 Dermatology Online Journal

